Abstract
In this paper, novel synthetic methods, including microwave O-alkylation, were used to produce several chrysin derivatives. These compounds were purified, characterised and tested on different cell lines and bacterial strains. From this family, 7-(2,4-dinitrophenoxy)-5-hydroxy-3-phenyl-4H-chromen-4-one (

Introduction
In the search for new drugs to overcome drug-resistant infections and cancers, phytochemicals have become the go-to source for inspiration. In plants, isoprenoids serve as the backbone from which many phytochemicals are biosynthesised via the cytosolic mevalonate (MVA) and the 2C-methyl-D-erythritol-4-phosphate (MEP) pathways. 1 Some phytochemicals, like flavonoids, are synthesised via the phenylpropanoid pathway, they exhibit both antimicrobial and anticancer activities. 2 The antimicrobial activity could be a result of impairment of the cell membrane integrity and cell agglutination. 3 The anticancer activity could be due to modulation of various mechanisms such as angiogenesis, apoptosis, metastasis, differentiation and cell proliferation.4,5 The increase in research targeted at developing new drugs for cancer and infectious diseases has gained momentum in the last decades.
Chrysin is a flavonoid predominantly present found in honey, blue passionflower and propolis. 6 In vitro studies on chrysin and chrysin derivatives have demonstrated interesting biological activity. 7 Chrysin has shown potential anticancer activities on several cancer cell lines including those derived from cancers of the prostate, pancreas, thyroid, glioblastoma, liver, cervical, nasopharyngeal, breast, lungs and haematological cancers.6,8 The mode of action of chrysin has been shown to be via apoptosis, cell proliferation and removal of inflammatory responses, suppression of NF-ĸB and angiogenesis.9–11 Antibacterial activity of flavonoids, including chrysin, has been reported against Gram-positive and Gram-negative bacteria.12–16
7-O-Alkylchrysin derivatives have been explored for anticancer activity. 17 Our group has extensive expertise in synthesising different flavonoid derivatives.18–20 In this paper, we explored the effect on the biological activity of different functional groups added on the position 7 in chrysin. To compare different moieties and carbon length chains, we synthesised both known and novel chrysin derivatives. 7-O-Bromochrysin and 7-O-alkylchrysin derivatives were explored employing green microwave processes in majority of the cases. The aim of this study was to investigate the antibacterial and anticancer activity of a group of chrysin derivatives with a range of different substituents.
Results and discussions
Chemistry
Chrysin (shown in Figure 1) was used as the core flavone structure, modifications were undertaken at the 7-hydroxyl group using different methods. Different reaction solvents such as acetone, acetonitrile, dimethylformamide as well as different equivalents were used for addition of the linkers. Potassium carbonate (K2CO3) has been widely used in O-alkylation synthetic methods as a base reagent with the intention of producing a phenoxide anion on the chrysin scaffold. 21 This nucleophile will then react with the electrophilic R groups (Br-alkyl groups). Different equivalents were used with some reactions favouring 2 equiv. or 4 equiv. to chrysin. Microwave chemistry has been at the front of green chemistry synthetic methods as it allows for reduction of solvent used, time and electricity as well as making some reactions more favourable. 18 O-alkylation in chrysin is favoured on position 7, this is due to the interaction between the carbonyl oxygen in position 4 and the hydroxyl in position 5. 19 The proximity of these two groups allows the formation of hydrogen bonding which requires harsher conditions for O-alkylation. Steric hindrance also plays an important role, while the hydroxyl in position 7 is free to interact with bulky groups, only small groups would be possible to interact with the hydroxyl in position 5.22–24
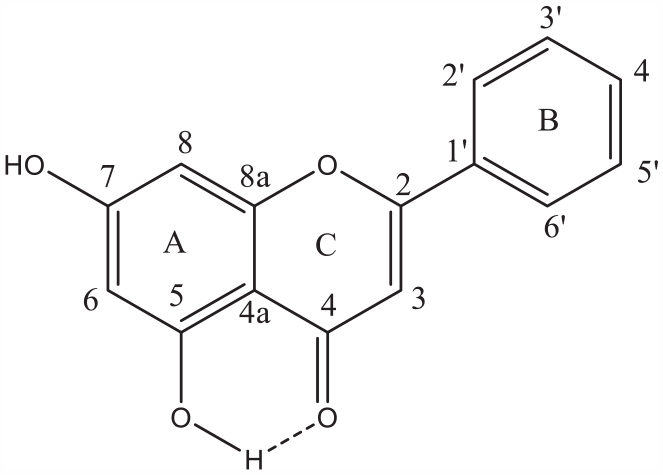
Chrysin, showing the traditional numbering of positions.
