Abstract
Binary transition metal oxides exhibit improved properties including good redox potentials and electrical conductivities compared with single metal oxides as electrode materials in energy storage. Herein, ZIF-67 is prepared by a one-step method using Co2+ as the central metal ion, 2-methylimidazole as the organic ligand, and methanol as an organic solvent at room temperature. Hollow NiCo2O4 and sheet-like Co3O4/NiCo2O4 derived from bimetallic imidazolate framework precursors were synthesized by adding cobalt and nickel ions in appropriate proportions. A hollow and porous structure is achieved for the reaction between a nickel salt and ZIF-67, and this unique nanostructure provides a high active surface area, which is beneficial to the electrochemical properties. Several samples are prepared and used as electrode materials for electrochemical tests in 6 M KOH. As a result, the Co3O4/NiCo2O4 electrode with a sheet nanostructure showed a high specific capacitance of 846 F g−1 at a current density of 0.5 A g−1. This Co3O4/NiCo2O4 electrode material is promising for future studies on high-performance supercapacitors to solve emerging energy-related problems.

Introduction
Compared with traditional batteries, supercapacitors exhibiting excellent power density, fast charging and discharging performance, long cycle stabilities, and environmental friendliness are considered to be very promising for energy storage.1,2 Although supercapacitors can provide significantly higher power than traditional batteries of the same volume, the small energy density of supercapacitors limits their practical applications.3,4 To resolve this problem, it is necessary to research hybrid supercapacitors based on both the energy storage mechanisms of chemical batteries and supercapacitors. 5
Transition metal oxides and conductive polymers are typical electrode materials in hybrid supercapacitors. In particular, transition metal oxides are more widely used due to their high stabilities and different valence states.6,7 However, for single metal oxides, the limited electron transport between the electrode materials and the electrolyte hinders its electrochemical performance. Classical binary metal oxides such as NiCo2O4, 8 ZnCo2O4, 9 and NiFe2O410 have attracted attention because of their numerous active sites that promote the redox reactions of electrode materials, making them suitable candidates for flexible electrode materials. 11 In previous reports, NiCo2O4 complexes with various morphologies have been synthesized, such as nanosheets, nanoneedles, and yolk-shell nanostructures. Xu et al. 12 synthesized hierarchical Co3O4@NiCo2O4 core-shell nanosheets as supercapacitors, which showed excellent rate capability and compelling cycling performance. Researchers synthesized a needle-like Ni-Co precursor on Ni foam by using the hydrothermal method and then obtained Ni foam@NiCo2O4 after an annealing process. Besides, carbon nanotubes aerogels are coated on the surface of Ni foam@NiCo2O4 by utilizing van der Waals and viscous forces and exhibit a high specific capacitance at 20 A g−1. 13 To achieve electrode materials with abundant adsorption sites, the common way is to expand organic linkers resulting in metal-organic frameworks (MOFs) with ultrahigh porosity. 14 However, expanded metal links can also cause fragile frameworks and penetration between different lattices, precluding high porosity.14,15
