Abstract
Alkylphosphinates have received extensive attention in the past few decades because of their very useful mechanical properties, electrical properties, low density, and low toxicity, and have been widely used in flame-retardant materials and other fields. In this work, aluminum diethylphosphinate is successfully synthesized under atmospheric pressure. More importantly, two novel dialkylphosphinates, aluminum dioctylphosphinate and aluminum didecylphosphinate, are first synthesized and characterized. The structures of these aluminum dialkylphosphinates are confirmed by nuclear magnetic resonance, Fourier-transform infrared spectroscopy, and high-resolution mass spectrometry, and the microscopic morphology and thermal stability are analyzed by scanning electron microscopy and thermogravimetric analysis, respectively. Furthermore, conditions for the synthesis of aluminum dioctylphosphinate are optimized. Compared with the traditional method of synthesizing dialkylphosphinates under high pressure, the method reported in this paper has the advantages of high safety, easy operation, and low economic cost, which makes it suitable for industrial production.
Keywords
A novel method of synthesizing aluminum dialkylphosphinates under mild conditions is developed. The conditions for the synthesis of aluminum dioctylphosphinate are optimized. Compared with the traditional method of synthesizing dialkylphosphinates under high pressure, the method reported in this paper is suitable for industrial production.

Introduction
Organic polymeric materials have been widely used in various fields in society, including high-end manufacturing in the fields of aviation, electronic components, and the automotive and construction industries due to their low density and easy fabrication. 1 However, the limiting oxygen index (LOI) of most organic polymeric materials is less than 25%, which means they are easy to combust. 2 In addition, organic polymeric materials have a high heat release rate of combustion and readily drop molten droplets and release flue gas, which can result in huge economic losses and casualties.3,4 Therefore, high-efficiency flame retardants capable of enhancing the properties of such polymeric materials are of significant importance. In recent years, with continuous concern regarding fire safety and environmental protection, and also with increasing stringent regulations, the development of halogen-free flame retardants is an important research field.5–10 Organic phosphinates are considered to be one of the most promising flame retardants as alternative halogen-based flame retardants, which have been developed very rapidly in recent years.11,12 Alkylphosphinates are halogen-free, hydrophobic, and have high flame-retardant efficiency, good thermal stability, and little impact on the electrical performance and mechanical properties of materials, which makes them especially suitable addition to thin-walled electronic materials. 13
There are four methods to synthesize metal salts of alkylphosphinates. (1) The Grignard reaction method, (2) the aluminum chloride catalysis reaction, (3) the metal coordination catalysis reaction, and (4) free-radical addition. 14 Dialkylphosphoric acids can be prepared with high stereospecificity through the Grignard reaction method. In addition, high yields and purities can be achieved. 15 Unfortunately, due to the harsh reaction conditions and the complex process involved, the cost is extremely high, which is not conducive to large-scale industrial production. The reaction conditions for the aluminum chloride catalysis reaction are mild since the reaction can be carried out at room temperature. However, the AlCl3 catalyst is strongly corrosive, and requires equipment with high corrosion resistance, while the yields of the metal salts of alkylphosphonates are low. Meanwhile, the product is difficult to separate from the reaction mixture and large amounts of waste water are produced during the process. Hence, the method is not suitable for large-scale production. 16 Giving high product yields, metal coordination catalysis utilizes mild conditions and the experimental process is simple. However, the high price of the catalysts has limited its application, and it is generally applied for the synthesis of monoalkylphosphoric acids. 17 Free-radical addition is currently the most widely used method for the industrial synthesis of metal salts of dialkylphosphonates. In the presence of a free-radical initiator, hypophosphorous acid, monoalkylphosphoric acid, or its base metal salt react with alkanes to produce the target products, the procedure for which is simple to operate. 18 More importantly, this type of free-radical addition reaction delivers high yields and high purities, and the products are easy to separate. Therefore, the free-radical addition reaction is widely used in industrial production. Bauer et al. 19 have developed a method for synthesizing aluminum diethylphosphinate (ADEP) via a free-radical addition reaction, with the product being obtained in an excellent yield of more than 90%; however, it was necessary to carry out the reaction under high pressure. Zheng et al. successfully synthesized dialkylphosphoric acids using a Grignard reagent and dioctylphosphinic acid was obtained in 65% yield. However, the experimental process involved four steps and the reaction conditions were relatively complex. 20 Deprèle et al. studied the effects of Pd2(dba)3/xantphos as the catalyst on the hydrophosphinylation of alkenes and obtained compounds with a P-C bond. Although the method affords yields of circa 79%, the catalyst was only effective in the synthesis of monoalkylphosphoric acids. 21
As the most widely used flame retardant consisting of a metal salt of an alkylphosphinate, ADEP has attracted significant attention in the past decades. ADEP has excellent thermal stability and remains stable at 350 °C, while the 5% decomposition temperature can reach more than 400 °C. It can be applied to thermoset resins, nylons, and many other organic polymer materials, and is especially suitable for thin-walled electronic components and transparent films.13,22 Compared with inorganic flame retardants, polymeric materials with smaller amount of ADEP can show satisfactory flame-retardant properties, and ADEP has better compatibility in polymeric materials and has less adverse effects on the mechanical and electrical properties of materials. In addition, it was found that the combination of ADEP and other types of flame retardant such as nitrogen flame retardants and silicon flame retardants resulted in better flame-retardant effects.23,24 ADEP was also studied from the perspective of toxicology and environmental protection, and it was also found that it had no adverse effects. 25 Free-radical addition reactions under high pressure have been widely employed over the past decades. However, the utilized high-pressure conditions have led to a number of safety hazards and the high-pressure reactor requires significant energy consumption and costly maintenance.
