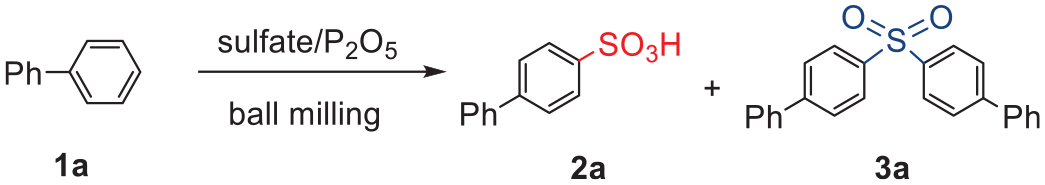
Abstract
A series of aromatic sulfonic acids is synthesized by subjecting arenes and NaHSO4·H2O to high-speed ball milling in the presence of P2O5. It is suggested the aromatic sulfonation occurs via in situ generated H2SO4 to give aromatic sulfonic acids. In some cases, formation of diaryl sulfones was observed.

Introduction
The sulfonation of aromatic compounds is of significant importance in both industry and academia.1–3 Traditional methods for the synthesis of aromatic sulfonic acids involve the sulfonylation of aromatic compounds with sulfonating agents such as concentrated sulfuric acid, 4 trifluoroacetic acid–sulfuric acid, 5 sulfur trioxide in dichloromethane, 6 silica–sulfuric acid, 7 the sulfur trioxide–dioxane complex, 8 oleum, 9 sulfur trioxide in nitrobenzene, 10 and 1,3-disulfo-1H-imidazol-3-ium chloride in aqueous media. 11 Other alternative methods include the sulfonation of Grignard and lithium reagents, 12 the Vilsmeier–Haack reaction in acetonitrile, 13 destructive oxidation of aryl thioglycolate esters, 14 and oxidation of thiols and disulfides using HOF·MeCN. 15 Despite their usefulness, these methods have limitations such as the requirements of harsh reaction conditions, prefunctionalized starting materials, high temperatures, expensive catalysts, and toxic solvents. Hence, the development of a general, facile, and efficient synthetic protocols to prepare sulfonic acid is still highly desirable.
Along with the rapid development of green chemistry, mechanochemical organic synthesis has attracted increasing attention from organic chemists.16–22 As a green alternative to traditional solution-based synthetic approaches, mechanochemical organic synthesis exhibits many advantages over its liquid-phase counterpart in terms of higher product yields, better selectivity, shorter reaction times, simple work-up procedures, elimination of harmful organic solvents, and so on. During our exploration of high-speed ball milling (HSBM) reactions,23–29 we recently found that arenes could be converted into diarylsulfones using 3CdSO4·xH2O/P2O5 as the sulfonating reagent, and noted that aromatic sulfonic acids were formed as by-products during the reaction. 29 We speculated that a higher yield of the aromatic sulfonic acid could be achieved by optimizing the HSBM conditions. We report herein the facile synthesis of aromatic sulfonic acids via the reaction of arenes and NaHSO4·H2O under HSBM conditions.
Results and discussion
Following our previous work on the synthesis of diarylsulfones, biphenyl was chosen as the standard substrate and the molar ratio of the biphenyl substrate, sulfate salt, and P2O5 was fixed at 1:3.5:11. The reactions were conducted in a standard 2.5-mL screw-capped milling beaker and milled with one stainless steel ball of 6.0 mm diameter in a high-energy vibrational MM400 mixer mill at a frequency of 28 Hz for 5 h at room temperature. A variety of different sulfate salts were tested under HSBM condition (Table 1, entries 1–7). It was found that CaSO4·0.5H2O, CdSO4·H2O, NiSO4·6H2O, MgSO4·7H2O, and Co2(SO4)3·7H2O were all ineffective in producing [1,1’-biphenyl]-4-sulfonic acid (
Optimization of the reaction conditions. a

Reaction conditions: biphenyl (0.1 mmol), reaction frequency (28 Hz), 5 h.
Molar ratio of biphenyl/sulfate/P2O5.
Yields were determined by 1H NMR.
Biphenyl (0.05 mmol) was used.The bold values indicate the optimized HSBM reaction condition.
