Abstract
This work reports density functional theory calculations of the optimized geometries, molecular reactivity, energy gap, and thermodynamic properties of molecular dyes fluorescein (FS), fluorescein attached with methoxy (FSO), fluorescein attached with amine (FSA), fluorescein attached with methane (FSM), fluorescein attached with ethene (FSE), and fluorescein attached with thiophene (FST) using the hybrid functional B3LYP and 6-311G basis sets. When donating groups are attached to the molecular dye, the bond lengths are slightly decreased which is important for easy transfer of electron from donating to the accepting group. For all dyes, highest occupied molecular orbital/lowest occupied molecular orbital analysis results in positive outcomes upon electron injection to semiconductors and subsequent dye regeneration by the electrolyte. The ionization potential increases with increasing conjugation; therefore, the molecular dye attached to thiophene has the highest ionization potential. Meanwhile, a donating group with increased conjugation results in low electron affinity.
Keywords

Introduction
Fluorescein (FS) is a metal-free organic dye which is a dark orange/red powder and is slightly soluble in water and alcohol. The molecular formula of FS [
FS has absorption spectra in water and alcohol from 380 to 512 nm, and emits from 510 to 517 nm. 2 It has high molar absorptivity at the wavelength of an argon laser (488 nm). It has a large fluorescence quantum yield and high photostability, making it a very useful and sensitive fluorescent label. Commercially, many derivatives of FS are available such as fluorescein isothiocyanate and fluorescein succinimidyl ester. 3 FS in an aqueous solution occurs in cationic, neutral, anionic, and dianionic forms as displayed in Figure 1.
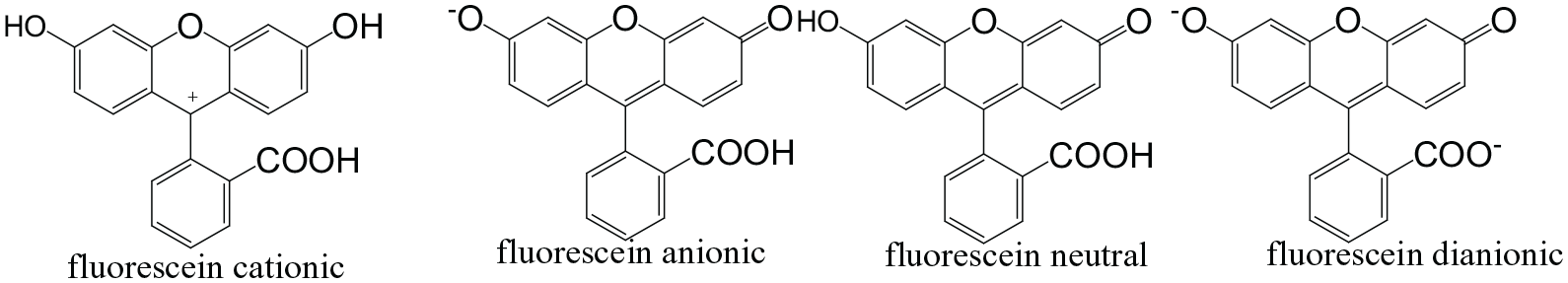
Different forms of fluorescein molecules.
When FS combines in acetonitrile solution, the absorption occurs at 460 nm, while emission occurs in the wavelength range of 500–540 nm. 4 FS has many applications such as in forensics and in serology to detect latent bloodstains. It is also used as a diagnostic tool in the field of ophthalmology and optometry. Topical FS is used in the diagnosis of corneal abrasions, corneal ulcers, and herpetic corneal infection. 5
Density functional theory (DFT) is an important method for the prediction of the thermodynamic properties and chemical reactivity of molecules. 6 Therefore, through applying the DFT method, several reactivity descriptors of FS derivatives can be discussed such as hardness, global softness, chemical potential, electronegativity, electrophilicity, electrofugality, nucleofugality, and other parameters of chemical reactivity.
Computational methods
The molecular structures were optimized in both gas and solvent phases by DFT using the B3LYP/6-311G basis sets. 7 All calculations were carried out using the Gaussian 09 software program.8,9 From these calculations, the optimized geometry and frequencies were also obtained. The calculations have been widely used to study different mechanism such as the reaction mechanism of the molecules. The visual representation of the densities of the highest occupied molecular orbitals (HOMOs) and the lowest occupied molecular orbitals (LUMOs) of all the studied molecules was accomplished with the Gaussian View 05 program. 10
Theoretical background
DFT is an important method for studying the theoretical chemical reactivity mechanisms of molecules.6,11 According to Koopmans’ theory, the ionization potential (IP) and electron affinity (EA) are the eigenvalues of HOMO and LUMO energy levels.11,12 Therefore, the IP and EA can be calculated through the following relations, respectively 13