We have explored all these conditions and synthetised a range of chrysin derivatives using stirring or microwave techniques, selecting the ones that presented the highest yields. Compounds

General method for the synthesis of chrysin derivatives. Common numbering of protons on flavonoids is shown on chrysin structure. Carbon chains are numbered in the supplementary material. Yields presented in %: C1 (80), C2 (52), C3 (46), C4 (75), C5 (49), C6 (47), C7 (56) and C8 (94).
7-O-alkylchrysin derivatives
Introduction of alkyl chains has the potential to form more lipophilic compounds, which could enhance the entrance of the compound into cells. This was explored for both carbon alkyl chains as well as halogen species opening up the possibility to form additional targets on further reaction.
27
Compound
A total of eight chrysin derivatives were synthesised and characterised. The structures of the products were confirmed by 1H NMR, 13C NMR, IR and Mass Spectral data. All the compounds were taken for biological studies.
Antibacterial activity
In this study, we investigated the activity of the new derivatives on Gram-negative bacteria (Escherichia coli, Pseudomonas aeruginosa, Pseudomonas fluorescens and Klebsiella pneumoniae) and Gram-positive bacteria (Staphylococcus aureus and Enterococcus). E. coli dwell mainly in the intestine of humans and animals; the pathogenic strains can cause bloody diarrhoea 23 vomiting and severe abdominal cramp. It is one of the major causes of food poisoning.29,30 The extended-spectrum β-lactamase (ESBL)-producing E. coli are of major importance due to their antibiotic resistance. ESBL are enzymes that confer antimicrobial resistance to most β-lactam antibiotics such as the monobactam aztreonam, cephalosporins and penicillins. 31 P. aeruginosa causes ventilator-associated pneumonia and other nosocomial infections as well as sepsis syndrome.32,33 Recently, the World Health Organization (WHO) has identified P. aeruginosa as a priority pathogen due to the threat of extremely drug-resistant infections. 34 P. fluorescens is rarely a cause of human disease; however, it might affect patients with a compromised immune system caused by cancer, cases have been reported of contaminated blood given through transfusions. 35 This strain also produces antibiotics like mupirocin which are used in creams to treat skin MRSA-active infections. 36 K. pneumoniae is present in the intestine, skin and mouth as a normal flora of these tissues, and it can damage the lungs (alveoli) if inhaled. They are resistant to most drugs, 37 and it is the third microorganism commonly isolated in blood cultures of patients with sepsis. 38 S. aureus is found on the skin, respiratory tract and nose, and is responsible for food poisoning, abscess, skin and soft tissue infections, wound and respiratory infections. It is also responsible for toxic shock syndrome, cellulitis, impetigo and boils.39,40 The introduction of penicillin reduced the high mortality but led to the development of β-lactamases which destroy penicillin. A semisynthetic penicillin analogue – methicillin was introduced; however, S. aureus resistant to methicillin emerged rapidly (MRSA). 41 Enterococcus is associated with nosocomial infections as it rapidly acquires antibacterial resistance. 42 The Candida albicans fungus is a lifelong, harmless commensal member of the normal human microbiome. However, C. albicans can cause infections that range from superficial infections of the skin to life-threatening systemic infections under certain circumstances. 43 Several activities and factors have been identified which contributes to the pathogenic nature of this fungus. This includes biofilm formation, secretion of hydrolases, molecules which mediate invasion and adhesion to the host cells, phenotypic switching and thigmotropism. 43 C. albicans can cause two major infections in humans: superficial infections and oral candidiasis and life-threatening systemic infections which could spread into vital organs. 44 C. albicans (up to 75%) is present in the oral cavity and remains benign in normal humans but could cause recalcitrant oral cavity infection in immunocompromised individuals. 43
Minimum Inhibitory Concentration (MIC) is defined as the lowest concentration of an ingredient that prevents visible growth of bacteria, used to evaluate the antimicrobial efficacy of compounds. As shown in Table 1, chrysin showed very weak antibacterial activity on the tested strains. This is in agreement with different studies, in where several flavonoids including chrysin, have shown modest studies.
44
Effect of chrysin derivatives on Escherichia coli, Pseudomonas aeruginosa, Klebsiella, Staphylococcus aureus, Enterococcus and Bacillus cereus following 72 h of drug treatment MIC is recorded in µg mL−1.

MIC: Minimum Inhibitory Concentration; Chr: chrysin; G: gentamicin, this aminoglycoside has a wide range of antibacterial activity making it ideal as a positive control on activity tests;44,45 F: fluconazole, this antifungal agent is widely used to treat C. albicans infection presenting a variable range of MIC. 46
No observed inhibition at MIC values ⩽250 µg mL−1.
