Abstract
We propose a new method for the selective detection of the antibiotic metronidazole (MNZ) using CB[7]-JAT (cucurbit[7]uril = CB[7] and JAT = jatrorrhizine) as a fluorescent probe, which is based on the competitive reaction between MNZ and JAT for the occupancy of the CB[7] cavity. The proposed method gives a good calibration curve in the concentration range of 0.38–60 μM, and the limit of detection for MNZ is 65 ng mL−1 with those obtained by the standard curve method. Moreover, the proposed method was successfully applied for the determination of MNZ in liquid milk. Most importantly, due to the high binding affinity between CB[7] and MNZ, the proposed method shows great anti-interference capacity to accurately detect MNZ in the presence of other antibiotics.
A rapid and sensitive fluorometric method was first established for the determination of metronidazole (MNZ) using CB[7]-JAT (cucurbit[7]uril = CB[7] and JAT = jatrorrhizine) as a fluorescent probe and shows great anti-interference capacity.

Introduction
Metronidazole (MNZ) (Figure 1) belongs to nitroimidazole antibiotic group and has been extensively used for the treatment of various bacterial infections in humans and animals.1–5 However, the long-term use of this drug may do serious harm to the body, such as sputum, peripheral neuropathy, and ataxia.6,7 Its metabolites are also potentially mutagenic, toxic and carcinogenic, while the continuous release of MNZ has caused high levels of residues in environmental and biological systems. So far, numerous analytical techniques—including surface-enhanced Raman scattering (SERS), 8 gas chromatography (GC),9,10 pH titrations, 11 and high-performance liquid chromatography (HPLC)12,13—have been used for the determination of MNZ. However, these techniques require either expensive equipment and professional technical personnel, or complicated sample processing. Furthermore, some other methods are not sensitive enough. Spectrofluorimetry is considered as one of the most convenient analytical techniques in pharmaceutical analysis. This is due to the low cost of analysis, and its inherent simplicity, fast signal response, ready availability, and high sensitivity in most quality-controlled and clinical laboratories.

Structure of CB[7], JAT, and MNZ.
Cucurbit[n]urils (CB[n]s, n = 5–8, 10) are a family of pumpkin-shaped macrocyclic molecules composed of n glycoluril units connected with 2n methylene groups.14–16 As highly symmetrical host molecules, CB[n]s are equipped with a rigid hydrophobic cavity and two identical polar portals possessing ureido carbonyl groups, making them excellent binders of cationic organic guests.17–23 Among different CB[n]s, CB[7] (Figure 1) is the most widely used host molecule due to its good water solubility and its ability to the formation of the stable inclusion complexes.24–26 Recently, CB[7]-based host–guest complexes have been utilized as optical sensors in drug detection.27–31 Nevertheless, there are a few reports on the selective detection of drugs based on CB[7] host–guest complexes in the presence of other antibiotics.
Jatrorrhizine (JAT) (Figure 1) is a type of isoquinoline alkaloid, in this work, we employ CB[7]-JAT inclusion complex as a fluorescent probe to detect non-fluorescent MNZ based on the competitive reaction between MNZ and JAT for the occupancy of the CB[7] cavity. To our knowledge, the use of JAT as a fluorescent probe for the determination of MNZ has not been reported. The proposed method was also tested for the determination of MNZ in liquid milk samples, which indicated promising applications in various practical scenarios.
Results and discussion
Spectral characteristics
Solution of JAT and MNZ showed undetectable fluorescent emission in aqueous solutions. Upon the addition of CB[7] to JAT, a dramatic increase in the fluorescence intensity was observed, indicating that the formation of a fluorescent inclusion complex between CB[7] and JAT (Figure 2(a)). Interestingly, when gradually adding MNZ to the CB[7]JAT solution, the fluorescence intensity of CB[7]-JAT rapidly decreased (Figure 2(b)), which is presumably due to the competitive reaction between JAT and MNZ for the occupancy of the CB[7] cavity. Hence, these results indicate that CB[7]-JAT can serve as a promising fluorescence probe for sensing MNZ.