As for organic mechanochemical reactions, the mechanochemistry process parameters usually have a strong influence on the outcomes.30–37 So, the influence of the ratios between the milling ball and the milling materials, as well as a study of the combined assessment of the grinding time and vibration frequency were carried out. To investigate the influence of the ratios between the milling ball and the milling materials, stainless steel balls of different sizes were selected for this HSBM reaction (Figure 1(a) and Table S1 in the Supporting Information). When the weight ratio between the milling ball and the milling materials was 4.45 (the diameter of the ball is 6.0 mm), the best yield of 55% of

Optimization of the mechanochemical parameters. (a) Influence of the weight ratio of the milling ball and milling materials. (b) Influence of the time and frequency.
With optimized reaction conditions in hand, we next extended the methodology to various substituted arenes. The results are summarized in Table 2. Arenes with only one activating substituent all gave a sulfonic acid as the major product (Table 2, entries 2–4). Toluene gave a 55% yield of 4-methylbenzenesulfonic acid (
Preparation of sulfonic acids

Reaction conditions:
Biphenyl (1.0 mmol) was used and reaction was carried using a polytetrafluoroethylene milling ball.
Arenes bearing multiple alkyl groups, such as o-, m-, and p-dimethylbenzene (
Naphthalene (
In order to investigate the applicability of our protocol on a larger scale, the sulfonation of a larger quantity of biphenyl (1 mmol) was performed in a polytetrafluoroethylene milling vessel at the same molar ratio as described above (Table 2, entry 16). As shown, our sulfonation reaction also worked well and [1,1’-biphenyl]-4-sulfonic acid was obtained in 58% yield.
Compared with our previous work, when 3CdSO4·xH2O/P2O5 was chosen as the sulfonating reagent under the same HSBM conditions, toluene (
It is interesting to note that the sulfonic acid was afforded along with diarylsulfones in this work, which is in contrast to our previous work, wherein the desired diarylsulfones were obtained along with sulfonic acids.
29
It appears that diarylsulfones can be converted into the corresponding sulfonic acids under HSBM conditions. In order to gain a deeper insight into our sulfonation reaction, we investigated the possibility of the decomposition of diarylsulfones, using 4,4’-sulfonyldibiphenyl (
Optimization of the conversion of diarylsulfone
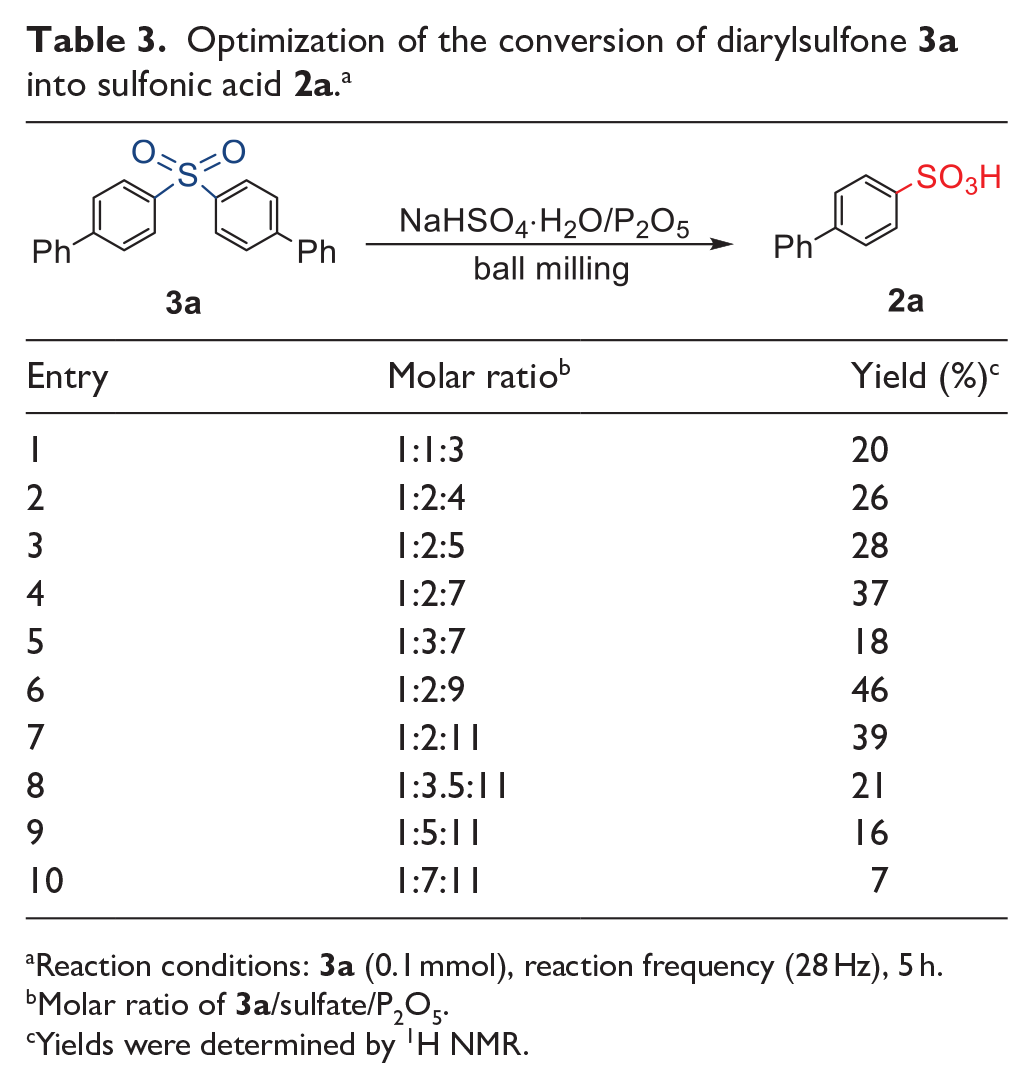
Reaction conditions:
Molar ratio of
Yields were determined by 1H NMR.