MIC values are expressed as a mean value ± SD from two independent experiments performed in triplicate.
Li et al. 46 had previously synthesised chrysin derivatives containing 3-carbon spacers attached to different positions. Their results on 7-OH derivatives showed they were the most effective with MIC values ranging from 3.13 to 50 μg mL−1 against S. aureus and E. coli, respectively. Another study using piperazine chrysin derivatives showed IC50 μg mL−1 values in the range of 1.30 to 4 for S. aureus, 1.15 to 7 for E. Coli and 10 to >50 for P. aeruginosa. 47 Only a handful of studies include chrysin or chrysin derivatives include investigation on MRSA, Alhadrami et al. 48 reported MIC values of 15 and 250 μg mL−1 for chrysin and hesperidin, a chrysin derivative, respectively. Similarly to the MRSA, flavonoids including chrysin have not been extensively studied on P. fluorescens; some reports related more to the food industry and packaging have reported propolis presenting antibacterial effect for P. aeruginosa and P. fluorensces. 49 Zhu et al. 50 reported the synthesis of several chrysin derivatives with antimicrobial activity; results of MIC were in the range of 50 μg mL−1 for K. pneumoniae and 3 mg mL−1 for C. albicans.
Anticancer activity
The growth inhibition activities of this family of chrysin derivatives were determined in a series of tumour and non-tumour cell lines. These cell lines represent some of the most common cancer types including leukaemia (myeloid K562 and lymphoid MOLT-4), colorectal (HCT-116 and Caco-2), breast (MCF-7 and MDA-MB 468), hepatocellular cancer (HepG2), lung cancer (A549), malignant mesothelioma (Mero-14), along with an immortalised normal bronchial epithelium cell line (BEAS-2B). BEAS-2B was used as a control to determine if the growth inhibition activity was specific to cancerous cell lines, an ideal anticancer agent would not be active on non-cancerous cells.
The results are shown in Table 2. Chrysin was very active in our studies with IC50 values ranging from 3 to 50 µM, including the non-cancerous cell line BEAS-2B. On colorectal cancer model HCT-116, all compounds were relatively active, especially
Effect of chrysin derivatives on the growth of HepG2, MCF-7, HCT116, MOLT-4, K562, A549, Mero-14, MDA-MB468 and BEAS-2B cell lines following 72 h of drug treatment.

SD: standard deviation; Chr: chrysin; CPZ: chlorpromazine, positive control.Values are the concentration required to cause a 50% decrease in cell growth compared to untreated control (IC50).
No bioactivity at doses >200 µM.
IC50 values are expressed as a mean value ± SD from two replicates.
The results showed that chrysin was an active compound, but it is also toxic to non-cancerous cells. From the derivatives,
Although anticancer agents bearing a nitro moiety are generally considered toxic, some compounds with NO2 are currently being investigated, for example, misanidazole.
56
The fact that compound
Chrysin alongside derivatives have been studied on our selection of cell lines. Chrysin has shown IC50 higher than 200 µM on leukaemia cells lines including K562 and MOLT-3; MOLT-3 and MOLT-4 are cell lines originated from the same patient with T-cell acute lymphoblastic leukaemia, having some differences in CD (cluster of differentiation) expression and chromosomal rearrangement.57,58 Samarghandian et al. 59 reported IC50 values close to 20 µM for breast cancer cell line MCF-7 while similar value was reported by Androutsopoulos et al. 60 for MDA-MB468. On a different study, Samarghandian et al. 61 reported IC50 values in the order of 40 µM for chrysin on A549. Although chrysin has been tested for anticancer activity pathway elucidation on colon cancer cell models, not many have reported quantifiable values. 62 A study using Chinese and Brazilian propolis reported IC50 values higher than 50 µM for Caco-2 and 12–40 µM for HCT116, respectively. 63 Chrysin has also been investigated on hepatic model HepG2, with Zhang et al. 64 reporting an IC50 value of 98 µM. To the best of our knowledge, no studies using flavonoids, propolis nor chrysin have been reported through the scientific literature, so we present here first ever studies of chrysin and its derivatives on this mesothelioma cell line.
