Abstract
Aryl thioesters are synthesized via nickel-catalyzed carbonylation of thioacetates with aryl iodides. Alkyl thioacetates undergo coupling with carbon monoxide and aryl iodides to produce the desired aryl thioesters in moderate yields. This catalytic carbonylative coupling process provides a cost-effective and direct approach for the preparation of useful thioesters.

Introduction
The thioester unit is a very useful and powerful building block in organic chemistry 1 and is also an important component of many natural products 2 and agrochemicals. 3 Specifically, thioesters have often been employed as intermediates for the synthesis of amides, esters, and ketones.4–8 In addition, thioesters also serve as important intermediates in the biosynthesis of polyketides and nonribosomal polypeptides. 9 Due to their many applications, numerous synthetic methods have been developed for the preparation of thioesters, for example, palladium-catalyzed carbonylation of aryl halides and alcohols, 10 palladium-catalyzed carbonylation of thioacetates and aryl iodides, 11 palladium-catalyzed intermolecular transthioetherification of aryl halides with thioethers, 12 condensation of carboxylic acids and alcohols, 13 copper-catalyzed esterification of disulfides and acyl chlorides, 14 and decarboxylative coupling of α-oxocarboxylic acids and disulfides. 15 These methods suffer from certain disadvantages such as harsh conditions, high costs, formation of by-products, and the use of excess reagents. More recently, new progress was reported in this field, in which some novel protocols were developed for the synthesis of thioesters.16–19 Herein, we report a novel process for the synthesis of aryl thioesters via nickel-catalyzed carbonylation of thioacetates or dialkyl disulfides with aryl iodides under mild conditions.
Results and discussion
In previous work, Kim et al.
11
reported aryl thioacetates as the source of sulfur in the palladium-catalyzed carbonylation of aryl iodides for the formation of thioesters under CO (8 atm). Inspired by this result, we tried to use nickel as the catalyst for this transformation under 1 atm of CO; however, the reaction did not occur at all. To our surprise, alkyl thioacetates were compatible with the Ni catalyst, unlike aryl thioacetates under the same conditions. Thus, we chose iodobenzene (