MOFs can be used as support substrates for nanomaterials or as sacrificial templates/precursors for the generation of various nanostructures. 16 MOFs are coordinated supramolecular materials, composed of coordinated metal ions, nodes, and organic ligands. 17 Because of their large specific surface areas and abundant oxidation-active sites, they have attracted widespread attention in the field of electrochemical energy storage.18,19 MOFs, especially ZIF-67, are a type of promising porous carbon product that can be used as electrode materials in EDLCs (electrochemical double-layer capacitors). 1 ZIF-67 (a Co-based zeolitic imidazolate framework), with a cubic crystal nanostructure, can be prepared with a metal cobalt ion as the central atom and 2-methylimidazole as the organic ligand under appropriate external conditions. 20 Sundriyal et al. 21 prepared nickel- doped ZIF-67 in the presence of rGO, and finally ZIF-67/rGO generated a composition as an electrode, the specific capacitance of which was 304 F g−1 at a current density of 1 A g−1. Bimetallic metal oxide materials derived from MOFs possess high stability and conductivity, which provides more active sites and makes them ideal supercapacitor materials. 10
The bimetallic mixture derived from an MOF has a more complex structure because metal ions with different functions are concentrated in a MOF framework, and the first type of metal ion may be replaced by the second type leading to provide new functions, which depend on the new metal ion. 22 In addition, other researchers found that a polyhedron with a hollow structure could be synthesized by selenizing the precursors of Ni- Co bimetallic hydroxides that were derived from ZIF-67, with the generated material showing high specific capacitance and excellent high energy density as a supercapacitor. 23 This electrode material based on an MOF has a unique structure that can greatly shorten charge transport paths to improve the electron transport and diffusion, thus strengthening the catalytic performance of the electrode materials. 19
Inspired by these results, it is of value to develop a facile and effective method to construct materials with high conductivity based on MOFs. In this study, a simple template-assisted method has been employed with ZIF-67 as the precursor, adding cobalt and nickel ions in the appropriate proportions to synthesize products with interesting nanostructures based on coordinating etching and precipitating processes. The combination of ZIF-67 and binary metal oxides shows synergistic effects for capacitance with enhanced performance, such as higher capacitance (846 F g−1) with respect to single ZIF-67 or NiCo2O4.
Results and discussion
The procedure for fabricating ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 is presented in Figure 1, showing the formation of NiCo2O4 and Co3O4/NiCo2O4 based on ZIF-67 through the template-assisted method. Figure 2(a) shows the crystalline phases of ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 precursors according to X-ray diffraction (XRD) measurements. It shows that NiCo2O4 and Co3O4/NiCo2O4 before annealing keep the traces of ZIF-67. From Figure 2(b), the XRD pattern types are in agreement with the crystal planes of (110), (111), (220), (311), (400), (422), (511), and (440), respectively, of NiCo2O4 (JCPDS 73-1702) and Co3O4 (JCPDS 80-1535). For ZIF-67, the peaks at 7.4°, 10.4°, 12.7°, 14.7°, 16.4°, 18.0°, 22.1°, 24.5°, 25.5°, 26.6°, 29.6°, 30.5°, 31.4°, and 32.3° are unambiguously assigned to the reported patterns of crystalline ZIF-67.24–27 Furthermore, the oxidation states of Ni, Co, and O are explored in the X-ray photoelectron spectroscopy (XPS) spectrum in Figure 3(a). The XPS results in Figure 3(b) show that the oxidation states of Ni belong to Co3O4/NiCo2O4 with the signals at 853.7 and 870.9 eV conforming with Ni2+. Two other peaks at 855.3 and 872.3 eV are due to the Ni3+ species. 28 As observed in Figure 3(c), two satellite peaks located at 779.2 and 780.6 eV fit well with Co 2p3/2, while the other peaks located at 794.4 and 780.6 eV are the satellite peaks of Co 2p1/2. Similarly, the O 1s spectrum of Co3O4/NiCo2O4 is well resolved into three components at 529.3, 531.7, and 533.3 eV.