In this work, a novel method for the synthesis of aluminum dialkylphosphinates by the free-radical addition method under mild conditions is developed. ADEP was prepared by a new synthetic method. To further explore the characteristics of other aluminum dialkylphosphinates, aluminum dioctylphosphinate (ADOP) and aluminum didecylphosphinate (ADDP) containing long alkyl chains with good thermal stability were also synthesized. The synthetic route has the advantages of low cost, simple synthesis, safe operation, and low energy consumption, which has the potential to be applied in industrial production.
Results and discussion
Synthesis of aluminum dialkylphosphinates
The synthesis of ADEP is depicted in Scheme 1. ADEP was synthesized in two steps, and was explored under different reaction conditions. In the first procedure, sodium hypophosphite (SHP) monohydrate was dissolved in acetic acid with stirring, and the solution was then heated to 90 °C. Ethylene gas was continuously bubbled into the reaction solution and dibenzoyl peroxide (BPO) dissolved in acetic acid was added dropwise. In the following study, it was found that the yield of the second step was higher than 90% and remained quite stable. Therefore, the change trend of the total yield of the two steps was considered to be the same as that of the first step. It was found that as the reaction time increased from 11 to 43 h, the yield of ADEP increased significantly from 8% to 71% (Table 1, entries 1–4) when BPO was used as the radical initiator. However, when BPO and 2,2′-azoisobutyronitrile (AIBN) were employed together as co-initiators, the yield of ADEP decreased to 30%. Sodium diethylphosphinate (SDEP) was obtained after the reaction mixture had been evaporated in vacuo. In the second step, SDEP was dissolved in water to obtain an aqueous solution. It was found that the pH of the aqueous solution had a significant influence on the yield of the product. When the pH of the SDEP aqueous solution was not deliberately adjusted, the pH of the aqueous solution was about 3 and the ADEP yield was only about 15% after adding the aqueous solution of aluminum sulfate. Thus, the pH of the aqueous solution of SDEP was adjusted to 7 before further operation and there was a sharp increase in the yield of ADEP. It was assumed that acidic conditions were not conducive to the production of ADEP.

Synthesis of aluminum dialkylphosphinates.
Optimization of the reaction conditions for the synthesis of ADEP a .
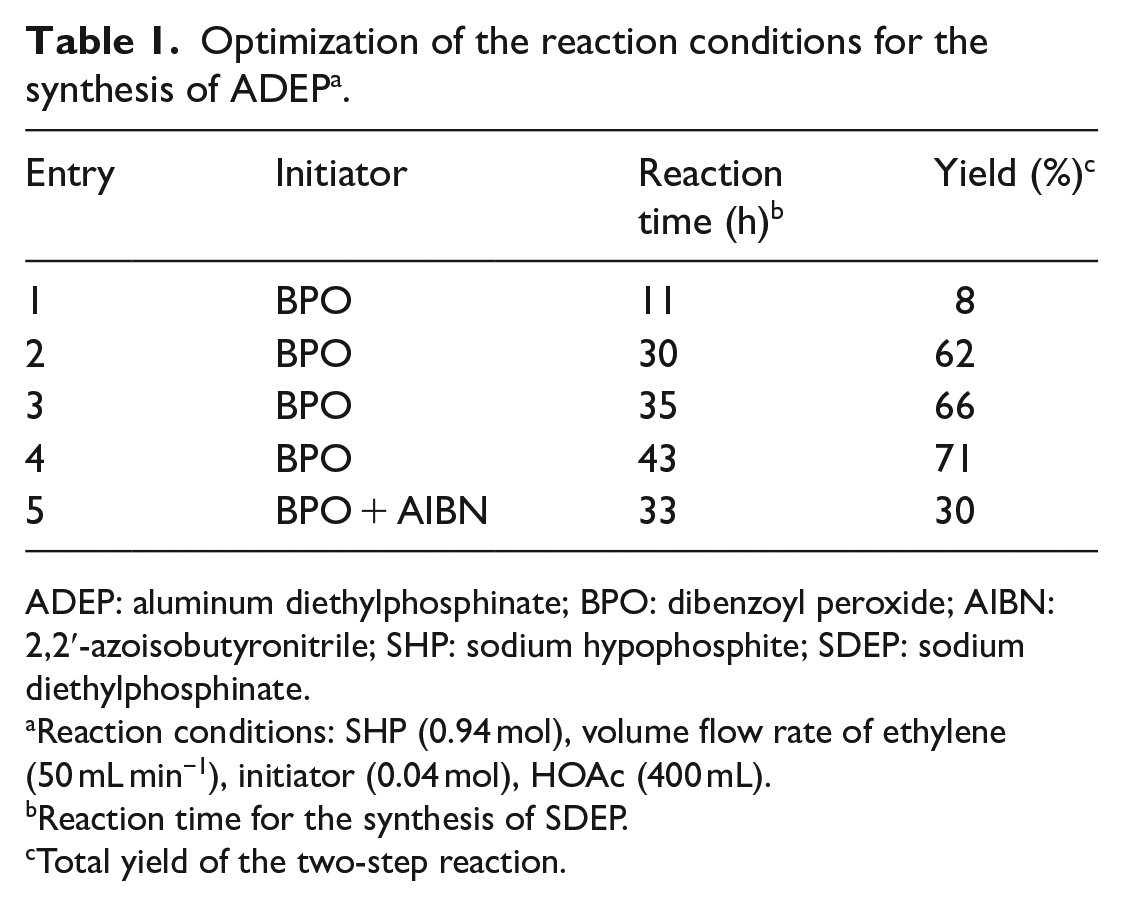
ADEP: aluminum diethylphosphinate; BPO: dibenzoyl peroxide; AIBN: 2,2′-azoisobutyronitrile; SHP: sodium hypophosphite; SDEP: sodium diethylphosphinate.
