Abstract
Investigation of the non-polar (rich in chlorophyll) and polar fractions of the ethyl acetate extract of Gymnosporia senegalensis (Lam.) Loes. leaves led to the isolation of nine compounds, including one new megastigmane derivative, (3R*,5S*,6R*,7E,9ξ)-7-megastigmene-3,6,9-triol-3-O-β-D-(6′-O-galloyl)glucopyranoside (

Introduction
The continuous dissemination of multi-drug-resistant (MDR) strains drastically reduces the efficiency of antibiotics and consequently increases the frequency of therapeutic failure. 1 In many regions affected by these MDR strains, local and indigenous plants are often the only available means of treating such infections. Herbal antioxidant compounds play a defensive role by preventing the generation of free radicals. This action is therefore extremely beneficial to relieve infectious diseases generating free radicals as well as diseases caused by oxidative stress such as cardiovascular diseases, diabetes, inflammation, degenerative diseases, cancer, anemia, and ischemia.2,3
Medicinal plants typically contain mixtures of different chemical compounds that may act individually, additively, or in synergy to improve health. It has been suggested that these compounds may have been synthesized by the plant as part of its defense system. Plants that survive under harsh and variable environmental conditions are expected to accumulate a wide range of secondary metabolites. Gymnosporia senegalensis (Lam.) Loes. (Celastraceae) is one of such a plant which is traditionally used as astringent, to treat toothaches, stomachaches, typhoid, malaria, and inflammatory disorders. It is widely distributed in semi-arid regions of Africa.4,5 G. senegalensis (Lam.) Loes. is also called Maytenus senegalensis (Lam.) Exell.
6
Previous biological studies reported that extracts from various parts (stem, leaves, bark, and root) of G. senegalensis exhibited anti-inflammatory,5,7,8 antimicrobial,5,8 antiplasmodial,
9
antioxidant,
8
and antiproliferative
8
activities. In addition, compounds of different classes, including alkaloids, terpenoids, sugars, lignans, and flavonoids, have been reported from M. senegalensis.5,10 Tatsimo et al.
11
recently reported the isolation of glycerol, cyanogenic, and phenolic derivatives from leaves of G. senegalensis. In our effort to investigate the potential of G. senegalensis as source of lead compounds, we fractionated the non-polar (rich in chlorophyll) and polar fractions of ethyl acetate leaf extract. We report herein the isolation, antimicrobial, and antioxidant activities of one new megastigmane derivative (