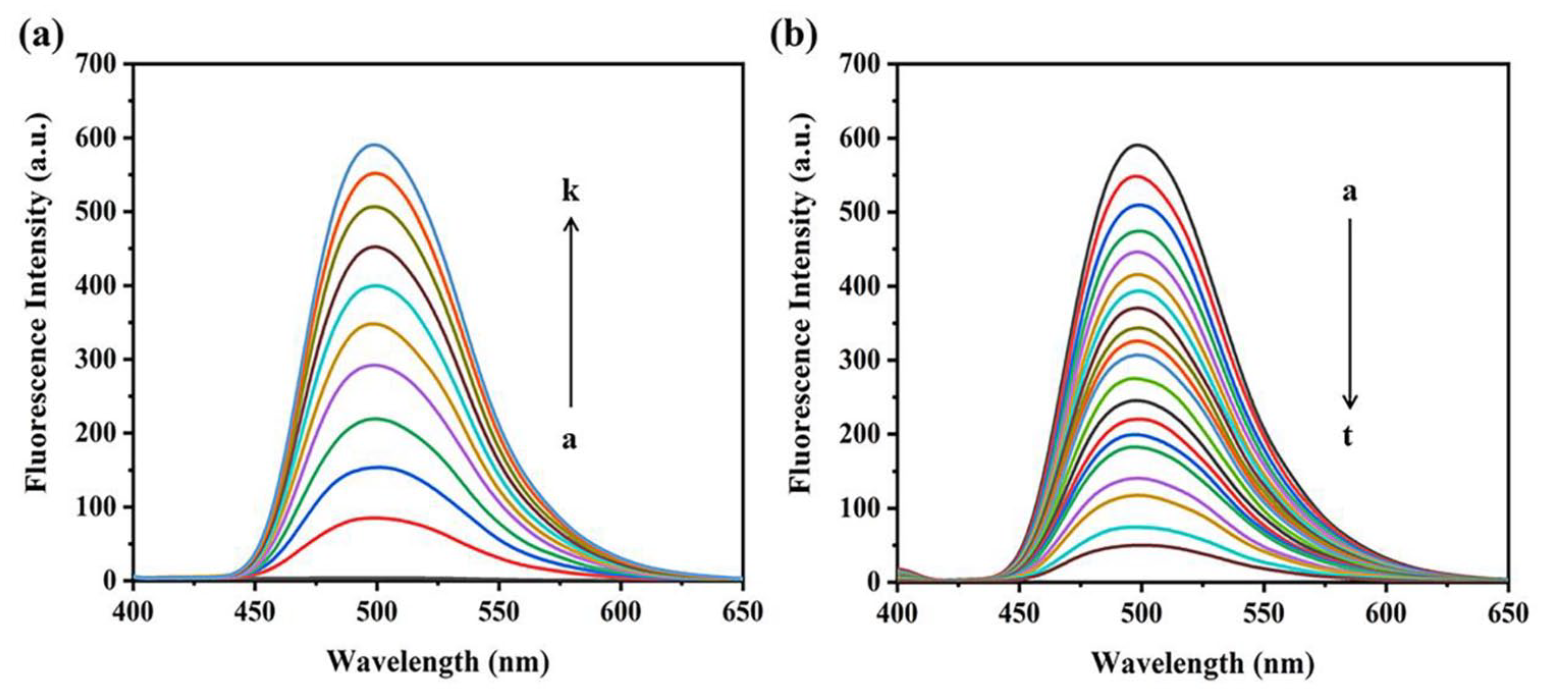
(a) The fluorescence spectra of JAT in the absence and presence of increasing concentrations of CB[7] (a = 0, b = 1, c = 2, d = 3, e = 4, f = 5, g = 6, h = 7, i = 8, j = 9, and k = 10 μM). (b) The fluorescence spectra of JAT–CB[7] complexes upon addition of MNZ (a = 0, b = 10, c = 20, d = 30, e = 40, f = 50, g = 60, h = 70, i = 80, j = 90, k = 100, l = 120, m = 140, n = 160, o = 180, p = 200, q = 250, r = 300, s = 400, and t = 500 μM), (λex = 340 nm).
Characterization of CB[7]-JAT inclusion complex
JAT exhibits weak fluorescence in aqueous solution because the isoquinoline ring and the substituted benzene ring are not in the same plane. Thus, the JAT molecule cannot form a conjugated system. When CB[7] was added to an aqueous solution of JAT, the apolar isoquinoline ring part of JAT penetrate into the hydrophobic cavity of CB[7], causing an electrostatic attraction between the high electron density of the carbonyl oxygens of CB[7] and the positive charge of the heterocyclic nitrogen of JAT. Hence, the degree of freedom of motion of the JAT molecule is reduced, and further reduced the probability of radiationless transitions, which leads to fluorescence enhancement.