Lipophilicity (clogP)
Lipophilicity has a significant impact on various drug properties including absorption, distribution and permeability. 66 For a drug to reach its target, needs to penetrate the lipid bilayer of the cellular membranes, and one of the first barriers a drug encounters is situated in the enterocytes in the intestinal epithelia. In general, a rule of thumb indicates that lipophilic drugs have good absorption. 67
Lipophilicity indicates partition between two immiscible phases. In the case of enterocytes, the drug needs to be absorbed through the apical side, for this being a lipophilic molecule might be helpful; but once the drug is inside, it needs to travel through the cytoplasm to be able to exit through the basolateral side of the cell. In this situation, there needs to be a delicate equilibrium between lipophilicity and lipophobicity, if the molecule is highly lipophilic, it might not be able to leave the lipid layer. One way of early detection during the drug design process is to employ the Lipinski’s Rule of Five, for which the calculated log10P is ⩽5. 68 Lipophilicity can be predicted using software, for example, ChemDraw, where a highly lipophilic drug would have a clogP >3. 69 Highly lipophilic drugs would also have solubility problems. The clogP of several molecules in the market for oral delivery present clogP in the ranges of −1 to 5, with the maximum between 1.5 and 3. 70 In the case of antibacterial commercial drugs, it appears that general physicochemical properties for these compounds are outside the range of other types of pharmaceutical molecules. An in-depth analysis of marketed antibacterial molecules showed that for around 34.4% of them, their logP were in the range of 0–5. 71
Chrysin has a clogP of 2.29 and is widely known to have absorption problems, therefore affecting its bioavailability, calculated to be 0.003%–0.02% through the oral route of administration.72,73
Conclusion
Chrysin is certainly a promising molecule for anticancer studies and can be an excellent scaffold for drug design as some derivatives offer incredible biological activities.50,74 The synthetic procedures employed to produce the chrysin derivatives were designed and optimised to produce high yields of the novel compounds utilising greener chemistry conditions. In our microbiological studies,
In our anticancer studies, we tested the compounds on a wide range of cancer models, like breast, hepatic, lung, leukaemia, colon cancer and malignant mesothelioma as well as non-cancerous cells. While chrysin was very active on the cancer models, it also showed toxicity to non-cancerous cells, maybe due to lack of selectivity for any target. Again,
Some antibiotics have been shown to also exert anticancer activity, so during early drug design it is important to assess new chemical entities for activity on both types of targets.75,76 The identification of the lead
Experimental
Materials and instruments
Unless stated otherwise all chemicals and reagents were used as received. Synthetic reagents were purchased from Sigma-Aldrich, Thermo Fisher Scientific, Apollo Scientific or TCI-UK. 1H and 13C NMR spectra were measured on a Bruker Avance DPX 400 MHz spectrometer using DMSO-d6 or CDCl3 as the solvent, and tetramethylsilane (TMS; δ = 0) as the internal reference. All solvents used for NMR analysis were purchased from Cambridge Isotope Laboratories Incorporated. J-values are given in Hz. High-Resolution Mass Spectrometry for novel compounds were outsourced, and they were analysed using the positive electrospray ionisation time-of-flight mass spectrometry (TOF MS ES) on Waters I-Class UPLC at Cambridge Analytical Services, University of Cambridge, UK. Calculated M + H were performed using ChemDraw 20.1 software. Infrared spectra were measured on a Thermo Scientific Nicolet iS10 and melting point analysis was performed on a Stuart Melting Point SMP20. Microwave-assisted reactions were performed in a CEM Discover SP microwave reactor. Silica gel chromatography was performed using silica gel 60 Å with a pore size of 40–63 µm (Fluorochem Limited, Glossop, UK). Silica thin-layer chromatography was performed on pre-coated aluminium sheets with a 0.2-mm thickness obtained from Thermo Fisher Scientific, UK. Anhydrous tetrahydrofuran was distilled over sodium and benzophenone prior to use. All other anhydrous solvents were purchased from Thermo Fisher Scientific.
Method A, synthesis of C1, C2 and C3
Chrysin (0.12 g, 4.7 mmol) was added to a suspension of potassium carbonate (4.0 equiv., 0.52 g, 8.4 mmol) in acetone (10.0 mL). 4-Bromobutyl acetate (2.0 equiv., 184 µL, 9.4 mmol), 1-(bromomethyl)-3,5-dimethylbenzene (2.00 equiv., 0.085 g, 6.4 mmol) or 1-bromo-2,4-dinitrobenzene (116.6 g, 4.7 mmol) were added to synthesise
Method B, synthesis of C4
To a solution of chrysin (0.10 g, 0.40 mmol), potassium carbonate (0.79 mmol; 2.0 equiv.) and tetra-n-butylammonium bromide (0.2 equiv., 0.025 g, 0.08 mmol) in dimethylformamide (10 mL) was added 1-bromo-2-methylpropane (2.0 equiv., 84 μL, 0.99 mmol). The reaction was allowed to stir for 72 h at 50 °C. After 72 h, the mixture was diluted with ice cold water, acidified with HCl (6 N) and extracted with ethyl acetate. The organic layer was concentrated to yield a yellowish-brown solid which was chromatographed on silica gel using ethyl acetate: petroleum ether (20:80 to 100:0) as mobile phase.