Results and discussion
Chemical analysis
Phytochemical investigation of chlorophyll-rich and polar fractions from ethyl acetate extract of G. senegalensis leaves led to the isolation of nine compounds including one new megastigmane (
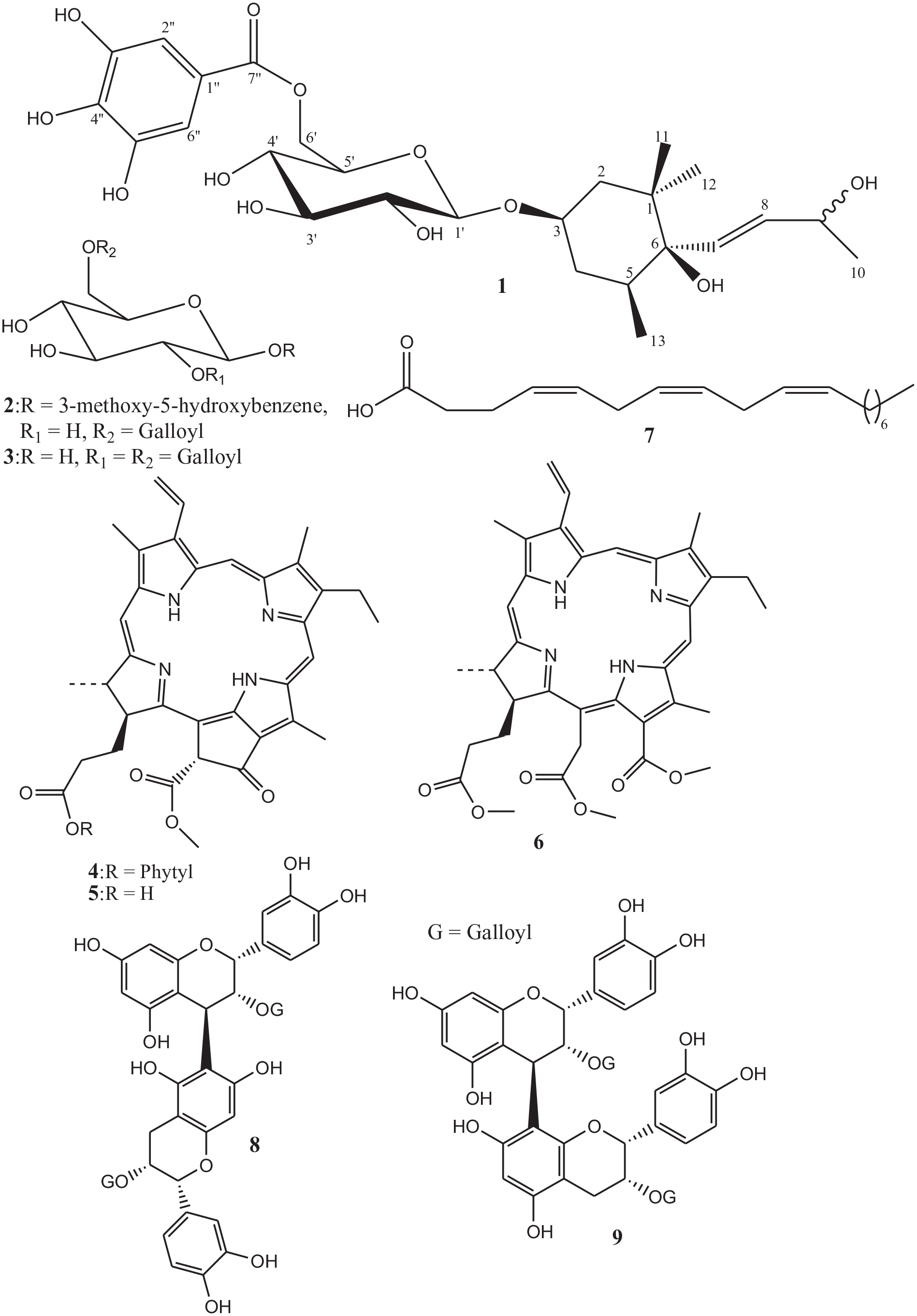
Structure of isolated compounds (
Compound

Connectivities and correlations deduced from 2D spectra.

MS/MS fragmentation pattern of compound
Antimicrobial and antioxidant activities
The extracts from G. senegalensis and their isolated compounds
Based on the antimicrobial cut-off points of pure compounds previously defined,
24
compounds
The results of the radical-scavenging activity (Figure S36) demonstrated that compound
Experimental
General experimental procedures
A JEOL ECX400 (1H, 400 MHZ; 13C, 100 MHZ) Delta spectrometer (JEOL, Tokyo, Japan) with TMS as an internal standard was used to measure NMR spectra. HR-MS and LC-MS spectra were recorded on a hybrid ion trap time-of-flight (IT-TOF) mass spectrometer (Shimadzu, Kyoto, Japan). A JASCO P2100 digital polarimeter (JASCO International Co. Ltd., Tokyo, Japan) and a JASCO FT/IR-460 Plus spectrophotometer (JASCO International Co. Ltd.) were used to measure optical rotations and IR spectra, respectively. The stationary phases for column chromatography (CC) were either Sephadex LH-20 (GE) or silica gel (Wakogel® C-200 or 400HG, Wako Pure Chemical Industries, Ltd., Osaka, Japan), while Biotage® SNAP Ultra 10 or 100 g (25 µm) Cartridge pack (silica gel packed column) were used for medium pressure liquid chromatography (MPLC) on a Biotage Isolera™ One apparatus (Biotage, Uppsala, Sweden). TLCs were carried out on precoated glass TLC silica gel 60F254 or RP-18 F254s plates (0.25 mm thickness, Merck, Darmstadt, Germany). Precoated glass PLC silica gel 60F254 plates (0.5 mm thickness) were used for Preparative TLCs. MPLC was performed using the following solvent systems (detection at 210 and 254 nm) as described by Tatsimo et al., 2019; Y1 (flow rate: 12 mL min−1; volume: 8 mL): water (A)-methanol (B):30% B for 8:45 min, 30% B to 100% for 23:45 min, and 100% B for 25 min; Y2 (flow rate: 12 mL min−1; volume of each fraction collected: 15 mL); H2O (A)-MeOH (B): 40% B for 7:00 min, 40% B to 100% for 10:30 min, and 100% B for 10 min.
