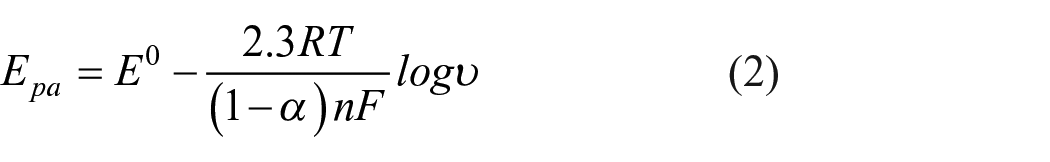Abstract
A simple and efficient electrochemical sensor based on a homemade reshaped micropipette tip carbon paste electrode is reported. Molecularly imprinted polymer membranes of graphene oxide and polypyrrole are synthesized and modified on the surface of micropipette tip carbon paste electrode. The merit of the method is evaluated under optimized conditions via differential pulse voltammetrics. The prepared sensor exhibits remarkable sensitivity toward dopamine with a linear range of 6.4 × 10−8–2 × 10−4 M, with a limit of detection as low as 1 × 10−8 M. The proposed method is applied for the determination of dopamine in urine samples by the standard addition route. A range of 1 × 10−7–1 × 10−4 M is obtained from these samples. The relative recoveries are in the range of 95.2%–104%. The proposed method has acceptable performance for the determination of dopamine in real samples with excellent sensitivity and selectivity.

Introduction
Dopamine (DA) is an important neurotransmitter and plays a key role in hormones, kidney function, and in the central nervous system.1,2 Abnormal changes in DA concentration can lead to schizophrenia, Parkinson’s disease, Alzheimer’s disease, and Huntington’s disease.1,3 Therefore, selective quantification of DA is essential for the diagnosis of these types of disorders. Many analytical techniques have been developed for DA sensing such as colorimetric, 4 fluorescence, 5 gas chromatography-mass spectrometry, 6 high-performance liquid chromatography, 7 Raman spectroscopy, 8 chemiluminescence, 9 and photoluminescence 10 techniques. Despite their accuracy and stability, these techniques suffer from various other disadvantages, mainly in terms of selectivity, cost, calibration, time-consuming procedures, and requirement of technical equipment. Therefore, these analytical methods are not amenable for miniaturization and hence are not suitable for point-of-care testing. 11 Electrochemical techniques benefit from being low cost, label-free, highly sensitive and selective, practical, and give fast and reliable responses. In addition, they are amenable to miniaturization and therefore have been proven to be a better choice.
Recently, modified electrodes prepared from a bare electrode (3 mm diameter) were used for electrochemical detection in a mL-scale volume cell. 12 The miniaturization of electrochemical analysis systems is an important topic worthy of study. Molecular imprinted polymers (MIPs) are very important ligands with high affinity for targeting in electrochemical sensor detection methods. 13 Because of the obvious advantages, such as thermal stability, physical robustness, low cost, and easy preparation, MIPs have proven to be promising for biomolecule recognition. 14 However, electrochemical sensors based on MIPs generally have the disadvantages of complex synthetic steps. In addition, because of the low surface area and conductivity of MIPs-modified electrodes, electrochemical sensors based on MIPs exhibit weak sensitivity. To address this issue, electrochemical sensors based on MIPs need to incorporate new material with high conductivity and high specific surface area. Graphene oxide (GO) has attracted significant attention, since it demonstrates many interesting mechanical, 15 thermal, 16 and electrical 17 properties. It has recently been recognized as an ideal material that can be used as a supplement for the preparation of MIPs resulting in the many advantages mentioned above. 18
Herein, a method for the simple synthesis of an MIP ligand on the surface of an electrode and a convenient process to prepare a reshaped electrode with a small volume are discussed. On the basis of GO, a simple and efficient one-step process for the synthetic MIP preparation of a modified micropipette tip carbon paste electrode (PTE) to detect DA with higher sensitivity has been achieved. The homemade PTE with a cylindrical shape was perfectly shaped with a micropipette tip. The PTE possessed an adjustable electrochemical active surface through using various types of micropipette tip. The minimized GO-MIP modified PTE (GO-MIP-PTE) working electrode can realize electrochemical analysis on a 500-μL scale (Scheme 1).

Process of preparing GO-MIP-PTE.
Results and discussion
Characterization of the fabricated electrode surfaces
To investigate the morphology of the GO- and GO-MIPs-modified PTE (GO-PTE, GO-MIP-PTE), scanning electron microscopic (SEM) analysis was carried out and the results are shown in Figures 1(a) and (b). It can be seen from Figure 1(a) that GO has a well exfoliated, wrinkled structure surface morphology, and can form an ultrathin and uniform graphene film. 19 Figure 1(b) represents the polymerized pyrrole on the graphene surface. It is observed that the polymerized pyrrole is uniformly deposited on the surface of grapheme particles, offering a highly accessible surface area for the targets.