Reaction conditions: SHP (0.94 mol), volume flow rate of ethylene (50 mL min−1), initiator (0.04 mol), HOAc (400 mL).
Reaction time for the synthesis of SDEP.
Total yield of the two-step reaction.
It was also found that the reaction time in the synthesis of SDEP had obvious effects on the purity of ADEP. Figure 1 shows the Fourier-transform infrared (FTIR) spectra of ADEP obtained at different reaction times. The absorption bands between 2970 and 2880 cm−1 are attributed to the C–H stretching vibrations of saturated methyl and methylene groups. The bands at 1463 and 1409 cm−1 are assigned to the deformation vibrations of C–H. The peaks at 1153 and 1079 cm−1 correspond with the P=O and P-O groups, respectively. The band located at 1271 cm−1 is due to P=O and that at 1232 cm−1 is due to P-C absorptions, while the peak at 777 cm−1 is attributed to P-C absorptions.26,27 It was observed that the peak at 2353 cm−1, which corresponds with the P-H absorption, gradually became smaller as the reaction time increased, indicating that the purity of the ADEP product increased with less monoalkyl product and less aluminum hypophosphite. When the reaction was carried out for 43 h, the peaks due to P-H absorptions barely existed, which indicates that the product was highly pure (see Figures S1–S7 in Supporting Material).
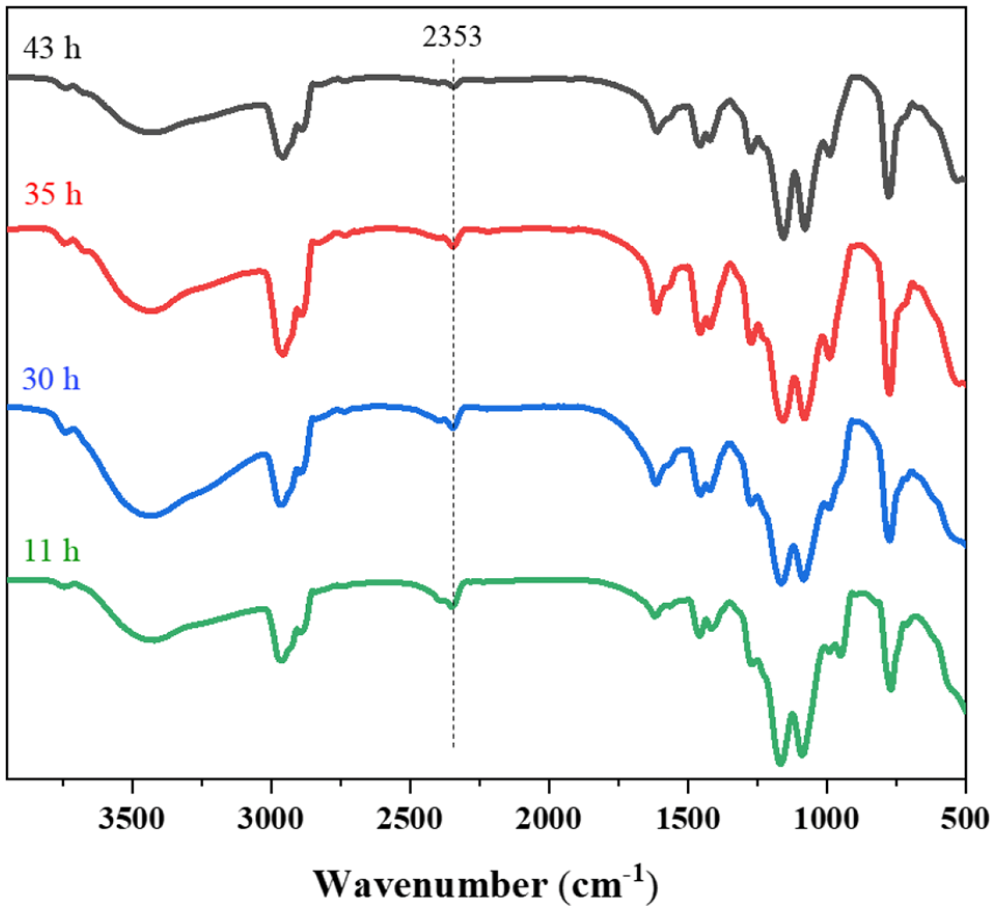
FTIR spectra of ADEP obtained at different reaction times.
Ethylene is a gas at room temperature and needs to be pressurized so that it can be stored in a gas cylinder, which brings more danger and higher costs to the production process. After successfully synthesizing ADEP under atmospheric pressure, we attempted to synthesize dialkylphosphinates containing long alkyl chains, which might have analogous properties to ADEP, using a similar method. What is more, since the addition of ADEP has an adverse impact on the mechanical properties of organic polymer materials, we deduced that the structure of the long carbon chain might enhance the compatibility of aluminum dialkylphosphinate in organic polymer materials. Based on the above results, the synthesis of ADOP and ADDP under atmospheric pressure was further investigated, and the synthetic conditions for ADOP were optimized. The synthesis of ADOP and ADDP is described in Scheme 2.

Synthesis of aluminum dialkylphosphinates.