Method C, synthesis of C5, C6 and C7
Potassium carbonate (2.00 equiv., 0.26 g, 4.2 mmol) was suspended in acetonitrile (10.0 mL) containing chrysin (0.12 g, 0.47 mmol). Two equiv. of bromoalkane (1-bromooctane (142.6 μL, 9.4 mmol) for
Method D, synthesis of C8
Potassium carbonate (4.00 equiv., 0.52 g, 8.4 mmol) was suspended in acetonitrile (10.0 mL) and stirred in a microwave tube for 30 min. Chrysin (0.12 g, 0.47 mmol) and 1 equiv. of 1,4-dibromopentane (93 μL, 4.72 mmol) were added and the resulting suspension stirred for 24 h at 80 °C. The resulting suspension was allowed to cool to room temperature and poured into crushed ice, leading to the formation of pale-yellow precipitate which was left at 4 °C for 24 h. After 24 h, the precipitate was filtered and washed with ice cold water, followed by petroleum ether, and eluted in silica gel using 2:1 ethyl acetate:petroleum ether, v/v. The solvent was evaporated, and the precipitate dried in an oven for 24 h at 50 °C.
4-((5-hydroxy-4-oxo-2-phenyl-4H-chromen-7-yl)oxy)butyl acetate (C1)
Yield: 80%; m.p. 122–124 °C; clogP: 2.83. 1H NMR (CDCl3, δ ppm) 1.83–1.94 (4H, m, 3"–H2 and 4"–H2), 2.09 (3H, s, 8"–H3), 4.06 (2H, t, J = 4.4 Hz, 5"–H2), 4.24 (2H, t, J = 5.88 Hz, 2"–H2), 6.38 (1H, s, 6–H), 6.51 (1H, s, 8–H), 6.69 (1H, s, 3–H), 7.55 (3H, m, 2'–H, 3'–H, 4'–H), 7.92 (2H, d J = 7.3, 2'–H, 6'–H), 12.9 (1H, s, 5-OH). 13C NMR (CDCl3, δ ppm) 20.9 (8"), 25.3 (3"), 25.6 (4"), 63.9 (5"), 67.9 (2"), 76.7–77.3 (CDCl3), 93.1 (8), 98.5 (6), 105.73), 105.9 (4a), 126.3 (3'/5'), 129.1 (4'), 131.3 (2'/6'), 131.8 (1'), 157.8 (8a), 162.2 (5), 163.9 (2), 164.9 (7), 171.1 (7"), 182.4 (4). IR (cm−1): 1165.42 (C–O–C, large ring C–O stretch), 1031.25, 1242.32 (C–O, phenolic), 1588.12 (C=C aromatic), 1605.21 (C=O, ketone), 1751.27 (OC=O). HRMS (ESI) m/z: calculated for C21H20O6 [M + 1] = 369.1293, found 369.1330.
7-((3,5-dimethylbenzyl)oxy)-5-hydroxy-2-phenyl-4H-chromen-4-one (C2)
Yield: 52%; m.p.: 122–124 °C; clogP: 5.26. 1H NMR (CDCl3, δ ppm) 2.25 (6H, s, 9"–H3, 10"–H3), 4.95 (2H, s, 4"–H2), 6.34 (1H, d, J = 2.4 Hz, 6–H), 6.48 (1H, d, J = 2.4 Hz, 8–H), 6.57 (1H, d, J = 2.7 Hz, 3–H), 6.92 (1H, s, 6"–H), 6.97 (2H, s, 2"–H, 8"–H), 7.45 (3H, m, 3'–H, 4'–H, 5'–H), 7.80 (2H, dd, J = 7.3, 2.6 Hz 2'–H, 6'–H), 12.80 (1H, s, 5-OH). 13C NMR (CDCl3, δ ppm) 23.3 (9"/10"),70.4–77.4 (CDCl3), 93.5 (8), 100.9 (6), 105.9 (3), 105.9 (4a), 125.3 (4"/8"), 126.3 (3'/5'), 129.1 (4'), 130.1 (2'/6'), 131.4 (1'), 131.4 (6"), 135.5 (5"/7"), 136.4 (3"), 157.7 (8a), 162.2 (5), 164.0 (2), 164.8 (7), 182.5 (4). IR (cm−1): 1158.49 (C–O–C, large ring C–O stretch), 1246.45 (C–O, phenolic), 1583.85 (C=C aromatic), 1602.10 (C=O, ketone), 2854.31 (C–H, alkane), 3001.46 (C–H, aromatic). HRMS (ESI) m/z: calculated for C24H22O4 [M + 1] = 373.1395, found 373.1433.