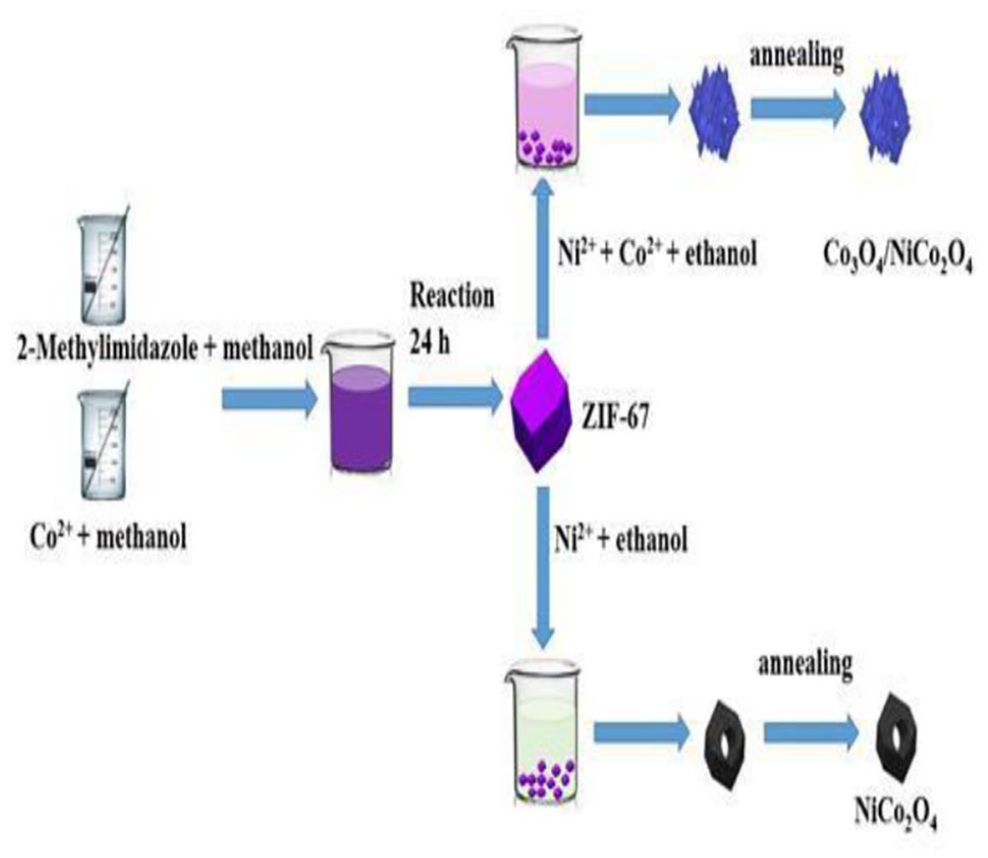
A schematic diagram of the processes involved in product formation.

XRD patterns of (a) ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 precursor and (b) NiCo2O4 and Co3O4/NiCo2O4.
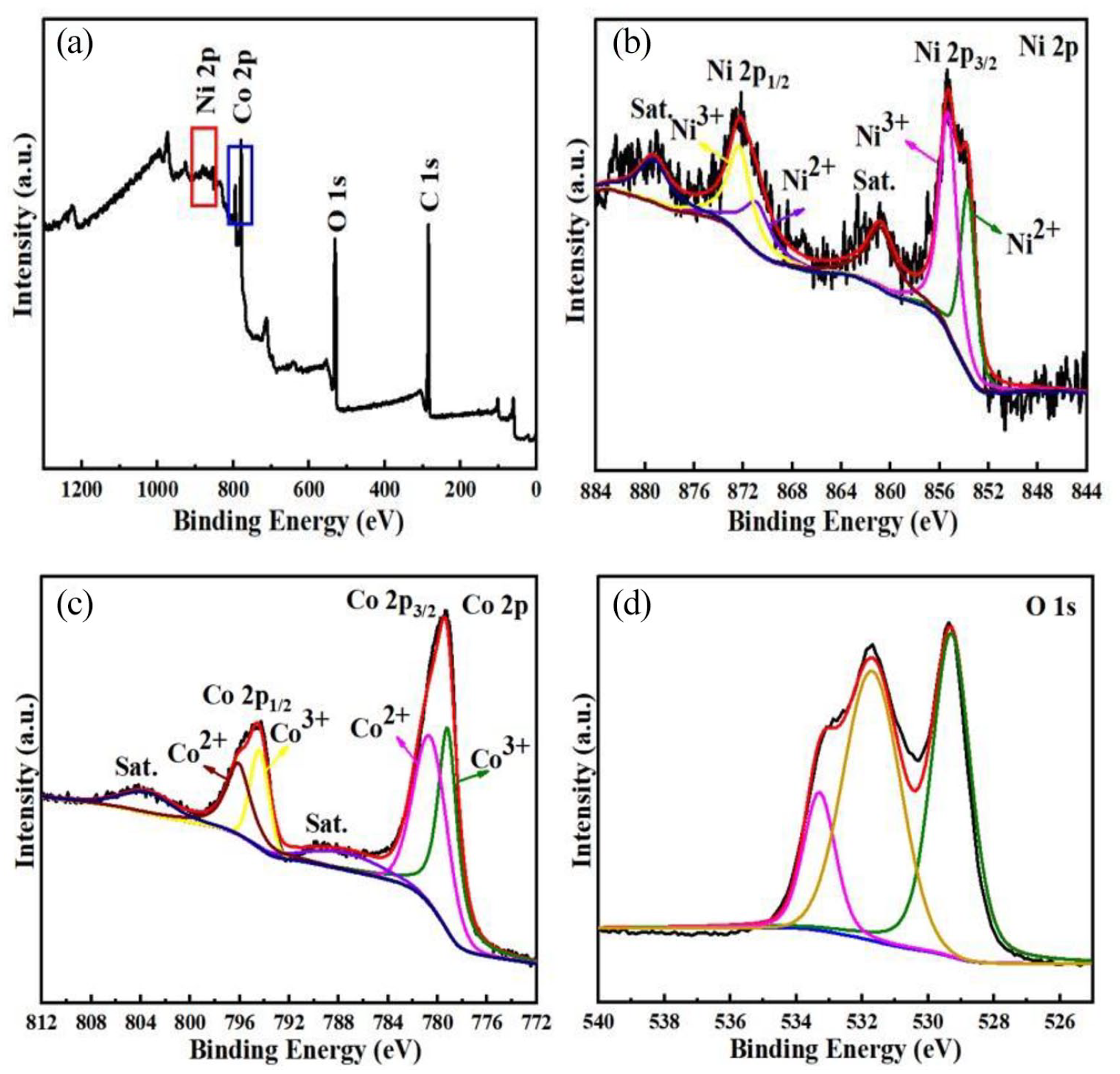
The full survey XPS spectrum of (a) Co3O4/NiCo2O4 and (b–d) XPS spectra for Ni 2p, Co 2p, and O 1s of Co3O4/NiCo2O4.
The morphologies and microstructures of ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 were analyzed by scanning electron microscopy (SEM) and transmission electron microscopy (TEM). As shown in Figure 4(a), it is observed that the microstructure of ZIF-67 exhibits a rhombic polyhedron structure with a size in the range of 500 nm. Along with the addition of Co(NO3)2 and methanol to 2-MIM, ZIF clusters can be generated by the deprotonation/coordination of imidazoles with Co2+. Next, the ZIF clusters are converted into ZIF-67 polyhedra becoming the skeleton template for NiCo2O4 and Co3O4/NiCo2O4. On addition of Ni(NO3)2 to the ZIF-67 polyhedron, a hollow structure assembled by nanosheets NiCo2O4 was prepared due to the etching reaction between released H+ (the hydrolysis of nickel ions and cobalt ions) and 2-MIM linkers of ZIF-67. 