Currently, it is not easy to monitor the mechanism of an organic mechanochemical reaction. In connection with our previous work, we have proposed a plausible mechanism to demonstrate the sulfonation of aromatic compounds with a sulfate salt under HSBM condition (Scheme 1). First, the sulfate reacts with P2O5 to produce H2SO4. Subsequently, the aromatic compound

A plausible reaction mechanism.
Conclusion
In summary, we have shown that the combination of NaHSO4·H2O and P2O5 can successfully convert aromatic compounds into sulfonic acids in moderate yields under HSBM conditions. The method is facile, eco-friendly, operationally simple, and can be performed safely on large scale. We propose that the in situ generated H2SO4 from the reaction of the sulfate and P2O5 participates in the aromatic sulfonation to afford the aromatic sulfonic acid.
Experimental
General
All chemicals and reagents were purchased from Sigma-Aldrich (Billerica, MA, USA), Alfa Aesar (Ward Hill, MA, USA), or TCI (Tokyo, Japan), and were used without further purification. The reactions were conducted in a stainless milling beaker (2.5 mL) using a stainless milling ball or in a polytetrafluoroethylene milling beaker (35 mL) using a polytetrafluoroethylene milling ball (ø = 19.05 mm) in an MM400 mixer ball mill (Retsch GmbH, Germany). Column chromatography was performed using 100–200 mesh silica gel purchased from the Qingdao Haiyang Chemical Co., Ltd. Appropriate solvent systems for chromatography were selected according to TLC analysis using UV light (254 nm) to visualize the reaction components. NMR spectra were recorded on Bruker AV 400 spectrometer at 400 MHz (1H NMR) and at 101 MHz (13C NMR). TMS was used as the internal standard. The peak patterns are designated as follows: s, singlet; d, doublet; t, triplet; q, quartet; dd, doublet-doublet; dt, doublet-triplets; m, multiplet. Coupling constants, J, are reported in Hz. The product was dissolved in an appropriate deuterium reagent with 3.5 μL of dibromomethane as a reference. An APEX II (Bruker Inc.) mass spectrometer was used for HRMS analysis.
General procedure of compounds 2 and 3
A mixture of arene (
[1,1’-Biphenyl]-4-sulfonic acid (2a )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:2) in 61% yield (71.37 mg); white solid; m.p. 138–141 °C (lit. 29 134–136 °C); 1H NMR (400 MHz, D2O): δ 7.88 (d, J = 8.5 Hz, 2H), 7.78 (d, J = 8.6 Hz, 2H), 7.72 (d, J = 7.2 Hz, 2H), 7.53 (t, J = 7.4 Hz, 2H), 7.46 (t, J = 7.3 Hz, 1H); 13C NMR (101 MHz, D2O): δ 143.59, 141.21, 139.36, 129.15, 128.34, 127.38, 127.17, 126.01; HRMS (ESI): m/z [M − H]− calcd for C12H9O3S: 233.0278; found: 233.0274.