7-(2,4-dinitrophenoxy)-5-hydroxy-3-phenyl-4H-chromen-4-one (C3)
Yield: 46%; m.p. 292–294 °C; clogP: 4.29. 1H NMR (DMSO-d6, δ ppm) 6.71 (1H, D, J = 3.5 Hz, 6–H), 7.11 (1H, d, J = 3.5 Hz, 8–H), 7.19 (1H, s, 3–H), 7.65 (4H, m, 3'–H, 4'–H, 5'–H, 3"–H), 8.12 (2H, d, J = 7.0 Hz, 2'–H, 6'–H), 8.57 (1H, dd, J = 10.5, 3.5 Hz, 4"–H,), 8.98 (1H, d, J = 3.5 Hz, 6"–H), 13.02 (1H, s, 5-OH). 13C NMR (DMSO-d6, δ ppm) 30.7–40.4 (trace cyclohexane/acetone), 93.0 (8a), 102.1 (6), 105.3 (4a), 107.8 (3), 122.0 (6"), 122.5 (3"), 126.6 (3'/5'), 129.2 (4'), 130.2 (2'/6'), 132.5 (1'), 140.7 (7"), 140.8 (5"), 152.4 (1"), 157.1 (5), 160.4 (2"), 161.2 (7), 182.5 (4). IR (cm−1): 1149.55 (C–O–C, large ring C–O stretch), 1248.32 (C–O, phenolic), 1535 (N=O, aromatic) 1593.47 (C=C aromatic), 1623.80 (C=O, ketone). HRMS (ESI) m/z: calculated for C22H16N2O8 [M + 1] = 421.0627, found 421.0661.
5-hydroxy-7-isobutoxy-3-phenyl-4H-chromen-4-one (C4)
Yield; 75%; m.p. 143–145 °C; clogP: 3.78. 1H NMR (CDCl3, δ ppm) 1.03 (6H, d, J = 6 Hz, 4"–H3, 5"–H3), 1.60 (trace water), 2.12 (1H, m, 3"–H), 3.83 (2H, d, J = 6 Hz, 2"–H2), 6.36 (1H, s, 6–H), 6.52 (1H, s, 8–H), 6.68 (1H, s, 3–H), 7.55(3H, d, J = 8.4 Hz, 3'–H, 4'–H, 5'–H), 7.91 (2H, d, J = 12 Hz, 2'–H, 6'–H), 12.78 (1H, s, 5-OH). 13C NMR (CDCl3, δ ppm) 19.2 (4"/5"), 28.1 (2"/3"), 74.9–77.3 (CDCl3), 93.1 (8), 98.7 (6), 105.6 (4a), 105.9 (3), 126.3 (3'/5'), 129.1 (4'), 131.4 (2'/6'), 131.8 (1'), 157.8 (8a), 162.1 (5), 163.9 (2), 165.3 (7), 182.5 (4). IR (cm−1): 1149.55 (C–O–C, large ring C–O stretch), 123.99 cm−1 (C–O, phenolic), 1587.00 (C=C aromatic), 1614.79 (C=O, ketone), 2859.02 (C–H, alkane), 2953.21 (CH3, alkane), 3076.84 (CH, aromatic). HRMS (ESI) m/z: calculated for C19H18O4 [M + 1] = 311.1239, found 311.1278.
5-hydroxy-7-(octyloxy)-3-phenyl-4H-chromen-4-one (C5)
Yield: 49.3%; m.p. 77–80 °C; clogP: 5.47. 1H NMR (CDCl3, δ ppm) 0.92 (3H, t, J = 7.0 Hz, 9"–H3), 1.35 (8H, m, 5"–H2, 6"–H2, 7"–H2, 8"–H2), 1.48 (2H, m, 4"–H2), 1.49 (2H, m, 4"–H2) 1.85 (2H, quint, J = 7.0 Hz, 3"–H2), 4.03 (2H, t, J = 7.2 Hz, 2"–H2), 6.47 (1H, d, J = 4.0 Hz, 6–H), 6.52 (1H, d, J = 4.0 Hz, 8–H), 6.68 (1H, s, 3–H), 7.27 (CDCl3) 7.55 (3H, d, J = 11 Hz, 3'–H, 4'–H, 5'–H), 7.91 (2H, d, J = 9 Hz, 2'–H, 6'–H), 12.9 (1H, s, 5-OH). 13C NMR (CDCl3, δ ppm) 14.1 (9"), 22.7 (8"), 25.9 (7"), 28.9 (6"), 29.2 (4"/5"), 29.3 (3"), 31.8 (2"), 68.7 (1"), 76.7–77.4 (CDCl3), 93.1 (8), 98.6 (6), 105.6 (4a), 105.8 (3), 126.2 (3'/5'), 129.1 (4'), 131.4 (2'/6"), 131.8 (1'), 157.8 (8a), 162.2 (5), 163.9 (2), 165.2 (7), 182.5 (4). IR (cm−1): 1169.51 (C–O–C, large ring C–O stretch), 1272.96 (C–O, phenolic), 1587.89 (C=C aromatic), 1607.79 (C=O, ketone), 2643.39 (C–H, alkane), 2918.76 (C–H, alkane), 3023.26 (C–H, aromatic). HRMS (ESI) m/z: calculated for C23H26O4 [M + 1] = 367.1865, found 367.1899.