29 Besides, two metals with different diffusion rates can form inner defects and assist in forming a hollow structure due to the Kirkendall effect. 30 From Figure 4(d), it can be seen that the morphology of NiCo2O4 inherits the hollow structure after completing the one-step annealing, but it exhibits larger cavities than the precursor because of decomposition of the organic components and structural reorganization during the calcination process. 31 The SEM images in Figure 4(c) and (e) reveal that the morphology of Co3O4/NiCo2O4 was changed after adding Ni(NO3)2 and Co(NO3)2 to ZIF-67, and Co3O4/NiCo2O4 exhibits an interconnected sheet-like shape. The mechanism of formation of nanosheets may be interpreted by the following processes. The protons produced by the hydrolysis of Ni2+ ions can bind to Co-MOF resulting in liberation of Co2+ ions. Meanwhile, part of the liberated Co2+ ions are oxidized to Co3+ ions by dissolved O2 and NO3− in the solution, which coprecipitate with Ni2+ ions to generate nanosheets.29,32,33 Besides, more nickel nitrate and cobalt nitrate were added in the process of preparing Co3O4/NiCo2O4 than the preparation of NiCo2O4. The reaction system has higher hydrolysis rate because of the lower pH value, 34 resulting in the formation of Co3O4/NiCo2O4 nanosheets.