Equation (1) is the basic equation for calculating different global chemical reactivity descriptors of the molecules like electronegativity (

where

Using Koopmans’ theory for closed-shell molecules, the electronegativity (

Also, the chemical potential (
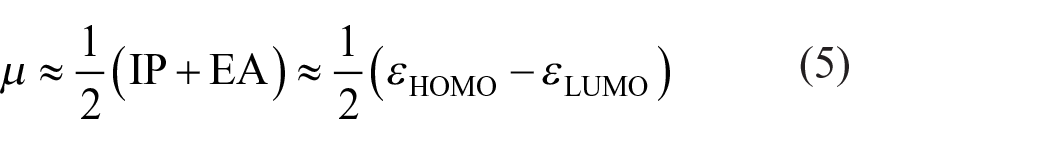
But the hardness (

where
As part of the calculations the eletrophilicity of the molecule can also be discussed. It is a descriptor of the reactivity that allows a quantitative classification of the global electrophilic nature of a molecule within a relative scale. In 2005, Parr et al. proposed that electrophilicity index as a measure of energy lowering due to the maximum of electron flow between the donor and acceptor. Therefore, the electrophilicity index can be defined through the following relation 15

where
The maximum amount of electron charge transfer (Δ

From equation (8), the maximum amount of electron transfer from the nucleophile to the electrophile can be evaluated for each studied molecule.
Finally, the nucleofugality (Δ


Results and discussion
Molecular geometry
All molecules were fully optimized by DFT using B3LYP with the 6-311G basis set. The optimization of the molecules was performed in the gaseous phase as well as in the solvent phase. Figure 2 shows the optimized structures of the studied molecules.

The optimized structures of the molecules calculated at the B3LYP/6-311G level.
After optimization, the bond lengths and bond angles were obtained (Table 1). Generally, the bond lengths of the studied molecules slightly decrease after the attachment of an electron-donating group. The decreasing of the bond length helps intramolecular charge transfer from the electron donor to the electron-accepting group. In fluorescein attached with amine (FSA), the C17–C18 bond length increased from 1.3966 to 1.4158 Å and that of other derivatives slightly increased as shown in Figure 3. Also, the C23–O25 bond length in FSA slightly increased in from 1.3735 to 1.3832 Å.
The bond lengths of the fluorescein derivatives determined.
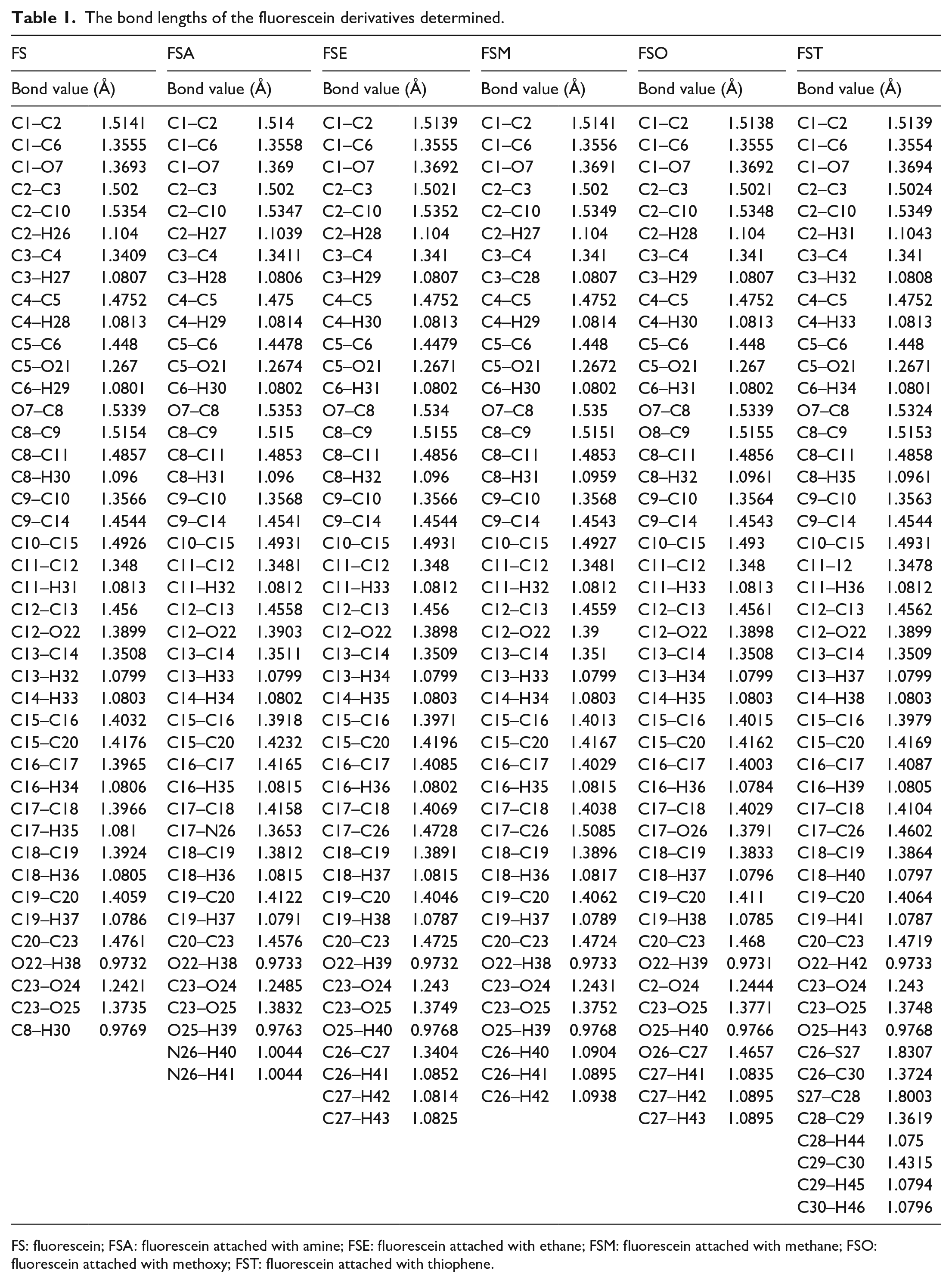
FS: fluorescein; FSA: fluorescein attached with amine; FSE: fluorescein attached with ethane; FSM: fluorescein attached with methane; FSO: fluorescein attached with methoxy; FST: fluorescein attached with thiophene.