4,4’-Sulfonyldi-1,1’-biphenyl (3a )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 75:1) in 27% yield (21.28 mg); white solid; m.p. 213–214 °C (lit. 29 213–216 °C); 1H NMR (400 MHz, CDCl3): δ 8.05 (d, J = 8.4 Hz, 4H), 7.72 (d, J = 8.5 Hz, 4H), 7.57 (d, J = 7.0 Hz, 4H), 7.50-7.35 (m, 6H); 13C NMR (101 MHz, CDCl3): δ 146.33, 140.45, 139.37, 129.26, 128.71, 128.32, 128.19, 127.55; HRMS (ESI): m/z [M + H]+ calcd for C24H19O2S: 371.1100; found: 371.1103.
4-Methylbenzenesulfonic acid (2b )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 2:3) in 55% yield (47.57 mg); white crystalline solid; m.p. 104–106 °C (lit. 41 99–102 °C); 1H NMR (400 MHz, D2O): δ 7.67 (d, J = 8.3 Hz, 2H), 7.35 (d, J = 8.0 Hz, 2H), 2.37 (s, 3H); 13C NMR (101 MHz, D2O): δ 176.61, 129.39, 125.32, 100.00, 20.33; HRMS (ESI): m/z [M − H]− calcd for C7H7O3S: 171.0121; found: 171.0124.
4,4’-Sulfonylbis(methylbenzene) (3b )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 65:1) in 37% yield (22.75 mg); white solid; m.p. 156–158 °C (lit. 29 155–158 °C); 1H NMR (400 MHz, CDCl3): δ 7.73 (d, J = 8.3 Hz, 4H), 7.16 (d, J = 8.1 Hz, 4H), 2.24 (s, 6H); 13C NMR (101 MHz, CDCl3): δ 143.73, 138.82, 129.65, 127.22, 21.21; HRMS (ESI): m/z [M + Na]+ calcd for C14H14O2SNa: 269.0607; found: 269.0605.
4-Ethylbenzenesulfonic acid (2c )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 5:6) in 57% yield (55.82 mg); brown crystalline solid; m.p. 109–111 °C; 1H NMR (400 MHz, D2O): δ 7.33 (d, J = 8.3 Hz, 2H), 6.90 (d, J = 8.3 Hz, 2H), 2.19 (q, J = 7.6 Hz, 2H), 0.73 (t, J = 7.6 Hz, 3H); 13C NMR (101 MHz, D2O): δ 148.28, 139.42, 128.04, 125.22, 27.84, 14.39; HRMS (ESI): m/z [M − H]− calcd for C8H9O3S: 185.0278; found: 185.0275.
4,4’-Sulfonylbis(ethylbenzene) (3c )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 120:1) in 28% yield (19.25 mg); white solid; m.p. 92–95 °C (lit. 29 93–95 °C); 1H NMR (400 MHz, CDCl3): δ 7.76 (d, J = 8.2 Hz, 4H), 7.18 (d, J = 8.1 Hz, 4H), 2.51 (q, J = 7.6 Hz, 4H), 1.05 (t, J = 7.7 Hz, 6H); 13C NMR (101 MHz, CDCl3): δ 149.84, 138.92, 128.43, 127.37, 28.41, 14.76; HRMS (ESI): m/z [M + Na]+ calcd for C16H18O2SNa: 297.0920; found: 297.0919.
4-Methoxybenzenesulfonic acid (2d )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:1) in 62% yield (58.65 mg); brown crystalline solid; m.p. 83–85 °C (lit. 41 88–90 °C); 1H NMR (400 MHz, D2O): δ 7.56 (d, J = 8.9 Hz, 2H), 6.84 (d, J = 8.9 Hz, 2H), 3.64 (s, 3H); 13C NMR (101 MHz, D2O): δ 161.01, 134.71, 127.21, 113.98, 55.38; HRMS (ESI): m/z [M − H]− calcd for C7H7O4S: 187.0071; found: 187.0070.
4,4’-Sulfonylbis(methoxybenzene) (3d )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 100:1) in 31% yield (20.89 mg); white solid; m.p. 125–127 °C (lit. 29 124–126 °C); 1H NMR (400 MHz, CDCl3): δ 7.81 (d, J = 8.9 Hz, 4H), 6.91 (d, J = 8.9 Hz, 4H), 3.78 (s, 6H); 13C NMR (101 MHz, CDCl3): δ 163.13, 133.96, 129.43, 114.49, 55.67; HRMS (ESI): m/z [M + H]+ calcd for C14H15O4S: 279.0686; found: 279.0683.
