Abstract
A practical method to access α-ketoesters from readily available aryl acetates is developed. In this approach, aqueous tert-butyl hydroperoxide and CuO are employed. No additional solvents are required and it was found that the peroxide side products in the reaction can be decomposed by pyridine.

Introduction
α-Ketoesters are important compounds because of their bioactivities 1 and applications in natural product synthesis 2 and chemical transformations. 3 There are several methods to synthesize α-ketoesters, such as reactions of organometallic reagents with oxalic acid derivatives, 4 oxidation of α-hydroxy esters, 5 double carbonylation of organic halides, 6 and transformations of aryl acetates.7–15 Among these synthetic methods, commonly used is the reaction of organometallic reagents with oxalic acid derivatives 16 but this needs harsh conditions. It is worthwhile to find a more flexible way to produce α-ketoesters.
Aryl acetates are ideal raw materials for commercialization. The conversion of aryl acetates into α-ketoesters has been reported via two main ways: direct oxidation and through a multistep process (Scheme 1). The first example of the oxidation of arylacetic esters to arylglyoxylic esters was carried out with a vanadium-pillared montmorillonite catalyst (V-PILC) in a dry solvent (Scheme 1(a)). 9 Rh, 10 Mo, 11 Cu12–14 and Co 15 can also be used to realize this procedure, but complex catalysts needed to be prepared or long reaction times were necessary. Examples of multistep syntheses of α-ketoesters include Wasserman and Ives’s 7 work involving enamino carbonyl systems (Scheme 1(b)) and Wang’s work 8 employing oxidation of a diazo group (Scheme 1(c)). One-step direct oxidations show higher efficiencies than multistep processes. In order to enhance the economy of the direct oxidation, we studied various oxidation systems and found a new and practical method to prepare α-ketoesters.

Approaches for the synthesis of α-ketoesters from aryl acetates: : (a) a direct oxidation in a dry solvent; (b) a multistep process involving enamino carbonyl systems; (c) a multistep process employing a diazo group; (d) this work.
In this work, we report a new method to access α-ketoesters from readily available aryl acetates using aqueous tert-butyl hydroperoxide (TBHP) as an oxidant and CuO as a catalyst under reflux conditions without any additional solvent (Scheme 1(d)). We also found that the peroxide side products could be removed with pyridine.
Results and discussion
Initially, 0.2 equiv. of CuI and 3.0 equiv. of aq TBHP
17
were used to oxidize methyl 2-phenylacetate (
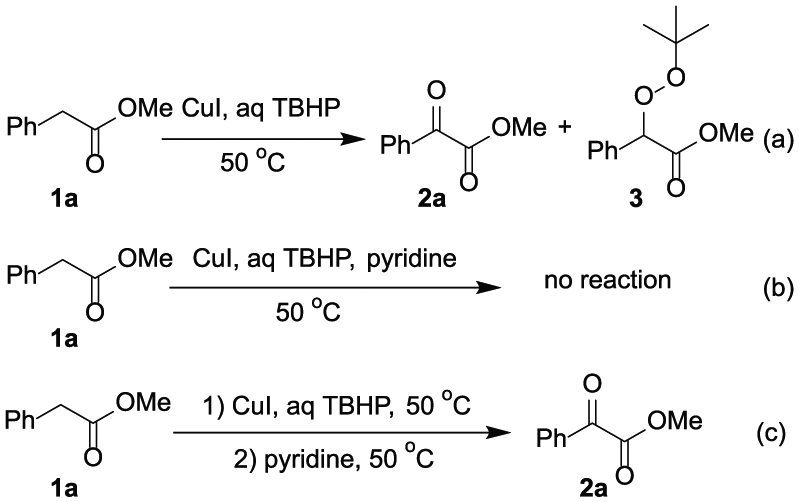
Initial results for the oxidation of methyl 2-phenylacetate: (a) the reaction of CuI, TBHP and 1a; (b) the reaction of CuI, TBHP, 1a and pyridine; (c) the reaction of CuI, TBHP and 1a was treated with pyridine.