SEM images of (a) ZIF-67, (b) the NiCo2O4 precursor, (c) the Co3O4/NiCo2O4 precursor, (d) NiCo2O4, and (e) Co3O4/NiCo2O4.
To confirm the presence of Ni, Co, and O in the composites, energy dispersive spectrometer (EDS) full mapping was completed. As shown in Figure 5(b)–(d) and Table 1, O, Co, and Ni are distributed in Co3O4/NiCo2O4, and values of 50.47 wt% (O), 39.18 wt% (Co), and 10.35 wt% (Ni) were determined. As revealed by the TEM images, the obtained Co3O4/NiCo2O4 presents a nanosheet structure, which corresponds to the SEM images. The microstructure of Co3O4/NiCo2O4 was further analyzed by HRTEM (Figure 6(d)–(f)). The lattice fringes are measured to be 0.20 and 0.29 nm, corresponding to the (400) and (220) planes of NiCo2O4.35,36 Furthermore, the measured lattice fringes of Co3O4 are about 0.24 and 0.47 nm, matching with (311) and (111) crystal plane. Meanwhile, the results of TEM and HRTEM matched the data from XRD.

SEM mapping image of (a) Co3O4/NiCo2O4 and (b–d) elemental mapping of O, Co, and Ni in Co3O4/NiCo2O4.
The element distribution in Co3O4/NiCo2O4.

TEM and HRTEM images for (a–c) NiCo2O4 and (d–f) Co3O4/NiCo2O4.
Figure 7(a) shows the cyclic voltammetry (CV) curves of ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 at a constant rate of 5 mV s−1 within the work potential window of 0.1–0.35 V. From each CV profile, it can be observed that redox peaks are more obvious in the case of Co3O4/NiCo2O4 as compared to ZIF-67 and NiCo2O4, and the CV integrated area of Co3O4/NiCo2O4 is the largest, indicating that it delivers a better specific capacitance and has higher redox activity than the other electrodes. 35 The CV curves of ZIF- 67, NiCo2O4, and Co3O4/NiCo2O4 under different scan rates from 5 to 80 mV s−1 are shown in Figure 7(b)–(d). On increasing the scanning speed, the CV curves still maintain a pair of representative redox peaks, while a positive shift of the oxidation peak and a negative shift of the reduction peak are also observed, indicating typical pseudocapacitive characteristics.37–39 For anodic and cathodic peaks, the mechanism of energy storage may be ascribed to the surface redox reactions of the Co2+/Co3+/Co4+ and Ni2+/Ni3+ redox couples in 6 M KOH electrolyte.40,41 The reactions can be plausibly suggested as follows9,42,43



The cyclic voltammograms of (a) ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 at a scan of 5 mV s−1, (b–d) ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 at different scan rates, and (e) square root of scan rate versus current density for Co3O4/NiCo2O4.