4-Fluorobenzenesulfonic acid (2e )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:3) in 59% yield (51.79 mg); brown solid; m.p. 89–92 °C (lit. 42 87 °C); 1H NMR (400 MHz, D2O): δ 7.92-7.75 (m, 2H), 7.24 (t, J = 8.9 Hz, 2H); 13C NMR (101 MHz, D2O): δ 163.86 (d, J = 248.9 Hz), 138.56, 127.88 (d, J = 8.9 Hz), 115.67 (d, J = 22.6 Hz); HRMS (ESI): m/z [M − H]− calcd for C6H4O3FS: 174.9871; found: 174.9878.
4,4’-Sulfonylbis(fluorobenzene) (3e )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 65:1) in 31% yield (19.71 mg); white solid; m.p. 99–102 °C (lit. 29 96–98 °C); 1H NMR (400 MHz, CDCl3): δ 7.94 (dd, J = 8.9, 5.0 Hz, 4H), 7.18 (t, J = 8.6 Hz, 4H); 13C NMR (101 MHz, CDCl3): δ 165.50 (d, J = 256.2 Hz), 137.72, 130.42 (d, J = 9.6 Hz), 116.75 (d, J = 22.7 Hz); HRMS (ESI): m/z [M + H]+ calcd for C12H9O2F2S: 255.0286; found: 255.0289.
4-Chlorobenzenesulfonic acid (2f )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 2:3) in 52% yield (49.96 mg); yellow solid; m.p. 87–91 °C (lit. 41 85–87 °C); 1H NMR (400 MHz, D2O): δ 7.72 (d, J = 8.7 Hz, 2H), 7.50 (d, J = 8.7 Hz, 2H); 13C NMR (101 MHz, D2O): δ 140.92, 136.95, 128.99, 127.02; HRMS (ESI): m/z [M − H]− calcd for C6H4O3ClS: 190.9575; found: 190.9578.
4,4’-Sulfonylbis(chlorobenzene) (3f )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 85:1) in 40% yield (29.04 mg); white solid; m.p. 145–148 °C (lit. 29 145–148 °C); 1H NMR (400 MHz, CDCl3): δ 7.67 (d, J = 10.8 Hz, 4H), 7.28 (d, J = 8.6 Hz, 4H); 13C NMR (101 MHz, CDCl3): δ 140.34, 139.85, 129.82, 129.29; HRMS (ESI): m/z [M + Na]+ calcd for C12H8O2Cl2SNa: 308.9514; found: 308.9511.
4-Bromobenzenesulfonic acid (2g )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:2) in 60% yield (70.75 mg); yellow solid; m.p. 91–94 °C (lit. 43 88–90 °C); 1H NMR (400 MHz, D2O): δ 7.72 (d, J = 9.0 Hz, 2H), 7.70 (d, J = 9.2 Hz, 2H); 13C NMR (101 MHz, D2O): δ 141.44, 132.04, 127.20, 125.40; HRMS (ESI): m/z [M − H]− calcd for C6H4O3BrS: 234.9070; found: 234.9072.
4,4’-Sulfonylbis(bromobenzene) (3g )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 50:1) in 34% yield (31.71 mg); white solid; m.p. 161–164 °C (lit. 29 161–165 °C); 1H NMR (400 MHz, CDCl3): δ 7.78 (d, J = 8.6 Hz, 4H), 7.64 (d, J = 8.6 Hz, 4H); 13C NMR (101 MHz, CDCl3): δ 140.36, 132.95, 129.37, 128.94; HRMS (ESI): m/z [M + Na]+ calcd for C12H8O2Br2SNa: 396.8504; found: 396.8501.
3,4-Dimethylbenzenesulfonic acid (2h )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:3) in 58% yield (53.77 mg); white solid; m.p. 85–88 °C (lit. 41 81–83 °C); 1H NMR (400 MHz, D2O): δ 7.34 (s, 1H), 7.28 (d, J = 7.9 Hz, 1H), 7.07 (d, J = 7.9 Hz, 1H), 2.06 (d, J = 4.1 Hz, 6H); 13C NMR (101 MHz, D2O): δ 142.06, 137.61, 135.78, 132.40, 126.61, 126.08, 20.03, 19.26; HRMS (ESI): m/z [M − H]− calcd for C8H9O3S: 185.0278; found: 185.0277.