5-hydroxy-7-(nonyloxy)-3-phenyl-4H-chromen-4-one (C6)
Yield: 47%; m.p. 73–76 °C; clogP: 5.88. 1H NMR (CDCl3, δ ppm) 0.92 (3H, t, J = 7.0 Hz, 10"–H3), 1.32 (10H, m, 5"–H2, 6"–H2, 7"–H2, 8"–H2, 9"–H2), 1.48 (2H, m, 4"–H2), 1.85 (2H, quint, J = 6.4 Hz, 3"–H2), 4.03 (2H, t, J = 7.6 Hz, 2"–H2), 6.37 (1H, s, 3–H), 6.50 (1H, s, 8–H), 6.68 (1H, s, 3–H), 7.28 (CDCl3) 7.55 (3H, m, 3'–H, 4'–H, 5'–H), 7.90 (2H, d, J = 13 Hz, 2'–H, 6'–H), 12.71 (1H, s, 5-OH). 13C NMR (CDCl3, δ ppm) 14.1 (10"), 25.7 (9"), 25.9 (8"), 28.9 (7"), 29.3 (6"), 29.3 (4"/5"), 29.5 (3"), 31.9 (2"), 68.7 (1"), 76.7–77.3 (CDCl3), 93.1 (8), 98.6 (6), 105.6 (4a), 105.9 (3), 126.3 (3'/5'), 129.1 (4'), 131.4 (2'/6'), 131.8 (1'), 157.8 (8a), 162.1 (5), 163.9 (2), 165.2 (7), 182.5 (4). IR (cm−1): 1169.51 (C–O–C, large ring C–O stretch), 1272.96 (C–O, phenolic), 1505.20 (C=C aromatic), 1660.79 (C=O, ketone), 2323.48 (C–H, alkane), 2643.39 (C–H, alkane), 2918.76 (CH, alkane), 3018.56 (C–H, aromatic). HRMS (ESI) m/z: calculated for C24H28O4 [M + 1] = 381.2021, found 381.2057.
7-(decyloxy)-5-hydroxy-3-phenyl-4H-chromen-4-one (C7)
Yield: 56%; m.p. 74–76 °C; clogP: 6.30. 1H NMR (CDCl3, δ ppm) 0.89 (3H, t, J = 4.9 Hz, 11"–H3), 1.32 (12H, m, 5"–H2 6"–H2, 7"–H2, 8"–H2, 9"–H2, 10"–H2), 1.48 (2H, m, 4"–H2), 1.85 (2H, quint, J = 7.4 Hz, 3"–H2), 4.06 (2H, t, J = 7.4 Hz, 2"–H2), 6.38 (1H, s, 6–H), 6.52 (1H, s, 8–H), 6.78 (1H, s, 3–H), 7.52 (3H, m, 3'–H, 4'–H, 5'–H), 7.90 (2H, d, J = 7.8 Hz, 2'–H, 6'–H), 12.75 (1H, s, 5-OH). 13C NMR (CDCl3, δ ppm) 14.1 (11"), 22.7 (10"), 25.9 (9"), 28.9 (8"), 29.3 (7"/6"), 29. 6 (4"/5"), 31.9 (3"), 68.7 (2"), 76.7–77.4 (CDCl3), 93.1 (8), 98.6 (6), 105.6 (4a), 105.9 (3), 126.3 (3'/5'), 129.1 (4'), 131.4 (2'/6'), 131.8 (1'), 157.8 (8a), 162.1 (5), 163.9 (2), 165.2 (7), 182.5 (4). IR (cm−1): 1169.51 (C–O–C, large ring C–O stretch), 1283.27 (C–O, phenolic), 1512.15 (C=C aromatic), 1663.49 (C=O, ketone), 2286.36 (C–H, alkane), 2323.33 (C–H, alkane), 2645.76 (C–H, alkane), 3014.51 (C–H, aromatic). HRMS (ESI) m/z: calculated for C25H30O4 [M + 1] = 395.2178, found 395.2208.