Selected bond lengths of the studied dye molecules.
At the point of attachment between FS and the electron-donating groups, the bond length values increased as indicated in Figure 4. The bonds formed in the FS derivatives (FSA, fluorescein attached with ethene (FSE), fluorescein attached with methane (FSM), fluorescein attached with methoxy (FSO), and fluorescein attached with thiophene (FST)) after addition of the substituent group are C17–N26, C17–C26, C17–C26, C17–O26, and C17–C26 and their values are 1.3653, 1.4728, 1.5085, 1.3791, and 1.4602 Å, respectively. FSE and FSM have the same bonds, but their values are different because FSE contains π-bond after C26 and FSM has no π-bond after C26. Thus, increasing of the π-conjugate spacer slightly decreases the bond length.

The bond lengths at the attachment point between the fluorescein and the donor group.
The HOMO/LUMO energies and energy gap (∆Eg )
The HOMO energy actually determines the ability of the molecule to donate an electron. In addition, the higher the value of the HOMO energy, the higher is the electron donation ability to the acceptor. But, the LUMO energy of the molecule indicates the ability to accept an electron. Therefore, the results show that the FSA and FST have the highest HOMO energies and that FSO has the lowest HOMO energy. This means that FST has higher ability to donate an electron, while FSA has the lowest ability to donate an electron. Table 2 displays the HOMO and LUMO energies, and the energy gap between the EA and the ionization energy of the studied molecules. The results indicate that FST has a low band gap (energy gap); this means that the ability to transfer an electron from the ground state to the excited state is very easy. If the energy gap is larger for the molecules, then the ability to transfer an electron from the HOMO to the LUMO will be lower.
The HOMO and LUMO energies, and the energy gap between ionization potential and electron affinity calculated at the DFT/6-311G level.

FS: fluorescein; FSA: fluorescein attached with amine; FSE: fluorescein attached with ethane; FSM: fluorescein attached with methane; FSO: fluorescein attached with methoxy; FST: fluorescein attached with thiophene.
Global molecular reactivity
IP and EA
The IP is the amount of energy required to remove an electron from a molecule, computed as the energy difference between the cation and the neutral molecule. It is negative value of the HOMO energy as displayed in equation (1). The lower the energy used to remove the electron from the molecule, the easier ionization of the molecule, but the higher the energy required to remove the electron, the more difficult the molecule is to ionize. Generally, the IP is affected by the size of the molecule, that is, the higher the molecular size, the lower the IP, and a smaller molecular leads to a higher IP. From the DFT results, it can be seen that the FST dye has a high IP and that the FSA dye has a low ionization (Table 3). The increasing order of IP for the studied dyes is FSA < FSM < FSE = FS < FSO < FST.
Calculated global quantities, ionization potential, electron affinity chemical potential, electronegativity, hardness, softness, and electrophilicity of the different dyes determined at the DFT/B3LYP/6-311G level.