SEM images of (a) GO-PTE and (b) GO-MIP-PTE. (c) CVs of the electrode modified by GO in 0.1 M KCl indicating that GO had been reduced. (d) Raman spectra of GO-PTE and GO-MIP-PTE.
Figure 1(c) shows the result of cyclic voltammetry (CV) of the GO-modified electrode in 0.1 M KCl solution. Ramesha and Sampath 20 have found that GO reduction can be achieved with only one scan. The electrochemical reduction is an irreversible process.
As shown in Figure 1(d), the structure of the composite material was studied by Raman spectroscopy. The Raman spectrum of the GO film displays a D-band at 1325 cm−1 and a broad G-band at 1600 cm−1. The D/G band intensity ratio expresses the atomic ratio of sp3/sp2 carbons, which is a measure of the extent of disordered graphite. 21 Studies have shown that the defect density is proportional to ID/IG. The ID/IG of the Raman spectra of GO-PTE, GO-NIP-PTE, and GO-MIP-PTE are 1.4, 1.23, and 1.11, respectively.
Electrochemical responses
The CV analysis of bare homemade PTE, GO-PTE, and GO-MIP-PTE was performed to study the increase in the effective surface area of the sensor. An electroactive redox probe couple [Fe(CN)6]−3/4− is normally used to study the electrocatalytic activity of modified electrodes. 22 The CVs of bare homemade PTE, GO-PTE, and GO-MIP-PTE in 6 mM K3Fe(CN)6 containing 1 M KNO3 by sweeping the potential from −0.2 to 0.7 V with a 100-mV/s scan rate are shown in Figure 2. The areas of the electrodes were evaluated in terms of the Randles–Sevcik equation from the CV 23
where Ip (A) is the anodic peak current, A (cm−2) is the surface area of the electrode, D (7.6 × 10−6 cm−2 s−1) is the diffusion coefficient, n (n = 1) is the electron-transfer number, υ (V s−1) is the scan rate, and C (mol/cm3) is the concentration of K3Fe(CN)6. 24 The calculated surface areas are 0.782, 1.56, and 3.69 mm2 for PTE, GO-PTE, and GO-MIP-PTE, respectively. The A value of GO-MIP-PTE is 371.9% larger than the geometrical surface area of bare PTE. This enhancement in electrode surface area is attributed to the modification process of PTE using the mixture of GO and MIP.

CVs of PTE, GO-PTE, and GO-MIP-PTE in 0.1 M KCl containing 6 mM K3FeCN6
Figure 3(a) displays the CV responses of GO-MIP-PTE at scan rates ranging from 25 to 500 mV/s in the presence of 5 × 10−5 M DA. It was observed that the increase in the redox current was dependent on an increasing scan rate. A more positive shift in the anodic peak potential occurred with increasing scan rates. It is noteworthy that there is a linear calibration curve between the log of the scan rate and the anodic/cathodic potentials.24,25 In Figure 3(b), a linear calibration curve was obtained with a linear regression coefficient (R2) of 0.935 and 0.923 for the anodic and cathodic peak potentials, respectively. The above observation predicts that the oxidation of DA is adsorption controlled at the surface of GO-MIP-PTE. 26

(a) CVs of GO-MIP-PTE in 0.1 M PBS (pH 6.8) in the presence of DA (5 × 10−5 M) at different scan rates. (b) Linear regression curves between the log of scan rate (mV/s) versus the anodic/cathodic peak. (c) The relationships of the peak potential Ep versus log υ.
Hence, the electron-transfer kinetic parameters, such as the electron-transfer coefficient (α) and the number of electronic transfers (n) of DA on GO-MIP-PTE, were calculated according to Laviron’s equation
where n represents the number of electrons transferred in the three reaction, R = 8.314 J/(mol·K), T = 298 K, and F (C mol−1) is the faraday constant.
The linear relationships between the oxidation peak potential (Epa) and reduction peak potential (Epc) with the logarithm values of υ (log υ) were established (Figure 3(c)), and the linear equations of DA are Epa (V) = −0.01369 log υ (V s−1) + 0.03764 (R = 0.954) and Epc = 0.01607 log υ (V s−1) + 0.1670 (R = 0.984), respectively. Next, according to equations (2) and (3), the values of α and n are calculated as 0.46 and 7.98, respectively.
Analytical parameters
Figure 4(a) is obtained by studying the effect of the adsorption time on the DPV peak current. With an incubation time from 0 to 30 min, the peak current increased rapidly, and then stabilized after 30 min. This result reveals a rapid response equilibrium between DA molecules and GO-MIP-PTE, which might be due to the surface binding sites of the GO-MIP-PTE composite through π–π stacking between the amino groups of DA and the oxygen-containing groups of the polypyrrole-loaded GO. 27