The effect of how the alkene was added to the reaction system on the yield of the sodium dialkylphosphinate was first investigated. The molar ratio of 1-octene and SHP was 2.5:1, the reaction temperature was 95 °C, the reaction time was 24 h, and the BPO dosage was 4% (mol percent of NaH2PO2). 1-Octene was directly added to the reaction system considering the simplicity of the operation. Sodium dioctylphosphinate (SDOP) was obtained in a yield of 17%. To further improve the yield of SDOP, 1-octene was added dropwise and the yield of SDOP increased significantly to 66%. It was assumed that a large amount of the excess alkene self-polymerized easily on initiation by a radical initiator, which led to a low yield of the target product. Therefore, the alkene was added dropwise into the reaction system to avoid the occurrence of side reactions. Unlike SDEP, SDOP is poorly soluble in water, likely due to it possessing long alkyl chains. Hence, a range of water-miscible organic solvents was selected to try to dissolve SDOP. It was found that SDOP had better solubility in tetrahydrofuran, so a mixed solvent of tetrahydrofuran and water with a volume ratio of 1:1 was chosen as the solvent to dissolve SDOP. Next, an aqueous solution of aluminum sulfate was added to the solution of SDOP and ADOP was obtained in a total yield of 75%. ADDP was obtained in a total yield of 69% under similar conditions.
It can be observed from Figure 2 that SHP has four characteristic absorption peaks. The peaks at 2352 and 2323 cm−1 are assigned to P-H, while the peaks at 1192 and 1056 cm−1 are attributed to P=O and P-O absorptions, respectively. In the FTIR spectra of ADOP and ADDP, the peaks at 2352 and 2323 cm−1 disappear, which indicates that the n-octyl or n-decyl groups have replaced H in P-H. In addition, the n-octyl group and n-decyl groups have stronger electron-donating abilities than H and the wavenumber of the characteristic absorption peak of P=O decreases and shifts from 1192 to 1156 cm−1. Besides, the typical P-O bond absorption peak shifts from 1056 to 1085 cm−1. The reason for this is that aluminum cations replace sodium cations in ADOP and ADDP, and the electron-donating ability of aluminum cations is weaker than that of sodium cations, resulting in an increase of the wavenumber of the characteristic absorption peak of P-O. In the FTIR spectra of ADOP and ADDP, new absorption peaks appear at 2926, 2855, 1467, and 1406 cm−1. Among them, the peaks at 2926 and 2855 cm−1 are attributed to the stretching vibrations of C–H in methyl or methylene groups, and the absorption peaks at 1467 and 1406 cm−1 are due to the bending vibrations of C–H in the n-octyl or n-decyl groups. 28 All the above results show that an alkylation reaction had occurred between SHP and 1-octene or 1-decene, and the carbon chain was introduced into the structure of SHP to form alkylphosphinates. The results of high-resolution mass spectrometry (HRMS) analysis and nuclear magnetic resonance (NMR) analysis (see Figures S8–S13 in the Supporting Material) proved that SDOP and ADOP had been successfully synthesized. The multiple peaks with chemical shifts between 1.60 and 1.42 ppm belong to the H atoms connected to the α-C and β-C of the carbon chain, the triplet with a chemical shift at 0.88 ppm belongs to H atoms in CH3, and the multiple peaks with chemical shifts between 1.35 and 1.12 ppm are attributed to the remaining hydrogens in the carbon chain. It was found in the 1H NMR spectrum that the chemical shifts of all the peaks shifted downfield because aluminum cations with weaker electron-donating ability replaced sodium cations. The 31P NMR spectra of SDOP and ADOP showed only a single peak, indicating the high purities of the products. The successful synthesis of sodium didecylphosphinate (SDDP) was proved according to HRMS and NMR (see Figures S14–S17 in the Supporting Material). The multiple peaks between 1.55 and 1.41 ppm belong to the hydrogens connected to the α-C and β-C atoms of the carbon chain, the triplet with a chemical shift at 0.88 ppm is due to the H atoms in CH3, and the multiplets with chemical shift between 1.32 and 1.22 ppm are attributed to the remaining H atoms in the carbon chain. Besides, the single peak in the 31P NMR spectrum of SDDP indicates that the product is highly pure. From the above analysis, it can be seen that the molecular structures of ADOP and ADDP are consistent with those expected.
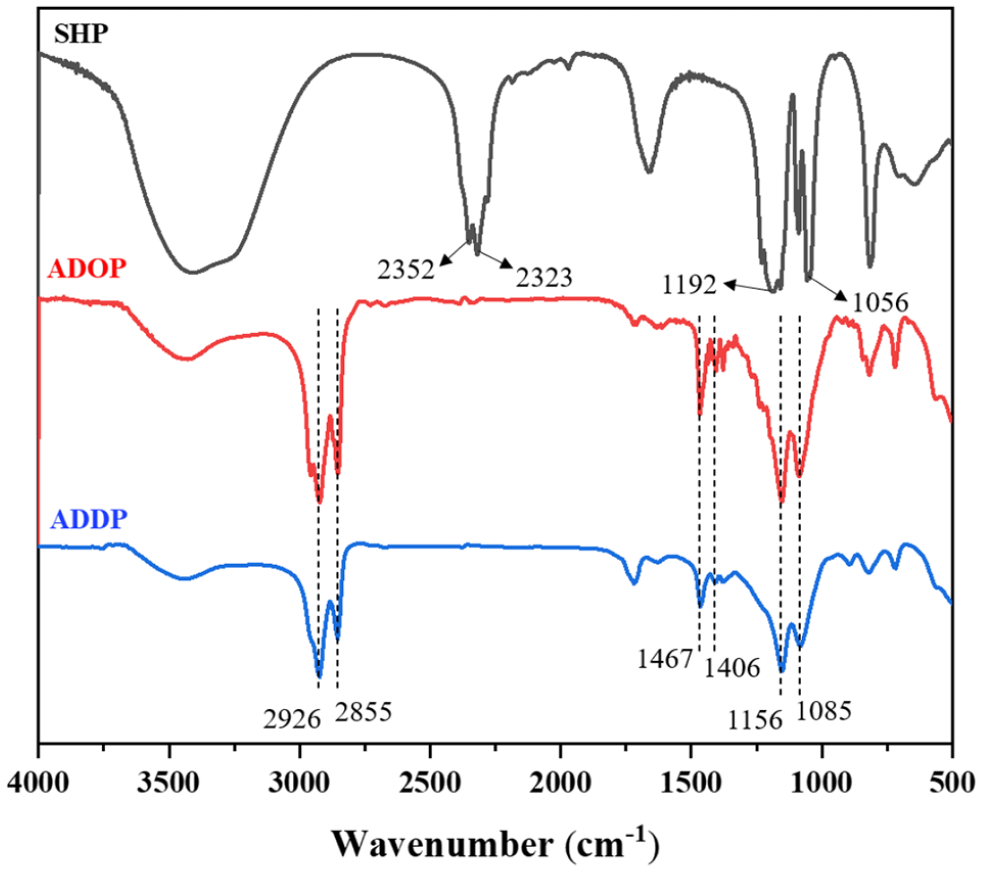
FTIR spectra of SHP, ADOP, and ADDP.