Sample collection
Leaves of G. senegalensis were collected in November 2015 at Mindif, Far North, Cameroon, and identified by Mr Tapsou, a botanist at IRAD (Institut de Recherche Agricole et de Développement) of Maroua, Cameroon. A voucher number No 22314/SRCam was obtained at the National Herbarium, Yaoundé, Cameroon.
Extraction and isolation of constituents
The extraction and fractionation were carried out as previously described.
11
Previously unstudied chlorophyll fractions, A (476.2 mg) and B (277.8 mg), and polar subfractions G [G5 (200.9 mg) and G6 (211.5 mg)] of ethyl acetate extract were investigated. Fraction A was subjected to SiO2 gel CC eluted with hexane-acetone gradient to give 150 subfractions of 12 mL each. Subfraction A121-150 (21.2 mg) was purified on reverse phase MPLC using system Y1 described above to give
(3R*,5S*,6R*,7E,9ξ)-7-megastigmene-3,6,9-triol-3-O-β-D-(6′-O-galloyl) glucopyranoside (
Antimicrobial evaluation
The antimicrobial activity was performed against one Gram-positive (Staphylococcus aureus ATCC25923) and three Gram-negative (Pseudomonas aeruginosa ATCC27853, Escherichia coli S2(1), Shigella flexneri SDINT) bacteria, and three yeast strains of Candida albicans ATCC10231, C. tropicalis PK233, and Cryptococcus neoformans H99. These microorganisms were taken from our local stocks. The broth microdilution method was used to determine the MICs and MMC as previously described. 28 Ciprofloxacin (Sigma-Aldrich, Steinheim, Germany) and nystatin (Merck, Darmstadt, Germany) were used as positive controls for bacteria and yeasts, respectively.
Antioxidant assay
Ferric reducing antioxidant power (FRAP) assay
The FRAP was performed by the Fe3+–Fe2+ transformation in the presence of extracts and compounds as previously described. 29 BHT was used as a standard antioxidant.
2,2'-diphenyl-1-picrylhydrazyl (DPPH) free radical–scavenging assay
The free radical–scavenging activity of extracts and isolates was carried out as previously described. 30 Vitamin C was used as a positive control.
Statistical analysis
The experimental results were analyzed by one-way analysis of variance followed by Waller-Duncan post hoc test. The results were expressed as the mean of three replicates ± standard deviation (SD). Differences between means were considered significant at p-value <0.05. The mean comparisons were performed using the Statistical Package for Social Sciences (SPSS, version 12.0) software.
Conclusion
The chemical investigation of ethyl acetate extract of G. senegalensis leaves led to the isolation of nine compounds, including one new megastigmane derivative. The isolates showed weak to high antimicrobial activities, with phaeophytin A (
Supplemental Material
sj-doc-1-chl-10.1177_17475198211072498 – Supplemental material for A new megastigmane, known porphyrinic and galloylated bioactive derivatives from the leaves of Gymnosporia senegalensis
Supplemental material, sj-doc-1-chl-10.1177_17475198211072498 for A new megastigmane, known porphyrinic and galloylated bioactive derivatives from the leaves of Gymnosporia senegalensis by Simplice Jöel Tatsimo Ndendoung, Jean-De-Dieu Tamokou, Kazufumi Toume, Léopold Havyarimana, Steve Endeguele Ekom and Katsuko Komatsu in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are grateful to the Matsumae International Foundation (MIF) for a postdoctoral fellowship awarded to Dr Tatsimo NSJ at the University of Toyama, Japan, and to the University of Maroua, Cameroon, for sponsoring some consumables used in this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