4,4’-Sulfonylbis(1,2-dimethylbenzene) (3h )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 65:1) in 32% yield (21.92 mg); white solid; m.p. 157–159 °C (lit. 29 159–161 °C); 1H NMR (400 MHz, CDCl3): δ 7.70-7.61 (m, 4H), 7.19 (d, J = 7.9 Hz, 2H), 2.23 (d, J = 5.4 Hz, 12H); 13C NMR (101 MHz, CDCl3): δ 142.55, 139.26, 137.87, 130.27, 128.19, 124.92, 19.73, 19.65; HRMS (ESI): m/z [M + H]+ calcd for C16H19O2S: 275.1100; found: 275.1102.
2,4-Dimethylbenzenesulfonic acid (2i )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 5:9) in 64% yield (59.56 mg); white solid; m.p. 65–68 °C (lit. 41 61–63 °C); 1H NMR (400 MHz, D2O): δ 7.71 (d, J = 8.0 Hz, 1H), 7.19 (s, 1H), 7.13 (d, J = 7.9 Hz, 1H), 2.54 (s, 3H), 2.31 (s, 3H); 13C NMR (101 MHz, D2O): δ 142.39, 137.75, 136.06, 132.59, 126.83, 126.28, 20.18, 19.35; HRMS (ESI): m/z [M − H]− calcd for C8H9O3S: 185.0278; found: 185.0273.
4,4’-Sulfonylbis(1,3-dimethylbenzene) (3i )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 76:1) in 25% yield (17.14 mg); white solid; m.p. 117–121 °C (lit. 44 122–125 °C); 1H NMR (400 MHz, CDCl3): δ 8.03 (d, J = 8.1 Hz, 2H), 7.13 (d, J = 8.1 Hz, 2H), 6.98 (s, 2H), 2.31 (s, 6H), 2.26 (s, 6H); 13C NMR (101 MHz, CDCl3): δ 144.04, 137.45, 136.27, 133.24, 129.89, 126.63, 21.35, 19.86; HRMS (ESI): m/z [M + H]+ calcd for C16H19O2S: 275.1100; found: 275.1106.
2,5-Dimethylbenzenesulfonic acid (2j )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:2) in 50% yield (46.53 mg); white solid; m.p. 95–98 °C (lit. 41 89–91 °C); 1H NMR (400 MHz, D2O): δ 7.57 (s, 1H), 7.19-7.09 (m, 2H), 2.44 (s, 3H), 2.21 (s, 3H); 13C NMR (101 MHz, D2O): δ 140.27, 136.03, 132.83, 132.00, 131.95, 127.03, 19.91, 18.97; HRMS (ESI): m/z [M − H]− calcd for C8H9O3S: 185.0278; found: 185.0279.
2,2’-Sulfonylbis(1,4-dimethylbenzene) (3j )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 55:1) in 27% yield (18.52 mg); white solid; m.p. 167–169 °C (lit. 44 163–165 °C); 1H NMR (400 MHz, CDCl3): δ 7.99 (s, 2H), 7.27 (d, J = 7.6 Hz, 2H), 7.09 (d, J = 7.7 Hz, 2H), 2.40 (s, 6H), 2.26 (s, 6H); 13C NMR (101 MHz, CDCl3): δ 138.71, 136.12, 134.63, 134.27, 132.65, 129.90, 21.07, 19.64; HRMS (ESI): m/z [M + Na]+ calcd for C16H18O2SNa: 297.0920; found: 297.0922.
2-Methyl-5-nitrobenzenesulfonic acid (2k )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:4) in 47% yield (50.98 mg); yellow solid; m.p. 137–139 °C (lit. 45 132–133 °C); 1H NMR (400 MHz, D2O): δ 8.25 (s, 1H), 7.82 (d, J = 8.4 Hz, 1H), 7.16 (d, J = 8.4 Hz, 1H), 2.38 (s, 3H); 13C NMR (101 MHz, D2O): δ 144.91, 144.56, 141.83, 133.01, 125.45, 121.77, 19.61; HRMS (ESI): m/z [M − H]− calcd for C7H6O5NS: 215.9972; found: 215.9978.