The thermal stability of aluminum dialkylphosphinates was evaluated by thermogravimetric analysis (TGA). The thermogravimetric curves of ADEP, ADOP, and ADDP are illustrated in Figure 3(a). It was found that the temperatures representing 5% weight loss of ADEP, ADOP, and ADDP are 401, 349, and 294 °C, respectively. Besides, the residual weight rates of all three at 595 °C were 35.64%, 19.75%, and 13.45%, respectively. It was observed that these aluminum dialkylphosphinates have high thermal stability, and on increasing the carbon chain length in the molecular structure, the temperature at which the mass loss of aluminum dialkylphosphinates was 5% and the residual weight rate at 595 °C had both decreased. The differential thermal gravity (DTG) curves of ADEP, ADOP, and ADDP are given in Figure 3(b). From Figure 3(b), it could be seen that the temperatures corresponding to the maximum thermogravimetric rates of ADEP, ADOP, and ADDP were 463, 478, and 483 °C, respectively, and that maximum thermogravimetric rates of the three were 2.08%, 1.18%, and 1.28% min−1. Although the decomposition temperatures of ADOP and ADDP were lower than those of ADEP, their maximum thermogravimetric rates were significantly lower than those of ADEP. It can be inferred that increasing the carbon chain length in the molecular structure reduced the thermal stability of aluminum dialkylphosphinates to a certain extent.

(a) TGA and (b) DTG analysis of ADEP, ADOP, and ADDP. Decomposition temperature (Td) is defined as the temperature with a 5% weight loss point.
Figure 4 shows the scanning electron microscopy (SEM) images of ADEP, ADOP, and ADDP. It can be observed that the synthesized ADEP solid exists as rod-shaped crystals with a diameter of about 120 nm, the ADOP solid is a stack of lamellar crystals with a diameter of about 700 nm, and the ADDP solid is a stack of irregularly shaped lamellar crystals. Also, as the length of carbon chains in the molecular structures increases, the degree of agglomeration of aluminum dialkylphosphinates crystal particles becomes higher.

SEM images of (a) ADEP, (b) ADOP, and (c) ADDP.
Optimization of the synthetic conditions for the preparation of ADOP
Synthesis of SDOP
The effect of the molar ratio of the substrates 1-octene and SHP on the yield of SDOP is shown in Figure 5(a). The optimum molar ratio of 1-octene to SHP was investigated under the following conditions: reaction temperature = 95 °C, reaction time = 24 h, and BPO amount = 4%. The ratio of 1-octene to SHP gradually changed from 2:1 to 4:1. It can be seen from the curve that when the ratio of 1-octene to SHP increased from 2:1 to 2.5:1, the yield of SDOP increased significantly and the maximum yield (66%) was obtained at a molar ratio of 3:5. This showed that increasing the amount of 1-octene can effectively improve the conversion of SHP and improve the yield of the target product. However, when the molar ratio continued to increase, the yield of SDOP started to decrease instead, which may be due to increased self-polymerization of the excess alkene, which was more likely to cause the formation of by-products. Therefore, the molar ratio of 1-octene to SHP of 2.5:1 was determined as the optimum conditions for subsequent experiments.

(a) Effect of the molar ratio on the yield of SDOP (reaction conditions: 50-mmol SHP, 2-mmol BPO, 95 °C, 24 h). (b) Effect of the reaction time on the yield of SDOP (reaction conditions: 50-mmol SHP, 250-mmol 1-octene, 2-mmol BPO, 95 °C).
Figure 5(b) shows the effect of the reaction time on the yield of SDOP. The optimum reaction time was investigated under the following conditions: ratio of 1-octene to SHP = 2.5:1, reaction temperature = 95 °C, amount of BPO = 4%. The reaction time ranged from 8 to 40 h. It was observed that when the reaction time was increased from 8 to 16 h, the yield of SDOP increased sharply. When the time continued to increase, the yield of the target product no longer improved and slightly decreased. It was inferred that when 1-octene and BPO were added to the reaction system at a rapid rate, the content of unreacted alkene in the reaction system increased, which increased the possibility for the alkene to self-polymerize due to initiation by the initiator. When the reaction time was longer than 16 h, the addition rate of the alkene and BPO was kept slow enough to make sure that the alkene could react with SHP in time after entering the reaction system, so a better yield of product was achieved. However, prolonging the reaction time may also increase the production of by-products. Hence, 16 h was selected as the best reaction time.
