Abstract
Three coordination polymers—{[Ag2(SIP)(4-pp)]·[Ag(4-pp)2]·H2O}n, [Pb4(SIP)2(μ3-OH)2(H2O)2]n, and {[Zn2(SIP)2(4-pp)4][Zn(4-pp)2(H2O)4]·4H2O}n (SIP = 5-sulfoisophthalic acid anion, 4-pp = 4-phenylpyridine)—are constructed under hydrothermal or solvothermal conditions using sodium m-phthalate 5-sulfonate (NaH2SIP) and 4-phenylpyridine as ligands. The structures of the three complexes are determined by single-crystal X-ray diffraction, powder X-ray diffraction, thermogravimetric analyses, Fourier-transform infrared analysis, and photoluminescence. The fluorescent properties of the complexes in the solid state are investigated at room temperature, and the results indicate that they all show photoluminescent properties.
Keywords

Introduction
The design and construction of coordination polymers (CPs) have been a very active area of materials research in recent years. The intense interest in these materials is driven by their potential applications as functional materials for luminescence sensing, gas storage and separation, drug delivery, catalysis, magnetism, and due to their intriguing structural diversity.1–6 It is well-known that many factors such as synthetic methods, central metals, ligands, and pH can influence the final structures of CPs.7–10 There is no doubt that organic ligands and the metal center play a significant role in the design and synthesis of CPs, while selecting the coordination geometry of the metal centers and organic ligands containing appropriate functional groups (such as polycarboxylic acid or multipyridine ligands) are also key for constructing CPs. Due to the high coordination numbers and flexible coordination geometries of transition-metal ions, they are able to produce a variety of structures. Moreover, the properties of CPs are highly dependent on the structures of the free ligands and the nature of the metal ions. The assembly of these metal–organic CPs allows for a wide choice of different parameters, including diverse electronic properties. A wide variety of CPs with interesting structures and desirable properties have been prepared through judicious choice of organic linkers and metal ions.
Recently, many CPs have been synthesized by employing polycarboxylate aromatic ligands because of their versatile coordination modes and thermal stability.11–15 Sodium m-phthalate 5-sulfonate (NaH2SIP) has inspired significant research efforts toward assembling coordination architectures. The approach to the supramolecular frameworks employed in this work involves the use the trifunctional linker NaH2SIP for the following reasons: it contains two carboxylate groups and one sulfonate group and is one of the most useful multifunctional ligands due to (1) structural rigidity, (2) strong bonding interactions, and (3) a rich diversity of coordination modes that can be utilized for extension of the metal ions into high-dimensional structures.
16
The sulfonate group is generally perceived as a weaker group with respect to its coordinating ability, and it has one more potentially coordinating oxygen atom. The weak coordination nature of sulfonate makes its coordination mode very flexible. In addition, the sulfonate group is a suitable hydrogen-bond acceptor. It can also be used as short bridge and long bridge between metal centers to construct a variety structures. It can be partially or completely deprotonated, and normally serves as a trigonal-shaped connector to construct diverse metal-supramolecular systems via different coordination and hydrogen-bonding modes.17–22 π–π stacking interactions between the aromatic rings may facilitate ordered, non-interpenetrated open frameworks. In this work, three complexes based on 5-sulfoisophthalic acid and 4-phenylpyridine—namely, {[Ag2(SIP)(4-pp)]·[Ag(4-pp)2]·H2O}n (
Results and discussion
Description of structure 1
Single-crystal X-ray structural analysis reveals that the asymmetric unit of complex

(a) Coordination environments for the Ag ions in
Selected bond lengths (Å) and angles (°) for complex

Symmetry codes: A: 4 − X, 1 − Y, −Z; B: 1 + X, +Y, +Z; C: 3 − X, 1 − Y, −Z.
Description of structure 2
Single-crystal X-ray diffraction analysis reveals that complex