7-((4-bromopentyl)oxy)-5-hydroxy-3-phenyl-4H-chromen-4-one or 7-O-2-bromopentylchrysin (C8)
Yield: 94%; m.p. 123–124 °C; clogP: 4.26. 1H NMR (CDCl3, δ ppm) 1.42 (trace cyclohexane), 1.62 (trace water),1.78 (3H, d, J = 9.6 Hz, 6"–H3), 2.08 (4H, m, 3"–H2, 4"–H2), 4.08 (2H, t, J = 4.8 Hz, 2"–H2), 4.22 (1H, s, 5"–H), 6.38 (1H, s, 6–H), 6.53 (1H, s, 8–H), 6.69 (1H, s, 3–H), 7.28 (CDCl3), 7.55 (3H, m, 3'–H, 4'–H, 5'–H), 7.91 (2H, d, J = 8.1 Hz, 2'–H, 6'–H), 12.80 (1H, s, 5-OH). 13C NMR (CDCl3, δ ppm) 26.6 (6"), 27.4 (3"), 37.3 (4"), 50.7 (5"), 67.7 (2"), 76.8–79.4 (CDCl3), 93.9 (8), 98.6 (6), 105.7 (4a), 108.9 (3), 126.3 (3'/5'), 130.1 (4'), 131.1 (2'/6'), 131.8 (1'), 157.6 (8a), 162.2 (5), 163.8 (2), 164.9 (7), 182.5 (4). IR (cm−1): 765.53 (C–Br), 1172.08 cm−1 (C–O–C, large ring C–O stretch), 1101.61 and 1270.95 cm−1 (C–O, phenolic), 1603.28 cm−1 (C=C aromatic), 1621.23 cm−1 (C=O, ketone), 2854.31 cm−1 (C–H, alkane), 2943.23 cm−1 (C–H, alkane), 2965.54 cm−1 (C–H, alkane), 3068.12 cm−1 (C–H, aromatic). HRMS (ESI) m/z: calculated for C20H19BrO4 [M + 1] = 405.0534, found 405.0521.
Cell culture and growth inhibition studies
DMEM supplemented with 10% heat inactivated FBS,
Bacterial and fungal growth and growth inhibition studies
Nutrient agar (agar, 15 g L−1, meat extract, 1 g L−1, peptone, 5 g L−1, sodium chloride, 5 g L−1, yeast extract, 2 g L−1) was used to culture Bacillus cereus ATCC 10876, S. aureus ATCC 25923, Enterococcus faecalis NCIMB 13280, P. aeruginosa NCTC 13437, P. fluorescens ATCC 13525, Extended Spectrum Beta-Lactamase (ESBL) producing E. Coli NCTC 13353, MRSA 252 and K. pneumoniae ATCC 13439, and Sabouraud Dextrose Agar (SDA) was used to culture C. albicans MTCC227. Strains were cultured overnight (22–25 h) at 37 °C on nutrient broth for the preparation of cell suspensions. Suspensions of bacterial cells (dissolved in PBS) were homogenised such that the spectrophotometry standard of 5 × 105 CFU mL−1 (0.5 McFarland standards) was obtained.
Minimum inhibitory concentration studies
MIC is considered the concentration of sample that prevents a colour change of the media and inhibited bacterial growth completely. All samples were dissolved in Mueller Hinton Broth (MHB)/DMSO with a DMSO final concentration of 2.5%. The resulting solution was added to MHB. Inoculum prepared in appropriate broth – MHB (100 µL) was then added. These plates were sealed and agitated using a plate shaker incubated for 18 h at 37 °C. A negative control (wells containing broth, DMSO and 100 µL of inoculum) was also prepared. Gentamicin was used as the reference antibiotic for the bacterial strains while fluconazole was the antifungal agents tested on C. albicans. The p-iodonitrotetrazolium chloride (INT) colorimetric assay was used to determine MIC. The assay measures the production of NADH. In order to determine MIC, 40 μL of 0.2 mg mL−1 of INT were added after incubation for 18 h at 37 °C and samples were screened for colour change from violet to red.
cLogP calculation
Calculated logP values were predicted using ChemDraw 20.1.
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211057467 – Supplemental material for Facile synthesis and biological evaluation of chrysin derivatives
Supplemental material, sj-pdf-1-chl-10.1177_17475198211057467 for Facile synthesis and biological evaluation of chrysin derivatives by Nicholas Omonga, Zakia Zia, Hesham Ghanbour, Abby Ragazzon-Smith, Howard Foster, John Hadfield and Patricia Ragazzon in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the technical staff at the University of Salford and Salford Analytical Services, in particular Mr Kirit Amin for his outstanding analytical contributions. The authors also express their gratitude to Dr Nanda Puspita for her outstanding help with cell culture.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Nigerian Government and the University of Salford.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