FS: fluorescein; FSA: fluorescein attached with amine; FSE: fluorescein attached with ethane; FSM: fluorescein attached with methane; FSO: fluorescein attached with methoxy; FST: fluorescein attached with thiophene.
The EA is the energy released when an electron is added to a neutral molecule, and it is computed as the energy difference between the neutral form and the anion. This parameter is the negative value of the LUMO energy and is calculated as indicated in equation (1). In the studied molecules, FST has the highest EA and FSA has the lowest EA. The decreasing order of EA of the dye is FST > FSE > FS > FSM > FSO > FSA. Table 3 displays the values of each molecule.
Chemical hardness (η)
This is an important descriptor for understanding the behavior of a chemical system, because it measures the resistance to change in the electron distribution of nuclei and electrons, 18 and it is calculated as indicated in equation (6). It is an important global reactivity indicator for predicting the susceptibility to and the type of reactions a reagent undergoes. 17 Table 3 displays the chemical hardness of the FS dye derivatives. The results show that the dye with a large energy gap has a large value of chemical hardness and that with a low energy gap has a low chemical hardness. Therefore, from the studied molecules, FSA has a high value of chemical hardness, and FST has a low value of chemical hardness.
Chemical potential (µ)
The chemical potential is the negative of the Mulliken electronegativity. 19 It is an indicator for a chemical reaction to take place. This means that a reagent with a high electronic chemical potential is a good electron donor, whereas a reagent with small electronic chemical potential is a good electron acceptor. 17 From the results in Table 3, it can be seen that FSA has a high chemical potential (−7.1535), which is a good electron donor compared with other derivatives. While the FST dye has a small chemical potential (−7.419), it is a good electron acceptor. The chemical hardness and the chemical potential are considered as intrinsic properties of the molecules, and help to give an important guide for the prediction of the product in a chemical reaction. 19
Electrophilicity (ω)
Electrophilicity is the change in energy that occurs when a reagent is placed in contact with a perfect nucleophile. This descriptor measures the property of electron accepting by molecules. A molecule with good electrophilicity has low chemical potential and low chemical hardness. 19 Equation (7) helps calculate the electrophilicity of a molecule, and the values are displayed in Table 3. The results indicate that the FSA dye has good electrophilicity properties compared with the other dyes and FST has poor electrophilicity property.
Thermodynamic properties
The thermodynamic properties of the molecular dyes discussed in this study are entropy, dipole moment, heat capacity, and energy of the molecules. The DFT results show that the entropy, heat capacity, and dipole moment of the molecules increased as the conjugation increases. Therefore, FST has a larger dipole moment, heat capacity, entropy, and energy compared with the other molecules (Table 4).
The energies of the dyes enthalpies, free energies, entropies, and dipole moments calculated at the DFT B3LYP/6-311G level.

FS: fluorescein; FSA: fluorescein attached with amine; FSE: fluorescein attached with ethane; FSM: fluorescein attached with methane; FSO: fluorescein attached with methoxy; FST: fluorescein attached with thiophene.
Conclusion
A donating group attached to a dye molecule results in decreasing of the bond length. A large energy gap results in a lowering of the transfer of electrons from the HOMO to the LUMO. For this case, a dye molecule will easily transfer an electron from the LUMO to a titanium dioxide (TiO2) electrode. Therefore, it will easily produce electricity.
The molecular geometry was determined through this experiment, and the result indicates that electron donating usually increases the bond length. For the molecule with π-conjugated spacer, the bond length slightly decreases which results in easy transfer of electron. Not only the bond length but also the energy gap helps to determine electron transfer from ground to excited state of the molecule.
The tendency of atom to accept the electron was determined using electrophilicity in which the FSA is good for accepting electron while FST is poor in electron acceptor. Also the experiment was aimed to determine the free energy, heat capacity, enthalpy, and dipole moment of dye which is very important for searching the good dye.
Footnotes
Acknowledgements
The University of Dodoma is acknowledged for providing the Computational Laboratory to conduct this work. The Muslim University of Morogoro is also acknowledged for allowing the practical work to be conducted at the University of Dodoma.
Author contributions
J.M.J. and S.A.H.V. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Zanzibar Higher Education Loans Board.