The linear fit is shown in Figure 7(e), which is obtained from the square root of the scan rate versus the current density. The correlation coefficient is R2 = 0.9990 for the cathodic and R2 = 0.9989 for the anodic peaks, suggesting that the obtained electrode system is controlled by a typical diffusion process. 44
The galvanostatic charge and discharge (GCD) curves of ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 at current densities of 0.5, 1.0, 1.5, and 2.0 A g−1 are shown in Figure 8. It can be observed that all the electrodes possessed no linear charge and discharge curves with notable discharge platforms, indicating the pseudocapacitance behavior of the charge- discharge process owing to the redox reactions of Co(Ⅱ) to Co(Ⅲ), Co(Ⅲ) to Co(Ⅳ), and Ni(Ⅱ) to Ni(Ⅲ). 45 The charge curve of the Co3O4/NiCo2O4 electrode exhibits a longer discharge platform among the three samples at a current density of 0.5 A g−1, which has a better faradaic redox reversibility and rate capability. 31 Figure 8(f) shows the specific capacitance of three electrodes at different current densities. The specific capacitance of Co3O4/NiCo2O4 is calculated to be 846 F g−1 at 0.5 A g−1, while the specific capacitance of NiCo2O4 and ZIF-67 are 522 and 354 F g−1. The specific capacitance (Cs) of Co3O4/NiCo2O4 was larger than those of ZIF-67 and NiCo2O4. The enhancement of the capacitive performance of Co3O4/NiCo2O4 can be attributed to its better redox reactions and higher electronic conductivity than the monometallic oxides. The valence bond exchange or charge hopping between nickel and cobalt ions can also increase electrochemical performance. 46

GCD curves (a) several electrodes at a current of 0.5 A g−1; (b–d) ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 in 6.0 M KOH with different current densities; (e) the electrochemical impedance spectra of ZIF-67, NiCo2O4, and Co3O4/NiCo2O4; and (f) the specific capacitance of ZIF-67, NiCo2O4, and Co3O4/NiCo2O4.
The electrochemical impedance spectroscopy (EIS) of the ZIF-67, NiCo2O4, and Co3O4/NiCo2O4 electrodes are shown in Figure 8(e), which can be simulated by an equivalent circuit. The abscissa of the intersection point of the impedance spectrum and the Z’ axis is the series resistance (Rs), describing the internal resistance of the electrodes, the resistance of the electrolyte, and the contact between the electrode and current collector. 47 The Rs values of Co3O4/NiCo2O4, NiCo2O4, and ZIF-67 were 0.687 Ω, 0.724 Ω, and 0.887 Ω, respectively. The slope of the straight line in the low frequency region represents the Warburg resistance (Zw), reflecting the diffusion into the electrode. A higher slope indicates a lower resistance to diffusion, evidently the Co3O4/NiCo2O4 electrode presents a bigger slope than the others.48,49 The charge transfer resistance (Rct) value of Co3O4/NiCo2O4 is estimated to be about 3.01 Ω, which is lower than those of NiCo2O4 (3.28 Ω) and ZIF-67 (3.59 Ω). In addition, NiCo2O4 shows a lower charge transfer resistance than ZIF-67, the reason is that its special hollow structure provides more active centers and improves the ion diffusion rate. This lower Rs and Rct value of Co3O4/NiCo2O4 indicates that it has a better electrochemical performance among the other electrodes. On the contrary, no completed semicircle in the high frequency region is observed in the plot for several electrodes because the electrochemical device is greatly affected by diffusive impedance from OH− ions. 50 The comparison of the Co3O4/NiCo2O4 electrode in this work with those electrodes previously reported is shown in Table 2.
Comparison of the electrochemical performance of Co3O4/NiCo2O4 in this work with previously reported studies.