Optimization of the reaction conditions a .

Reaction conditions:
DTBPy: 4,4’-di-tert-butyl-2,2’-bipyridine, Bpy: 2,2’-bipyridine, Phen: 1,10-phenanthroline.
GC yield.
1 atm CO balloon.
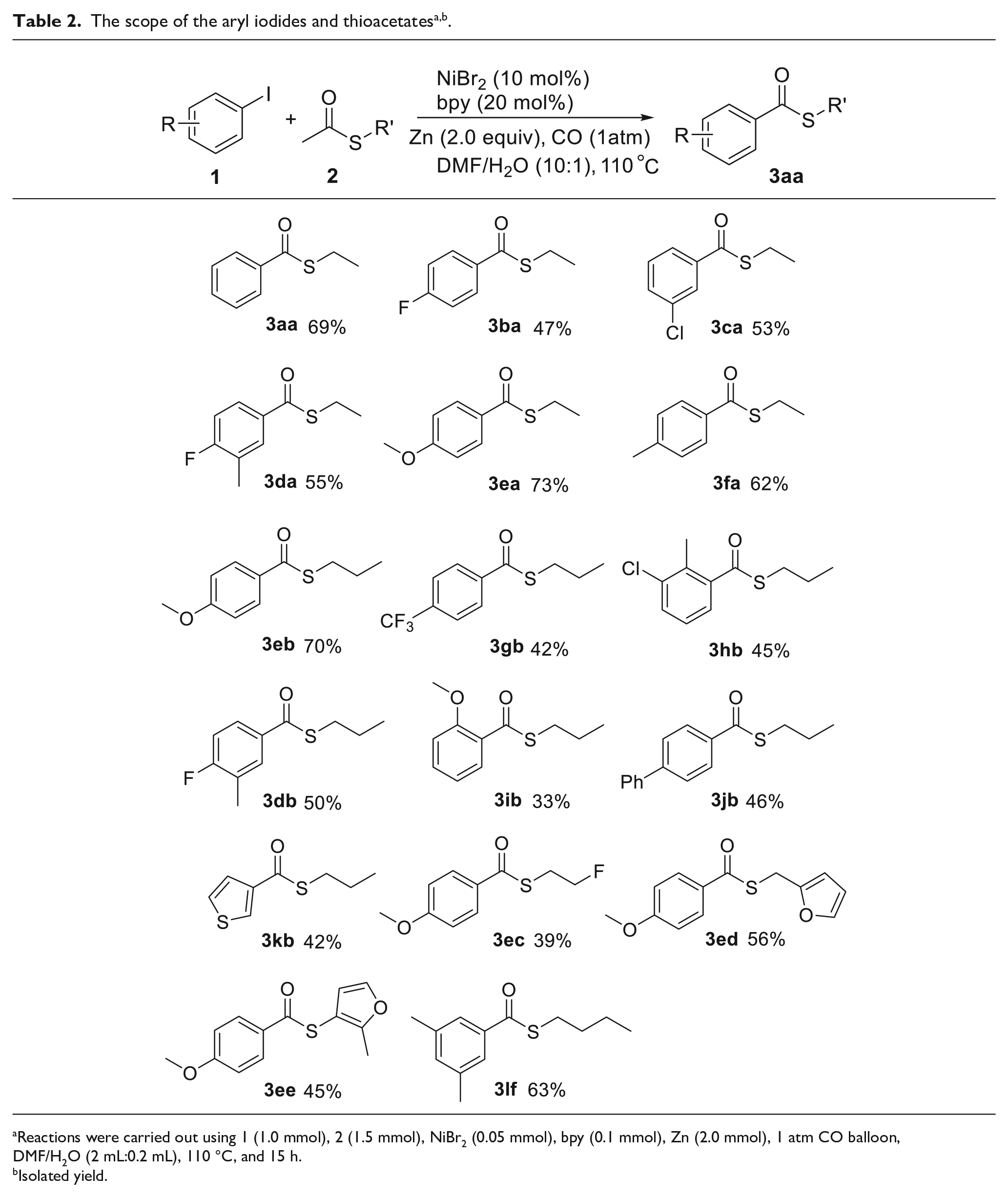
To investigate the scope of this transformation, many aryl iodides were examined for the formation of the corresponding aryl thioester under the optimized reaction conditions and the results are shown in Table 2. Both aryl iodides which have electron-donating and electron-withdrawing groups were found to react with thioacetates and gave the products in moderate yields. Generally, those with electron-donating groups on the aryl ring favored the transformation. For example, 1-iodo-4-methoxybenzene (
Reactions were carried out using 1 (1.0 mmol), 2 (1.5 mmol), NiBr2 (0.05 mmol), bpy (0.1 mmol), Zn (2.0 mmol), 1 atm CO balloon, DMF/H2O (2 mL:0.2 mL), 110 °C, and 15 h.
Isolated yield.
When S-propyl ethanethioate (
To study further the mechanism of this transformation, alkyl sulfides were selected as one of the substrates to carry out the reaction under the optimized conditions. To our delight, the results showed that the yields of the desired products were higher in comparison with those obtained when alkyl ethanethioates were employed as substrates (Scheme 1). For example, dibutylsulfane and S-butyl ethanethioate reacted with 1-iodo-3,5-dimethylbenzene under the same conditions, leading to the same product in 71% and 63%, respectively. These results indicated that insertion of Ni(I) into the S–S bond is easier and that the insertion of CO occurred after C–S or S–S bond cleavage.

Alkyl sulfides as substrates.
Based on the above the experimental data and previous work,
17
a plausible reaction mechanism is proposed in Scheme 2. First, an Ni(0) species is formed by reduction with Zn powder, then, the species inserts into the Ph–I bond of the iodobenzene to form the intermediate

Plausible reaction mechanism.