The formation of a CB[7]-JAT inclusion complex has been confirmed by 1H NMR spectroscopy. As shown in Figure 3, upon addition of CB[7] (1 equiv.) to the JAT solution, the resonances corresponding to JAT showed both upfield and downfield shifts compared with those of free JAT. The upfield shifts for Hb, Hc, Hd, Hk, and HƖ indicate that the protons were present deep inside the cavity of CB[7]. In contrast, the proton resonance of Ha, He, Hi, Hg, and Hh displayed down field shifts, and sit outside the carbonyl portals of CB[7]. It is worth noting that after 1:1 stoichiometry was reached, no more changes were observed in the NMR spectra (1–2 equiv.). These results suggest that a 1:1 CB[7]-JAT inclusion complex is formed.

1H NMR spectra of JAT (18.1 mM) and JAT-CB[7] (at a molar ratio of 1:1 and 1:2) in D2O.
The CB[7]-JAT inclusion complex was also investigated by powder X-ray diffraction (PXRD) and FTIR spectrum. The X-ray diffraction (XRD) patterns of pure CB[7], pure JAT, a CB[7]-JAT physical mixture, and CB[7]-JAT inclusion complex samples are shown in Figure 4(a); it revealed that crystallization peaks of JAT can still be detected in physical mixtures of both CB[7] and JAT. However, these peaks disappeared completely in the CB[7]-JAT inclusion complex, which indicates the formation of an inclusion complex between CB[7] and JAT. 32 The FTIR spectrum is depicted in Figure 4(b), free JAT bands are present at v = 1611 and 1453 cm−1, which are due to C=C vibrations of the aromatic ring, and at 1728 cm−1 due to the C=O of CB[7]. Their physical mixture has no characteristic absorptions compared to free JAT and CB[7]. In contrast, in the IR spectrum of CB[7]-JAT, a new absorption bands centered at 1457 cm−1 appeared. In particular, the peak at 1728 cm−1 corresponding to the C=O groups on the rims of pure CB[7] was shifted to 1721 cm−1 on account of the interaction between CB[7] and JAT, further confirming the formation of an inclusion complex.
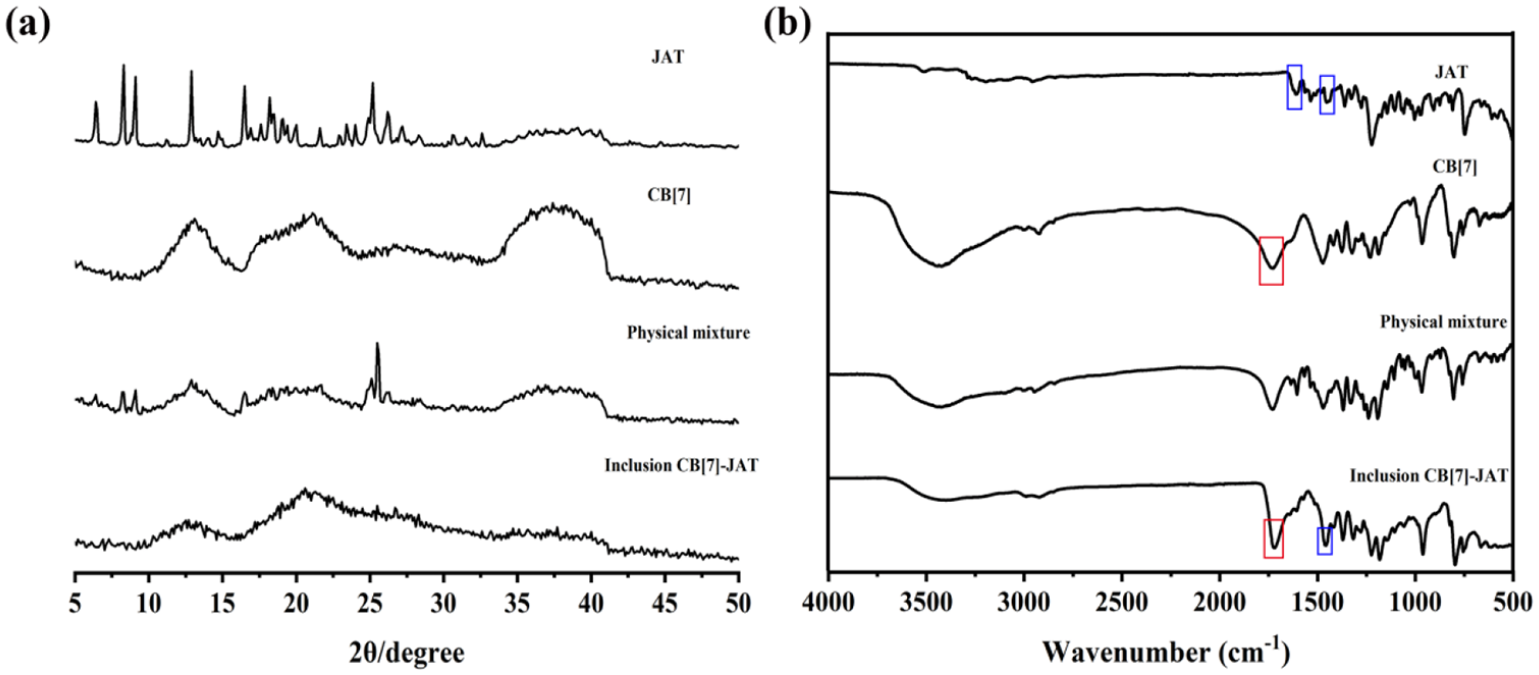
(a) XRD patterns for JAT, CB[7], a physical mixture of CB[7]-JAT, and the CB[7]-JAT inclusion complex. (b) A comparison of the FTIR spectra of JAT, CB[7], a physical mixture of JAT and CB[7], and the CB[7]-JAT inclusion complex.