As illustrated in Figure 6(a), the variation in the trend of SDOP yield with respect to the reaction temperature was investigated. The optimum reaction temperature was examined based on a ratio of 1-octene to SHP of 2.5:1, the reaction time was 16 h, and the amount of BPO was 4%. It can be observed from the curve that when the reaction temperature was 85 °C, the yield of SDOP was quite low (only 36%). When the reaction temperature continued to rise to 95 °C, the yield increased significantly to 67%. The maximum yield was 72%, achieved at 105 °C. On continuing to increase the temperature, the yield of the target product no longer increased, and even decreased slightly and then changed only marginally. Therefore, 105 °C was selected as the optimum temperature for the reaction.
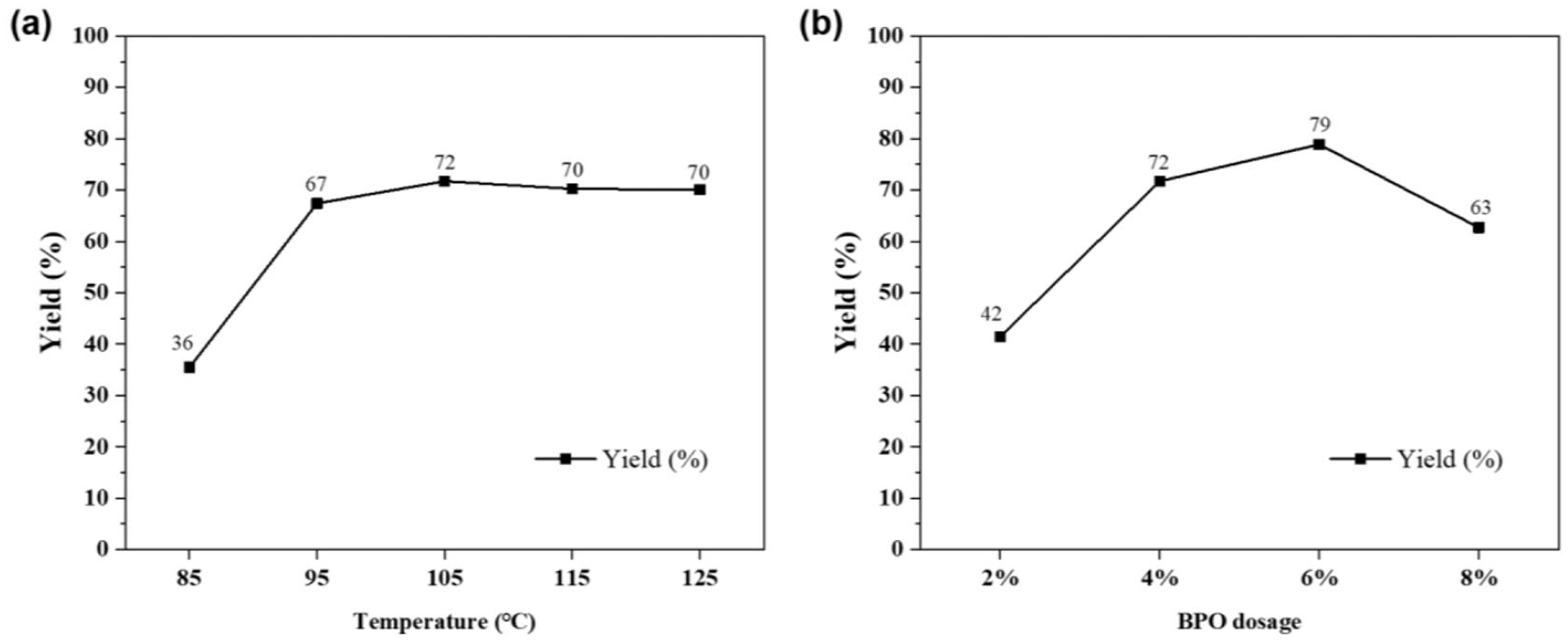
(a) Effect of the reaction temperature on the yield of SDOP (reaction conditions: 50-mmol SHP, 250-mmol 1-octene, 2-mmol BPO, 16 h). (b) Effect of the initiator dosage on the yield of ADOP (reaction conditions: 50-mmol SHP, 250-mmol 1-octene, 105 °C, 16 h, BPO dosage = n(BPO):n(NaH2PO2)).
The effect of the initiator dosage on the yield of SDOP is depicted in Figure 6(b). The optimum amount of BPO was examined under the following conditions: ratio of 1-octene to SHP = 2.5:1, reaction time = 16 h, and reaction temperature = 105 °C. The molar ratio of BPO to SHP varied from 2% to 8%. When the amount of BPO was 2%, the yield of SDOP was only 42%. On increasing the amount of BPO, the yield increased gradually and reached its highest value when the amount of BPO was 6%. The yield then decreased notably on further increasing the amount of BPO. Thus, it was inferred that a BPO dosage of less than 6% was insufficient, and more initiator needed to be added to provide adequate free radicals. Nonetheless, too many initiators would lead to the occurrence of a “cage effect,” which means that the initiators gather together in a “cage” formed by the surrounding molecules, and the primary free radicals collide easily with each other and cause side reactions to form stable compounds, resulting in the reduction of the efficiency of the initiator and the generation of more by-products. 29
Synthesis of ADOP
SDEP is readily soluble in water, while SDOP and SDDP have poor solubility in water. It was speculated that the length of the n-octyl and n-decyl carbon chains led to their strong hydrophobicity and low solubility in water. Since the aluminum sulfate needed to be dissolved in water, we tried to use a variety of water-miscible organic solvents to dissolve SDOP. It was found that SDOP had poor solubility in acetone and N,N-dimethylformamide, was slightly soluble in 1,4-dioxane, and had good solubility in tetrahydrofuran and ethanol. Therefore, mixed solvents of tetrahydrofuran and water or ethanol and water were adopted as the solvents (the volume ratio of organic solvent and water is 1:1) for the double decomposition reaction, respectively, and ADOP was successfully synthesized. When the mixed solvent of tetrahydrofuran and water was used as the solvent, the yield of ADOP was 94%. When a mixed solvent of ethanol and water was used, the yield was 93%. Considering the economic cost and toxicity, the latter was used as the reaction solvent in subsequent experiments.