(a) Coordination environments of the Pb ions in
Selected bond lengths (Å) and angles (°) for complex
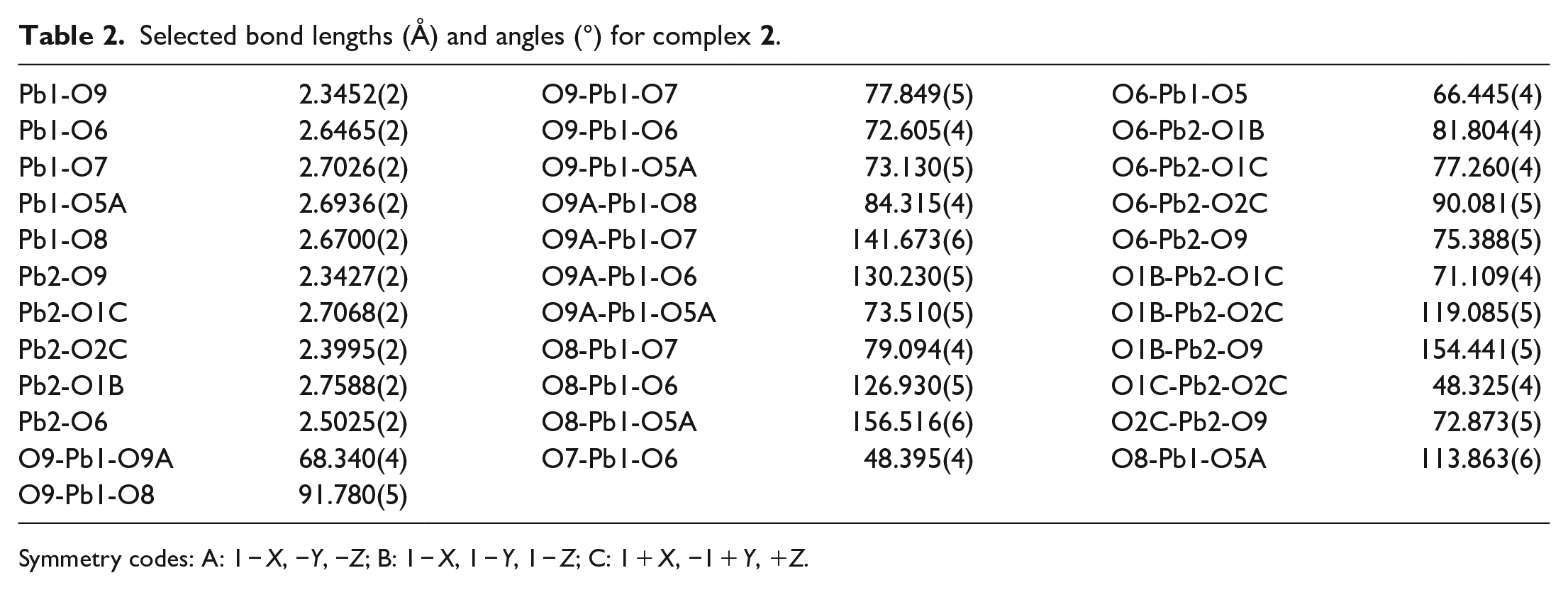
Symmetry codes: A: 1 − X, −Y, −Z; B: 1 − X, 1 − Y, 1 − Z; C: 1 + X, −1 + Y, +Z.
Description of structure 3
X-ray diffraction analyses revealed that