(a) Effect of the incubation time on the DPV peak current of GO-MIP-PTE in 0.1 M PBS (containing 1.6 × 10−6 M DA, pH = 6.8), (b) the DPV plots of the electrode modified by GO-MIP-PTE in 0.1 M PBS (containing a: 6.4 × 10−8, b: 3.2 × 10−7, c: 1.6 × 10−6, d: 8 × 10−6, e: 4 × 10−5, and f: 2 × 10−4 M DA), and (c) the negative logarithmic linear fitting graph of DPV peak value and concentration.
Figure 4(b) shows the DPV response of the GO-MIP-PTE-modified electrode after incubation in DA solution. The oxidation peak current increases with the increase in template molecule concentration. Figure 4(c) shows a good linear region with DA concentration in the range of 6.4 × 10−8–2 × 10−4 M. The linear regression equation is expressed as I (μA) = 30.45 + 3.24 log (CDA) with a correlation coefficient of R2 = 0.9945. The limit of detection (LOD) was 1.0 × 10−8 M based on the signal corresponding to three times the noise of the response. As shown in Table 1, the results obtained using the GO-MIP-PTE sensor used to detect DA are compared with other reported methods, which shows that the prepared GO-MIP-PTE sensor has high sensitivity and a low detection limit.
A comparison of the prepared GO-MIP-PTE-modified electrode with other electrochemistry methods used for the determination of DA.

GO-MIP-PTE: GO-MIP modified PTE; LOD: limit of detection.
Selectivity
In general, coexisting electroactive components, such as ascorbic acid (AA) and uric acid (UA), show serious interference in the electrochemical detection of DA. As shown in Figure 5, the DPV current response of DA is significantly greater than that of structural analogs, and the current responses of AA and UA are equivalent to 11.05% and 13.87% of the DA current response, respectively. The large current response of the sensor to DA is because GO-MIP-PTE specifically binds to DA, while the structural analogues are mainly non-specifically bound. Therefore, the measured current response is small, indicating that the developed sensor is sensitive and has high selectivity toward DA.

DPV current response of GO-MIP-PTE, GO-NIP-PTE, and GO-PTE in 0.1 M PBS containing 4 × 10−5 M dopamine (DA), ascorbic acid (AA), and uric acid (UA). The error bars indicate the standard deviation from three measurements.
Recovery measurements for real samples
In order to evaluate the applicability of the proposed sensor, a standard addition method was used to determine the concentration of DA in a human urine sample. The analytical results are shown in Table 2. The recovery rate is in the range of 95.2%–104%, indicating that the sensor has good accuracy and has potential practical application in the analysis of DA in actual samples.
Determinations of DA in human urine samples (n = 6).

Conclusion
A mixture of graphite and paraffin is filled in the micropipette tip to prepare PTE. PTE was modified with GO-MIP based on polypyrrole through a simple process. This novel electrochemical sensor has the advantages of a high current response, a low detection limit, and good selectivity for DA, with analytical recovery values in the range of 95.2%–104%. In addition, accurate analysis of actual samples also provides good prospects (relative standard deviation range of 0.63%–2.94%).
Materials and methods
Reagents and chemicals
Pyrrole, DA, AA, and UA were purchased from the Shanghai Aladdin Bio Chem Technology Co., Ltd., P. R. China. Analytical reagent (AR) grade potassium chloride, hydrochloric acid, hydrogen peroxide (30%), and graphite powders were purchased from Sinopharm Chemical Reagent Co. (China). Phosphate buffer solution (PBS, 0.1 M, pH = 6.8) was prepared with monobasic sodium phosphate (NaH2PO4) and dibasic sodium phosphate (Na2HPO4). Ultrapure water was used for the preparation of all solutions.
Apparatus
Electrochemical tests including CV and differential pulse voltammetry (DPV) were performed using a CHI 660E electrochemical workstation (Chen Hua Instruments Co., Shanghai, China) with a three-electrode system. Modified or bare, the PTE (1.5 mm diameter) is the working electrode, a saturated calomel electrode (SCE) is the reference electrode, and the graphite electrode is the auxiliary electrode. All measurements were performed at room temperature.
Preparation of GO-MIP and GO-MIPs-modified PTE
Graphite powder and paraffin oil, in a ratio of 70:30 (%, w/w), were added to a porcelain mortar and ground evenly. The micropipette tip was filled with the obtained mixture. 19 Copper wire was used as the conductive medium. Before use, the electrode was first polished on paper and then scanned over several cycles between −0.7 and 0.2 V in 0.1 M PBS (pH: 6.8 containing 0.1 M KCl) until the peak stabilized. To reduce the influence of other factors in the process of making the electrodes, the carbon paste on the surface of the PTE was removed.
GO was prepared from graphite powders using a modification of the Hummers method. 20 GO-MIPs were initiated by the addition of H2O2 (0.5 mL) to a sonicated GO aqueous solution (50 mL, 0.01 g/mL) containing pyrrole (0.05 mL) and DA (0.05 g) over 6 h. Subsequently, the composite dispersion (10 μL) was dropped on to the surface of the PTE and dried at room temperature. The embedded DA was eluted by scanning between +0.7 and −0.2 V in 0.1 M PBS (pH: 6.8) containing 0.1 M KCl over 30 cycles. After that, the was placed in 0.1 M PBS containing DA of different concentrations, incubated for 30 min and then tested by DPV. The sensor was stored at 4 °C. The method for the preparation processes of the GO-NIPs sensors was the same as that described above, but without the addition of DA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the collaborative innovation project of Anhui Provincial Department of Education (Grant No. GXXT-2019-034) and the Science Research Project of Anhui Provincial Education Department (Grant Nos. KJ2019A0758 and KJ2019JD08).