Figure 7 displays the effect of the temperature of the metathetical reaction on the yield of ADOP. The optimum reaction temperature was investigated under the following conditions: reaction time = 1 h and reaction solvent = ethanol/water (1:1). As can be seen, the reaction temperature was in the range of 40–80 °C. From the curve, it can be seen that when the temperature rose from 40 to 60 °C, the yield of ADOP increased to a certain extent, and the highest yield was 96% at 60 °C. When the temperature continued to rise, the yield decreased slightly, and then there was no obvious change. Consequently, a reaction temperature of 60 °C was chosen as the optimum temperature for obtaining a high yield of ADOP. Since the yield was sufficiently high and the product was highly pure (see Figures S8–S13 in the Supporting Material), it was not necessary to optimize the other reaction parameters.

Effect of the reaction temperature on the yield of ADOP (reaction conditions: 50-mmol SDOP, 8.33-mmol Al2(SO4)3, 80-mL solvent, 2 h).
Conclusion
In summary, ADEP has been successfully synthesized by free-radical addition under atmospheric pressure, which brings higher safety to the production process and reduces the economic cost. In addition, the synthesis of ADOP and ADDP was also investigated. Under the optimum conditions for the synthesis of ADOP, the total yield reached 76%. The rod-shaped structure of ADEP crystals and the lamellar stacking structure of ADOP and ADDP were observed by SEM. All products demonstrated good thermal stability and the temperatures of 5% weight loss of ADEP, ADOP, and ADDP are 401, 349, and 294 °C, respectively. In consideration of the good thermal stability of ADOP and ADDP, they are considered as potential flame retardants for polymeric materials. This study offers a useful method and guidelines for facilitating the synthesis of aluminum alkylphosphinates on a large scale.
Experimental
Materials and methods
SHP (98%) was purchased from Shanghai Lingfeng Chemical Reagent Co., Ltd. Acetic acid (99.5%), absolute ethanol (99.7%), and sodium hydroxide (96%) were purchased from Sinopharm Chemical Reagent Co., Ltd. Ethylene (99.99%) was purchased from Nanjing Shangyuan Gas Factory. 1-Octene (98%), 1-decene (95%), BPO (99%), 2,2′-azobis(2-methylpropionitrile) (98%), tetrahydrofuran (99%), and aluminum sulfate octadecahydrate (99%) were all purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Ethanol (95%) and deuterium oxide (99.9 atom% D) were purchased from General Reagent Co., Ltd. and Shanghai Macklin Biochemical Co., Ltd., respectively. Sulfuric acid-D2 (D, 99.5%, acidity 90%) in D2O and chloroform-d (D, 99.8%) were purchased from Adamas Reagent Co., Ltd.
FTIR spectral data were recorded on a Nicolet 5700 spectrometer in the range of 400–4000 cm−1. NMR spectra were measured on Bruker Avance III 400 MHz or Bruker Ascend 600 MHz spectrometers. HRMS data were acquired on an Agilent 1260-6224 Spectrometer. TGA data were obtained on a NETZSCH TG 209 F3, employing a heating ramp of 20 °C min−1 under an N2 atmosphere. The microscopic morphologies of the products were observed by SEM (Inspect F50, FEI, USA) at a voltage of 10 kV.
Synthesis of SDEP
SHP monohydrate (100 g, 0.943 mol) was dissolved in acetic acid (400 mL) with stirring, and the mixture was then heated at 90 °C. Ethylene was continuously bubbled into the reaction solution at a volume flow rate of 50 mL min−1. Meanwhile, a solution of BPO (10 g) in acetic acid (240 mL) was added dropwise to the reaction system. Ethylene and the initiator were then added completely over 43 h and the mixture was cooled to room temperature. The acetic acid was removed by evaporation in vacuo and the residue was then dried in vacuo at 100 °C for 24 h to obtain SDEP (105.0 g, 77% yield) as a white solid. m.p. > 320 °C. 1H NMR (600 MHz, D2O): δ 1.57–1.48 (m, 4H), 1.10–1.01 (m, 6H). 13 C NMR (150 MHz, D2O): δ 21.5 (d, J = 91.3 Hz), 5.9 (d, J = 4.8 Hz). 31P NMR (243 MHz, D2O): δ 50.02–49.47 (m). HRMS (ESI): m/z [(M-Na)-H]− calcd for [C4H10O2P]: 121.04184; found: 121.04312.
Synthesis of ADEP
SDEP (105.01 g, 0.729 mol) was dissolved in deionized water (600 mL) and the pH value of the solution was adjusted to between 7 and 8 with a 40% aqueous sodium hydroxide solution. After heating the reaction mixture to 90 °C, 200 mL of an aqueous solution containing aluminum sulfate octadecahydrate (104.8 g, 0.157 mol) was added dropwise over 1 h. After stirring at 90 °C for a further 2 h, the mixture was cooled to room temperature. The solid precipitate was separated by filtration, washed with distilled water (3 × 800 mL), and then dried in vacuo at 120 °C for 24 h to afford ADEP (87.5 g, 92%) as a white solid. m.p. > 320 °C. 1H NMR (400 MHz, D2SO4): δ 2.44–2.32 (m, 4H), 1.50–1.38 (m, 6H). 13 C NMR (151 MHz, D2SO4): δ 18.7 (d, J = 85.9 Hz), 4.9 (d, J = 5.0 Hz). 31P NMR (162 MHz, D2SO4): δ 84.23. FTIR (KBr, cm−1): 3435, 2960, 2880, 1462, 1409, 1271, 1232, 1153, 1079, 777.