(a) Coordination environment for the Zn(II) ions in
Selected bond lengths (Å) and angles (°) for complex
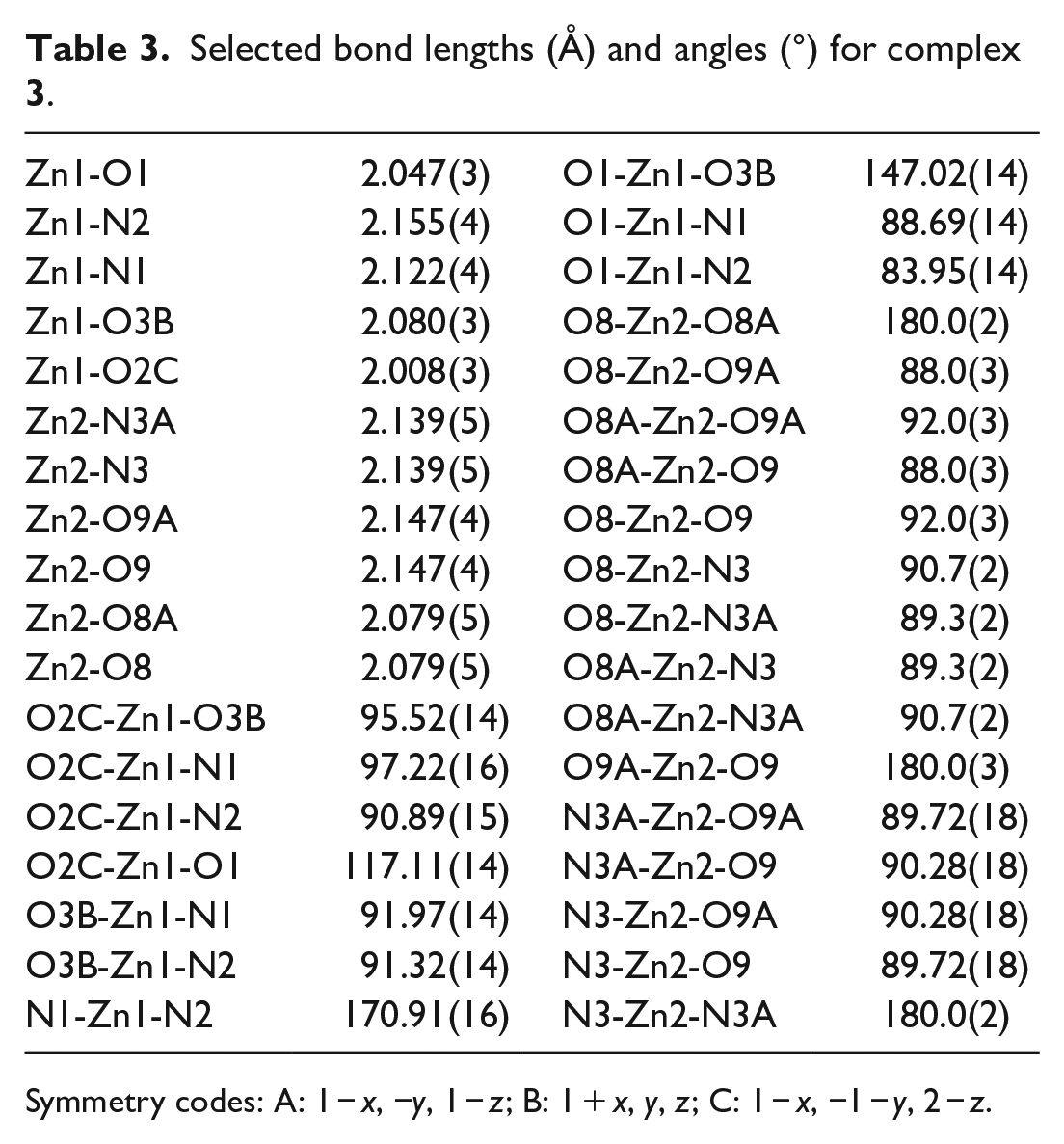
Symmetry codes: A: 1 − x, −y, 1 − z; B: 1 + x, y, z; C: 1 − x, −1 − y, 2 − z.
The hydrogen-bond lengths (Å) and angles (°) of 3.

Symmetry codes: A: 1 − x, −y, 1 − z; B: 1 + x, y, z; C: 1 − x, −y, 1 − z.
IR spectra
IR spectroscopy is an excellent approach to characterize and study crystallization, and hence, the IR spectra of complexes

IR spectra of complexes
Powder X-ray diffraction and thermal analyses
The Powder X-ray diffraction (PXRD) analyses of the synthesized crystalline samples of

PXRD patterns for the samples
TGA were carried out for complexes

TGA curves of complexes
Luminescent properties
It is well-known that CPs constructed with a d
10
metal center and conjugated organic linkers are promising candidates for photoactive materials,23–27 with potential applications such as chemical sensors and electro-luminescent materials. Therefore, the photoluminescence (PL) properties of the three complexes, together with the free ligands, are investigated at room temperature for comparison (Figure 7). The free NaH2SIP ligand exhibits a broad weak luminescent emission band with one emission peak at 310 nm (λex = 262.4 nm). The 4-pp ligand exhibits a broad weak luminescent emission band with one emission peak at 320 nm (λex = 264.4 nm) that can be ascribed to the π*–n or π*–π transitions, as previously reported.28,29 Complexes