Optimization of the experimental conditions
The effect of the pH in the range from 1.0 to 11.0 was investigated. The fluorescence quenching value (ΔF) changed slightly in the pH range of 1.0–11.0, and showed a maximum change at pH 7.0. Hence, a neutral solution was optimum in this experiment. In addition, ΔF reached a maximum of 20 min after the reagents were added and it remained constant for at least 12 h. The effect of the temperature on ΔF was investigated in the temperature range of 10–80 °C. The formed complexes were stable below 35 °C. However, at temperatures above 35 °C, the fluorescence intensity greatly decreased, which can be attributed to the dissociation of the complexes at high temperatures. For these reasons, the standard reaction conditions were set as pH 7.0 for 20 min at room temperature.
The selectivity of sensing MNZ
To check the selectivity of CB[7]-JAT toward MNZ, eight commonly used antibiotics including AMPB (amphotericin B), AZM (clotrimazole), NEO (neomycin sulfate), SDM (sulfamethazine), PNC-G (benzylpenicillin-G-sodium), ERY (erythromycin), CTM (azithromycin) and KMC (kanamycin) were selected, the chemical structures of which are shown in Figure S1. As shown in Figure 5, when MNZ was added to the CB[7]-JAT probe solution, the fluorescence was nearly completely quenched. Nevertheless, there were almost no changes in the cases of the other antibiotics, implying that CB[7]-JAT can be regarded as a potential candidate for selective sensing of MNZ. Moreover, the anti-interference experiment results showed that the quenching selectivity for MNZ is not disturbed by the addition of other antibiotics (Figure 6), further confirming that CB[7]-JAT can act as an excellent fluorescence probe for MNZ detection.

(a) Fluorescence intensity of the CB[7]-JAT probe and the fluorescence quenching intensity after the addition of antibiotics (λex = 340 nm). (b) Fluorescence quenching intensities of the CB[7]-JAT (10 μM) probe after the addition of other antibiotics (500 μM).