1H NMR spectra of different systems in CDCl3: (a) the crude mixture of the reaction of CuI, TBHP and
In order to improve the yield, different solvents (MeOH, EtOAc, THF, DMF, DMSO and CH3CN) and other oxidants (H2O2 and cumene hydroperoxide) were introduced to the reacting system, however, in all these cases, no
Optimization of the reaction conditions.
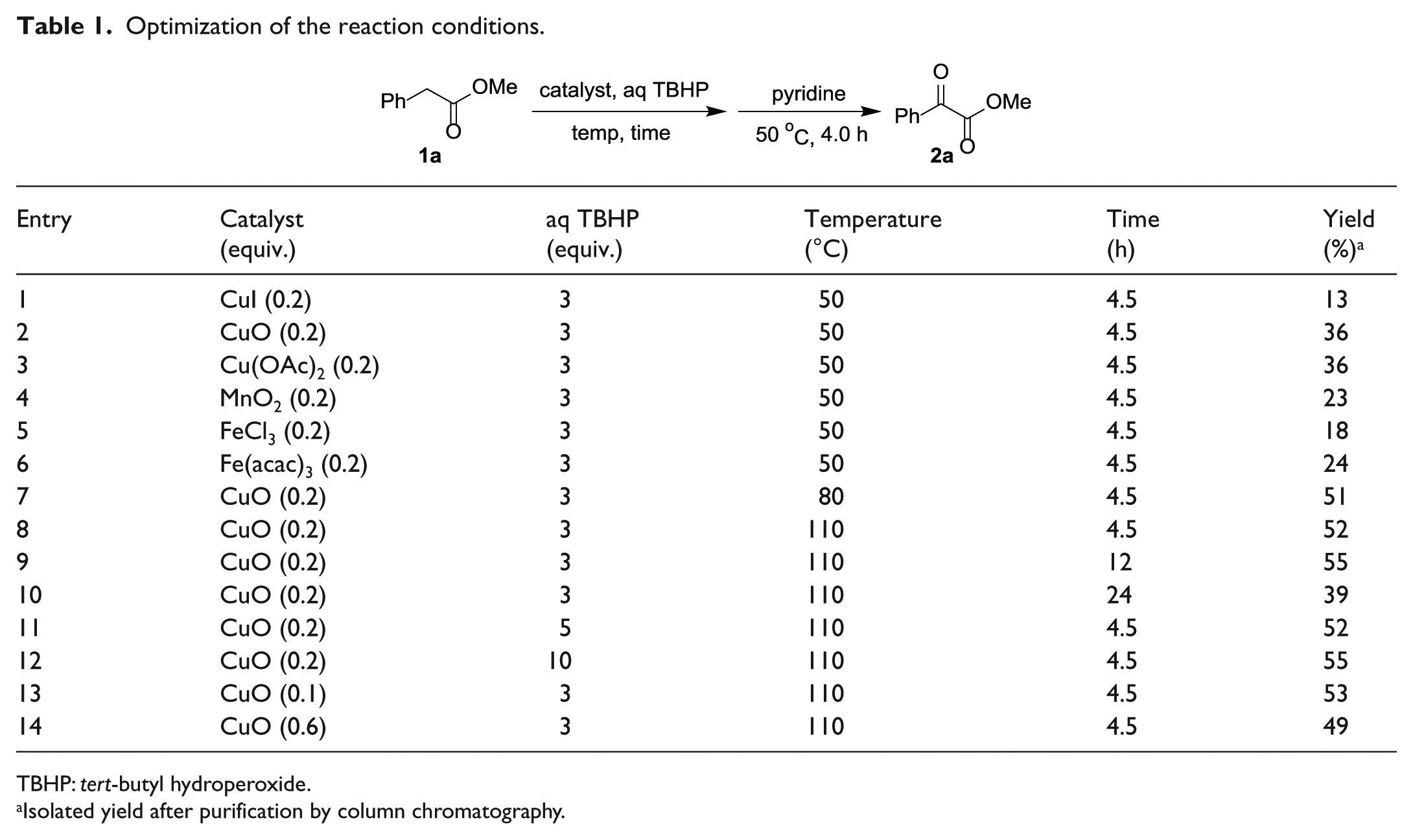
TBHP: tert-butyl hydroperoxide.
Isolated yield after purification by column chromatography.
With optimized reaction conditions in hand, various aryl acetates were used to synthesize α-ketoesters (Table 2). Methyl ester
Scope of the aryl acetates. a

Reaction conditions: (1)
All the products were identified by comparison of their analytical data with those reported in the literature.
Isolated yield.
Compounds

Large-scale preparation of

Oxidation of methyl 3-phenylpropanoate.
Based upon the above reaction facts and the relevant publications,18,19 a possible reaction mechanism is proposed in Scheme 5. Catalysed by CuO, aryl acetate

Possible reaction mechanism.
Conclusion
In conclusion, a practical method to access α-ketoesters from readily available aryl acetates under reflux conditions has been developed. In the approach, aq TBHP and CuO are employed and no additional solvents are required. Besides, the peroxide side products in the reaction could be decomposed to α-ketoesters by pyridine.
Experimental section a
Typical procedure for the synthesis of α-ketoesters
Aryl acetate
Supplemental Material
Supporting_Information_pdf – Supplemental material for CuO-catalyzed oxidation of aryl acetates with aqueous tert-butyl hydroperoxide for the synthesis of α-ketoesters
Supplemental material, Supporting_Information_pdf for CuO-catalyzed oxidation of aryl acetates with aqueous tert-butyl hydroperoxide for the synthesis of α-ketoesters by Jin Jiang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Sichuan University of Science and Engineering under grant number 2017RCL45.
Notes
Supplemental material
The spectra data for compounds 2a-p, H NMR spectra for compounds 2a-p and 4 is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