Solid-state fluorescent spectra of the ligands and complexes at room temperature.
Conclusion
In summary, three new metal complexes—{[Ag2(SIP)(4-pp)]·[Ag(4-pp)2]·H2O}n
Experimental
Reagents and physical measurements
All chemicals for the syntheses were purchased from commercial sources (Aldrich, Aladdin, or Alfa Aesar) and were used as received without further purification. The structures of the complexes were confirmed by G8910A CCD single-crystal X-ray diffraction analyses. Infrared spectra were obtained using a PerkinElmer spectrum one FTIR spectrometer. PL spectra were acquired on an RF-5301 fluorescence spectrophotometer (4000–400 cm−1). TGA were performed on a TGA Q600 Thermogravimetric Analyzer and heated from room temperature to 800 °C at a rate of 10 °C min−1. The PL spectra for the solid samples were measured at room temperature.
Synthesis of complexes 1–3
{[Ag2(SIP)(4-pp)]·[Ag(4-pp)2]·H2O}n 1
A mixture of NaH2SIP (0.0268 g, 0.1 mmol), NaOH (0.0080 g, 0.2 mmol), Ag(NO)3 (0.0170 g, 0.1 mmol), and 4-phenylpyridine (0.0156 g, 0.1 mmol) in distilled water (10 mL) was stirred for 10 min at room temperature. The resulting solution was sealed in a 25-mL Teflon-lined stainless steel container, which was then heated at 120 °C for 3 days. After the sample had cooled to room temperature at 10 °C h−1, colorless rod-shaped crystals were isolated. Yield: 55% (based on Ag). FTIR: (KBr, v, cm−1): 3477(w), 1602(s), 1550(s), 1356(m), 1206(m), 1039(s), 764(s), and 617(s). Anal. calcd for C41H32Ag3N3O8S (%): C, 46.84; H, 30.46; N, 3.98; S, 3.01; found: C, 46.88; H, 30.50; N, 3.94; S, 3.05.
{[Pb4(SIP)2(μ3-OH)2(H2O)2]}n 2
Colorless lump-shaped crystals were obtained following the same method used for the preparation of complex
{[Zn2(SIP)2(4-pp)4][Zn(4-pp)2(H2O)4]·4H2O}n 3
A mixture of NaH2SIP (0.0536 g, 0.2 mmol), 4-pp (0.0310 g, 0.2 mmol), and Zn(NO)3·6H2O (0.0595 g, 0.2 mmol) was added to a mixed solvent system of ethanol (3 mL) and H2O (10 mL). The solution was kept at 120 °C for 3 days. Colorless needle-shaped crystals were obtained. Yield: 43% (based on Zn). FTIR: (KBr, v, cm−1): 3403(s), 1614(s), 1556(s), 1422(m), 1366(w), 1224(s), 1196(m), 1044(m), 766(m), 696(m), and 625(m). Anal. calcd for C82H84N6O26S2Zn3 (%): C, 53.78; H, 4.59; N, 4.59; S, 3.50; found: C, 53.79; H, 4.53; N, 4.55; S, 3.52.
Crystal structure determinations
The crystal and molecular structures of the complexes were determined by single-crystal X-ray diffraction. Diffraction intensities for the complexes were collected at 298(2) K using an Agilent G8910A CCD area-detector diffractometer with MoKα (λex = 0.71073 Å) radiation. Using Olex2,32,33 the structure was solved with the SHELXL refinement package using full-matrix least-squares refinement techniques. 34 All hydrogens were added theoretically. Crystal data as well as details of the data correction and refinement for the three complexes are summarized in Table 5.
Crystallographic and experimental data for three complexes

Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors appreciate the financial support from the Nature Science Foundation of Guangxi Province of China (no. 2017GXNSFAA198268) and the National Nature Science Foundation of China (no. 61765005).