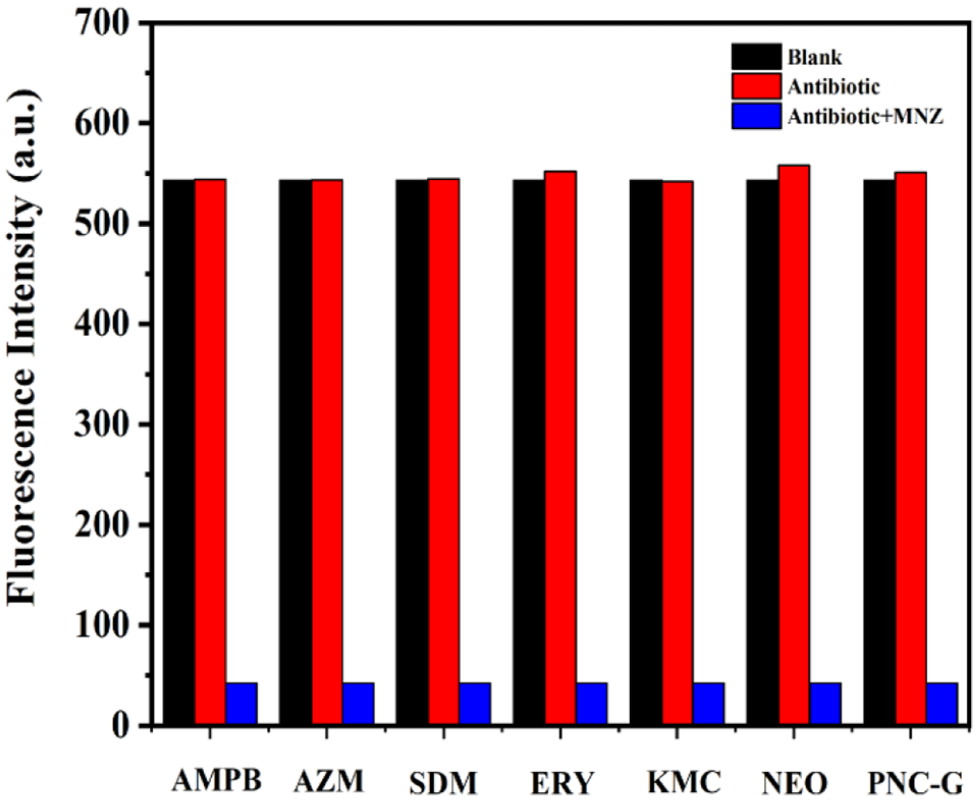
Fluorescence intensities of CB[7]-JAT (10 μM) aqueous solutions in the presence of individual antibiotic (500 μM) and a mixture of competing antibiotic including MNZ (500 μM).
The sensitivity of MNZ sensing
To further investigate about the sensitivity of the CB[7]-JAT probe for MNZ detection, quantitative fluorescence titrations experiments were carried out. As shown in Figure 2(b), with an increasing concentration of MNZ, the fluorescence intensities of CB[7]-JAT decreased gradually, and the ΔF values exhibited a good linear relationship with MNZ in the range of 0.38–60 μM. The linear regression equations are ∆F = 4.363C (μM) + 72.434 with correlation coefficients of 0.9968 (Figure 7), and the limit of detection (LOD) is 65 ng mL−1 (LOD = 3σ/S), where σ and S are the relative standard deviation (RSD) of the response and the slope of the calibration curve. The method reported herein is comparable and even more sensitive than any other methods reported in the literature for sensing of MNZ (Table 1).2,33–35

The standard calibration curve ΔF versus MNZ (0–60 μM), using 1.0 CB[7]-JAT (10 μM) as a fluorescence probe.
Comparison with other methods for the determination of MNZ.

MNZ: metronidazole.
The quenching mechanism of the fluorescent probe
The reason for the fluorescence quenching was attributed to competitive replacement of JAT from the CB[7] cavity by MNZ. As shown in Figure 8, after the addition of MNZ to the solution CB[7]-JAT, the signals of the JAT protons Ha, Hc, Hd, Hb, Hh, Hi, Hk, and HƖ were consistent with free JAT, and protons Hm, Hn displayed down field shifts, supporting the fact that JAT was displaced from CB[7] by MNZ. Meanwhile, the signals due to protons Ha and Hb of MNZ showed significant upfield shifts, while Hd was shifted downfield compared with free MNZ, thus being consistent with CB[7]-MNZ (Figure S2). These findings indicated that the MNZ molecules were present in the CB[7] cavity, and eventually lead to the fluorescence quenching of CB[7]-JAT. The association constant was calculated using a non-linear curve-fitting method (Figure 9), and the value was found to be 4.8 × 104 M−1, which is much lower than the association constant of CB[7]-MNZ 2.7 × 108 M−1, obtained via the same method. Hence, JAT molecules can be easy expelled from the CB[7] cavity by MNZ molecules. Besides, as shown in Figure 10, we can see that MNZ can be identified in 20 s and almost reached a stable level, indicating that the CB[7]-JAT probe can detect MNZ with a rapid response. These results suggest that CB[7]-JAT might be a potential fluorescence probe for detecting MNZ.