Conclusion
In summary, we have successfully fabricated hollow NiCo2O4 assembled by nanosheets and sheet-like Co3O4/NiCo2O4 through preparing the MOF as a carbon skeleton, which was removed after annealing. Compared with ZIF-67 and NiCo2O4, the prepared sheet-like Co3O4/NiCo2O4 as an electrode material has a maximum specific capacitance. The hollow NiCo2O4 surface reveals a porous structure that promotes the surface redox reaction. The accelerated electron migration and diffusion speed are conducive to improving the specific capacitance of the electrode materials. After adding cobalt and nickel ions to the MOF, the specific capacitance of the electrode material (Co3O4/NiCo2O4) is 846 F g−1 at a current density of 0.5 A g−1, and the transition metal oxide (Co3O4/NiCo2O4) can increase the electric capacity due to the multi-electron transfer reaction. In addition, Co3O4/NiCo2O4 has the longest charging and discharging time, showing its good electrochemical performance. Therefore, the Co3O4/NiCo2O4 prepared in this study has excellent electrochemical properties and can be used as an electrode material in supercapacitors.
Experimental
Chemicals and materials
Ethanol, methanol, potassium hydroxide, and isopropanol were obtained from Sinopharm Chemical Reagent Co., Ltd. 2-Methylimidazole, nickel nitrate hexahydrate, cobalt nitrate hexahydrate, polytetrafluoroethylene (5 μm), and acetylene black were purchased from Aladdin. All the chemicals are analytically pure and were used without any further purification. All experimental solutions were prepared using deionized water.
Sample preparation
2-Methylimidazole (40 mmol) and Co(NO3)2·6H2O (9 mmol) were dissolved in methanol (200 mL) by stirring. The two solutions were mixed thoroughly and aged at room temperature for 24 h. By centrifuging, the precipitates were collected and then dried at 60 °C for 12 h to give ZIF-67.
Ni(NO3)2·6H2O (1 mmol) and ZIF-67 (0.2 g) prepared above were dissolved in ethanol (100 mL) by magnetic agitation for 120 min. The mixture was washed three times with distilled water and ethanol, dried at 60 °C for 12 h, and ground into a powder. Finally, the composites were calcined at 350 °C for 120 min under an air atmosphere with a heating rate of 1 °C min−1 to give NiCo2O4. Co3O4/NiCo2O4 could be acquired in the same step by adding Co(NO3)2·6H2O (2 mmol) and Ni(NO3)2·6H2O (1 mmol).
Material characterizations
The morphologies of the synthesized nanomaterials were analyzed by SEM (Zeiss Sigma 300) and TEM (JEOL JEM 2100). The purities and chemical compositions of the samples were determined by XRD using SmartLab 9 kw, operating at 40 kV and 40 mA, and the scanning range of 2θ is 5° to 90°. The states of the elements were analyzed by XPS (Thermo Scientific K-Alpha). Electrochemical measurements including CV, GCD, and EIS were obtained on an electrochemical workstation (CHI760E, China) using a three-electrode system.
Electrode preparation and electrochemical measurements
The catalyst, carbon black, and polyethylene with a mass ratio of 8:1:1 were dispersed ultrasonically for 1 h in isopropyl alcohol (2 mL) to form a mixed solution. Next, the mixed solution was coated onto the surface of nickel foam (1 cm × 1 cm) and dried at 60 °C for 12 h. A typical three-electrode system was composed of a working electrode (nickel foam), a counter electrode (a platinum gauze electrode), and a reference electrode (a saturated calomel electrode) to determine all the electrochemical properties of the samples. The electrolyte is 6.0 M KOH aqueous solution.
The specific capacitance (Cs) of several electrode materials can be calculated according to Cs = I ∆t/m ∆V, where Cs (F g−1) is the specific capacitance, I (A) is the current, m (g) is the mass of active materials, ∆t (s) is the discharge time, and ∆V (V) is the potential range of the charge-discharge. EIS was measured in the range from 100 kHz to 0.01 Hz.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from the Postgraduate Research & Practice Innovation Program of Jiangsu University of Technology (XSJCX20_04), the National Natural Science Foundation of China (grant no. 31800495), the Natural Science Foundation of Jiangsu Province (grant no. BK20181040), and the College Student Innovation and Entrepreneurship Training Program of Jiangsu Province (202011463008Z) is gratefully acknowledged.