Synthesis of SDOP
SHP monohydrate (5.3 g, 0.05 mol) was dissolved in acetic acid (20 mL) with stirring. The solution was then heated to 105 °C. 1-Octene (14.0 g, 0.125 mol) and a solution of BPO (0.727 g) in acetic acid (13 mL) was added dropwise to the reaction mixture. 1-Octene and the initiator were then added completely at the same time over 16 h. Then, the resulting mixture was stirred at 105 °C for a further 2 h, and was cooled to room temperature. The acetic acid was removed by evaporation in vacuo and the residue was washed with ethanol (2 × 100 mL) and water (2 × 100 mL), and then dried in vacuo at 70 °C for 24 h to afford SDOP (12.3 g, 79% yield) as a white solid. m.p. > 320 °C. 1H NMR (600 MHz, CDCl3): δ 1.60–1.42 (m, 8H), 1.35–1.12 (m, 20H), 0.88 (t, J = 7.0 Hz, 6H). 13 C NMR (150 MHz, CDCl3): δ 32.1, 31.7, 31.6, 30.2, 29.5, 23.0, 22.9, 14.2. 31P NMR (243 MHz, CDCl3): δ 59.73. HRMS (ESI): m/z [(M-Na)-H]− calcd for [C16H34O2P]: 289.22964; found: 289.23100.
Synthesis of ADOP
SDOP (5.0 g, 0.016 mol) was dissolved in 80 mL of a mixed solvent of ethanol and water (ethanol/water = 1:1) with stirring, and the pH value of the solution was adjusted to between 7 and 8 with a 40% aqueous sodium hydroxide solution. The reaction mixture was then heated to 60 °C and 10 mL of an aqueous solution containing 1.78 g of aluminum sulfate octadecahydrate was added dropwise over 1 h. After stirring at 60 °C for a further 1 h, the mixture was cooled to room temperature. The resulting precipitate was separated by filtration, washed several times with ethanol (100 mL) and water (2 × 100 mL), and then dried in vacuo at 120 °C for 24 h to afford ADOP (4.59 g, 96%) as a white solid. m.p. > 320 °C. 1H NMR (400 MHz, D2SO4): δ 1.88–1.72 (m, 8H), 1.62–1.50 (m, 4H), 1.48–1.35 (m, 16H), 0.98 (t, J = 6.7 Hz, 6H). 31P NMR (243 MHz, D2SO4): δ 88.16. FTIR (KBr, cm−1): 3435, 2926, 2855, 1467, 1406, 1239, 1274, 1156, 1085.
Synthesis of SDDP
SHP monohydrate (5.3 g, 0.05 mol) was dissolved in acetic acid (20 mL) with stirring. The solution was then heated to 105 °C. 1-Decene (17.5 g, 0.125 mol) and a solution of BPO in acetic acid (0.727 g of BPO dissolved in 13 mL of acetic acid) was added dropwise to the reaction solution. 1-Decene and the initiator were then added completely at the same time over 16 h. Then, the resulting mixture was stirred at 105 °C for a further 2 h, and was cooled to room temperature. Acetic acid was removed by evaporation in vacuo and the residue was washed with ethanol (2 × 100 mL) and water (2 × 100 mL), and then dried in vacuo at 70 °C for 24 h to give SDDP (13.91 g, 76% yield) as a white solid. m.p. > 320 °C. 1H NMR (600 MHz, CDCl3): δ 1.55–1.41 (m, 8H), 1.32–1.22 (m, 28H), 0.88 (t, J = 7.0 Hz, 6H). 13 C NMR (150 MHz, CDCl3): δ 32.0, 31.1, 31.0, 29.7, 29.6, 29.5, 29.4, 22.8, 21.7, 14.2. 31P NMR (243 MHz, CDCl3): δ 59.48. HRMS (ESI): m/z [(M-Na)-H]− calcd for [C20H42O2P]: 345.29279; found: 345.29830.
Synthesis of ADDP
SDDP (5.0 g, 0.038 mol) was dissolved in 150 mL of a mixed solvent of tetrahydrofuran and water (tetrahydrofuran/water = 1:1) with stirring and the pH value of the system was adjusted between 7 and 8 with a 40% aqueous sodium hydroxide solution. The reaction mixture was heated to 60 °C, and 10 mL of an aqueous solution containing 4.20 g of aluminum sulfate octadecahydrate was added dropwise over 1 h. After stirring at 60 °C for a further 1 h, the reaction mixture was cooled to room temperature. The resulting solid precipitate was separated by filtration, washed several times with ethanol (100 mL) and water (2 × 100 mL), and then dried in vacuo at 120 °C for 24 h to afford ADDP (12.16 g, 91%) as a white solid. m.p. > 320 °C. FTIR (KBr, cm−1): 3453, 2926, 2855, 1467, 1406, 1156, 1085.
Supplemental Material
sj-docx-1-chl-10.1177_17475198211073275 – Supplemental material for Synthesis of aluminum alkylphosphinates under atmospheric pressure
Supplemental material, sj-docx-1-chl-10.1177_17475198211073275 for Synthesis of aluminum alkylphosphinates under atmospheric pressure by Mengting Zhang, Shengbin Shi, Yan Lei, Liuyu Gu, Lijing Gao and Guomin Xiao in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Key R&D Program of China (No. 2019YFB1504003).
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