1H NMR spectra of JAT (18.1 mM), 1.0 CB[7]-JAT, JAT-1.0 CB[7]-1.0 MNZ, and MNZ (18.1 mM) in D2O.
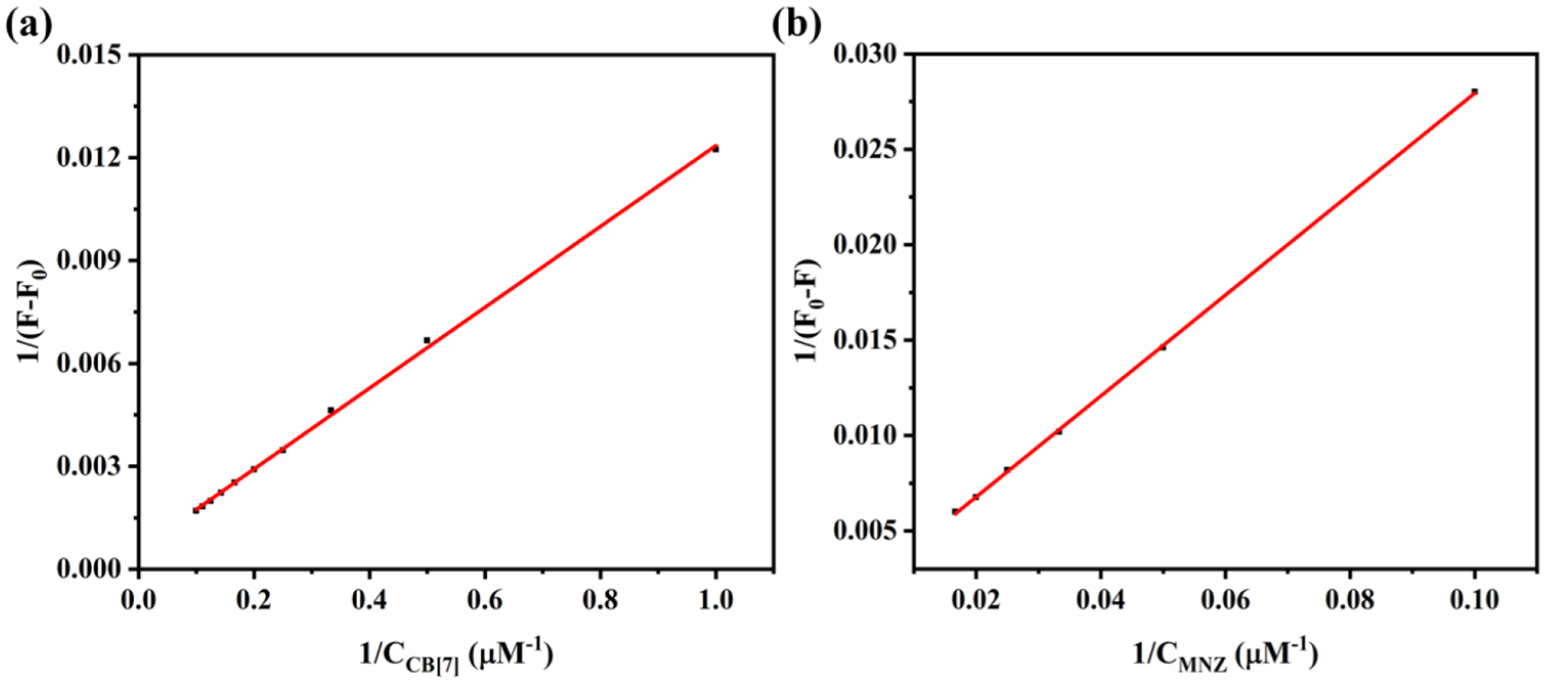
(a) A plot of 1/(F − F0) versus 1/CB[7] for CB[7]-JAT complex (CJAT = 10 μM). (b) A plot of 1/(F0 − F) versus 1/MNZ for CB[7]-MNZ complex (CMNZ = 10 μM).