4-Methyl-3-sulfobenzoic acid (2l )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:3) in 49% yield (53.17 mg); yellow solid; m.p. 262–265 °C; 1H NMR (600 MHz, MeOD): δ 8.04 (s, 1H), 7.98 (q, J = 8.4 Hz, 2H), 2.57 (s, 3H); 13C NMR (101 MHz, MeOD): δ 166.33, 151.99, 134.39, 133.57, 132.84, 127.93, 124.04, 18.35; HRMS (ESI): m/z [M − H]− calcd for C8H7O5S: 215.0019; found: 215.0015.
4-Methoxy-3-sulfobenzoic acid (2m )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:4) in 50% yield (63.76 mg); yellow solid; m.p. 256–257 °C (lit. 46 250–251 °C); 1H NMR (400 MHz, D2O): δ 7.39 (s, 1H), 7.03 (d, J = 8.7 Hz, 1H), 6.19 (d, J = 8.8 Hz, 1H), 3.07 (s, 3H); 13C NMR (101 MHz, D2O): δ 166.80, 159.63, 134.29, 129.39, 129.05, 120.65, 111.78, 55.62; HRMS (ESI): m/z [M − H]− calcd for C8H7O6S: 230.9969; found: 230.9965.
Naphthalene-1-sulfonic acid (2n )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:3) in 71% yield (73.84 mg); yellow solid; m.p. 80–85 °C (lit. 41 79–81 °C); 1H NMR (400 MHz, D2O): δ 7.91 (d, J = 5.2 Hz, 1H), 7.44 (q, J = 8.6 Hz, 2H), 7.41-7.35 (m, 1H), 7.91 (s, 1H), 7.12-6.98 (m, 2H); 13C NMR (101 MHz, D2O): δ 141.15, 135.46, 130.72, 130.36, 129.64, 129.25, 127.20, 123.47; HRMS (ESI): m/z [M − H]− calcd for C10H7O3S: 207.0126; found: 207.0121.
1,1’-Sulfonyldinaphthalene (3n )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 75:1) in 21% yield (33.39 mg); white solid; m.p. 144–146 °C (lit. 47 143–146 °C); 1H NMR (400 MHz, CDCl3): δ 8.34 (d, J = 4.4 Hz, 2H), 7.98-7.82 (m, 8H), 7.55 (dd, J = 6.5, 2.6 Hz, 4H); 13C NMR (101 MHz, CDCl3): δ 141.62, 134.27, 132.55, 128.63, 128.48, 127.69, 127.53, 126.91, 125.44, 122.76; HRMS (ESI): m/z [M + Na]+ calcd for C20H14O2SNa: 341.0601; found: 341.0607.
Thiophene-2-sulfonic acid (2o )
Isolated by silica gel column chromatography (dichloromethane/ethyl acetate = 1:4) in 77% yield (63.03 mg); yellow solid; m.p. 72–75 °C (lit. 48 71–72 °C); 1H NMR (400 MHz, D2O): δ 7.38 (d, J = 6.3 Hz, 1H), 7.26 (d, J = 3.7 Hz, 1H), 6.92-6.84 (m, 1H); 13C NMR (101 MHz, D2O): δ 129.39, 128.72, 128.26, 126.76; HRMS (ESI): m/z [M − H]− calcd for C4H3O3S2: 162.9529; found: 162.9526.
2,2’-Sulfonyldithiophene (3o )
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 86:1) in 12% yield (6.89 mg); white solid; m.p. 127–129 °C (lit. 29 123–125 °C); 1H NMR (400 MHz, CDCl3): δ 7.72 (d, J = 3.8 Hz, 2H), 7.65 (d, J = 5.0 Hz, 2H), 7.10-7.05 (m, 2H); 13C NMR (101 MHz, CDCl3): δ 133.95, 133.47, 131.08, 127.91; HRMS (ESI): m/z [M + Na]+ calcd for C8H6O2S3Na: 252.9422; found: 252.9420.
Supplemental Material
sj-docx-1-chl-10.1177_17475198211032571 – Supplemental material for Mechanochemical sulfonation of aromatic compounds using NaHSO4·H2O/P2O5
Supplemental material, sj-docx-1-chl-10.1177_17475198211032571 for Mechanochemical sulfonation of aromatic compounds using NaHSO4·H2O/P2O5 by Lu-Lu Zuo, Shuai Qin, Pu Zhang, Yu-Jun Qin and Zhi-Xin Guo in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are thankful to the Renmin University of China for providing necessary facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.