Fluorescence response versus time curve of MNZ (20, 40, and 60 μM) solutions added to the CB[7]-JAT (10 μM) fluorescence probe system.
Analytical application
The proposed fluorescent probe was further applied in the determination of MNZ in milk. To detect MNZ in milk samples, the standard addition method was used. As summarized in Table 2, the recovery ratio ranged from 99.1% to 105.0%, and the low RSD (n = 3) was less than 1.4%. These results revealed that the proposed method possess good precision and satisfactory reproducibility.
Determination of MNZ in milk (n = 3).

MNZ: metronidazole; SD: standard deviation.
Conclusion
In summary, a rapid and sensitive fluorescent method has been established for the determination of MNZ using a CB[7]-JAT inclusion complex as a fluorescent probe based on a host–guest competitive reaction. The high association constant of MNZ with CB[7] of up to 2.7 × 108 M−1 suggests the strong anti-interference capacity of the CB[7]-MNZ system in the presence of other antibiotics with a low LOD value of 65 ng mL−1. Moreover, the proposed probe was also successfully applied for the detection of MNZ in milk, with recoveries being in the range 99.1%–105.0%. This study is beneficial for the molecular recognition between CB[7] and drugs which have non-fluorescent or weakly fluorescent substances.
Experimental section
Chemicals and samples preparation
JAT and MNZ were obtained from the Chinese National Institute for the Control of Pharmaceutical. CB[7] was prepared and characterized according to the literature. 36 All other reagents were of analytical reagent grade. Stock solutions of JAT, MNZ, and CB[7] (100 μM) were dissolved in fresh double-distilled water (pH = 3). All the stock standard solutions can be stored stably for several weeks at room temperature. Standard working solutions were obtained by dilution of the stock solutions with fresh double-distilled water before use.
Apparatus characterization
The fluorescence spectroscopy was measured using an Edinburgh FLS55 spectrophotometer. The slit widths of the excitation and emission monochromators were set to 5 nm. All measurements were performed using a standard 1 cm path-length quartz cell at room temperature. The 1H NMR spectra were recorded using a Bruker AV-400 MHz NMR spectrometer in D2O. Infrared spectra were obtained with a Bruker IFS 66v/S FTIR spectrometer. PXRD measurements were obtained on a Rigaku/Dmax 2200 PC X-ray diffractometer using Cu–Kα radiation in the angular range of 2θ from 5° to 50°.
Spectra measurement procedure
A solution of 100 μM CB[7] 1 mL was poured into a 10-mL colorimetric flask, and then appropriate amounts of JAT solution were then sequentially added to the flask. The mixture was diluted to volume with double-distilled water and shaken for 30 min at room temperature. The fluorescence spectra and intensity measurements were recorded at an excitation wavelength of 340 nm.
Pretreatment of milk samples
Liquid milk was bought from a local supermarket (Mengniu milk, 5C20200905DE0201:34). The milk samples were pretreated according to the previous method. 37 First, 10 mL, 61 mM of trichloroacetic acid was added to 10 g of milk and the mixture was then sonicated for 15 min to remove the proteins. Next, the samples were centrifuged at 10,000 r/min for 10 min, and the upper clear liquid was filtered. Finally, the filtrate was concentrated on a rotary evaporator to 10 mL and filtered through a 0.45-μm microporous membrane to afford the sample. Known amounts of MNZ were added to the obtained milk samples to acquire different concentrations of MNZ solutions for analysis.
Supplemental Material
sj-docx-1-chl-10.1177_17475198211055103 – Supplemental material for A cucurbit[7]uril-based supramolecular fluorescent probe for the detection of metronidazole with high sensitivity and strong anti-interference capacity
Supplemental material, sj-docx-1-chl-10.1177_17475198211055103 for A cucurbit[7]uril-based supramolecular fluorescent probe for the detection of metronidazole with high sensitivity and strong anti-interference capacity by Xuemei Hu, Huaqing Zhang and Mei Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Research Project for the 13th Five-Year Plan of the Education Department of Jilin Province of China (no. JJKH20200649KJ).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