Conclusion
In conclusion, we have established a novel nickel-catalyzed carbonylation of aryl iodides and thioacetates for the synthesis of thioesters. Numerous aryl iodides reacted to give the desired S-alkyl thioesters in moderate yields under 1 atm of CO. This methodology is also useful with dialkyl sulfides and afforded the corresponding products in moderate yields under the same conditions. Advantageously, the reaction occurs in one pot, using a cheap catalyst, mild conditions, and a simple procedure. Unfortunately, this one-pot protocol for the formation of S-thioesters was unsuccessful with S-aryl ethanethioates as substrates as used in our previous work. 20
Experimental
All experiments were carried out using a Schlenk flask open to air. Aryl iodides and thioacetates were purchased from commercial suppliers and were used as received, unless otherwise noted. All solvents and other commercially available reagents were purchased from TCI company and used directly. Reactions were monitored by TLC (Qingdao Haiyang Chemical Co., Ltd., Silica gel 60 F254). Products were detected using a UV-Vis lamp (254 nm). Column chromatography was performed on Qingdao Haiyang Chemical Co., Ltd., Silica Gel 60 (200–300 mesh). The 1H and 13C NMR spectra were obtained on a Bruker 400 MHz NMR Fourier transform spectrometer. 1H NMR data are reported as: chemical shift (δ ppm), multiplicity, coupling constant (Hz), and integration. 13C NMR data are reported in terms of chemical shift (δ ppm), multiplicity, and coupling constant (Hz) where applicable. The spectra are referenced against the internal non-deuterated solvent (CDCl3, δ 1H = 7.26 ppm, 13C = 77.0 ppm). Data are reported as follows: s = singlet, d = doublet, t = triplet, q = quartet, and m = multiplet. ESI-MS spectra were recorded on a Bruker Esquire 3000 instrument.
General procedures for the one-pot synthesis of thioesters 3
A Schlenk flask equipped with a magnetic stir bar was charged with S-alkyl thioacetate
S-ethyl benzothioate (3aa ): Yield:
114 mg (69%); yellowish liquid (lit. 13 ). 1H NMR (400 MHz, CDCl3): δ 7.80 (d, J = 8.0 Hz, 2H), 7.58-7.57 (m, 1H), 7.49-7.45 (m, 2H), 3.13-3.08 (m, 3H), 1.38 (t, J = 8.0 Hz, 2H). 13C NMR (101 MHz, CDCl3): δ 14.75, 23.44, 127.16, 128.56, 133.20, 137.30, 192.08. HRMS (EI): m/z [M + H]+ calcd for C9H11OS: 167.0531; found: 167.0533.
S-ethyl 4-fluorobenzothioate (3ba ):
Yield: 86 mg (47%); yellowish liquid (lit. 21 ). 1H NMR (400 MHz, CDCl3): δ 8.03-7.99 (m, 2H), 7.14 (t, J = 8.0 Hz, 2H), 3.13-3.07 (m, 2H), 1.38 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 14.73, 23.57, 115.56 (d, JC-F = 22 Hz), 129.63 (d, JC-F = 9 Hz), 133.64, 167.12 (d, JC-F = 252 Hz), 190.57. HRMS (EI): m/z [M + H]+ calcd for C9H10FOS: 185.0436; found: 185.0432.
S-ethyl 3-chlorobenzothioate (3ca ):
Yield: 106 mg (53%); colorless liquid. 1H NMR (400 MHz, CDCl3): δ 7.95 (s, 1H), 7.87 (d, J = 8.0 Hz, 1H), 7.39 (t, J = 8.0 Hz, 1H), 3.14-3.08 (m, 2H), 1.38 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 14.66, 23.68, 125.29, 127.22, 129.88, 133.11, 134.52, 138.76, 190.90. HRMS (EI): m/z [M + H]+ calcd for C9H9ClOS: 200.0063; found: 200.0058.
S-ethyl 4-fluoro-3-methylbenzothioate (3da ):
Yield: 109 mg (55%); colorless liquid. 1H NMR (400 MHz, CDCl3): δ 7.85-7.81 (m, 2H), 7.06 (t, J = 8.0 Hz, 1H), 3.11-3.05 (m, 2H), 2.33 (s, 3H), 1.38-1.34 (m, 3H). 13C NMR (101 MHz, CDCl3): δ 14.47, 14.50, 14.75, 23.51, 115.35 (d, JC-F = 25 Hz), 125.49 (d, JC-F = 18 Hz), 127.03, 130.76, 133.32 (d, JC-F = 3.0 Hz), 165.70 (d, JC-F = 252 Hz), 190.74. HRMS (EI): m/z [M + H]+ calcd for C10H12FOS: 199.0593; found: 199.0588.
S-ethyl 4-methoxybenzothioate (3ea ):
Yield: 143 mg (73%); yellow liquid (lit. 13 ). 1H NMR (400 MHz, CDCl3): δ 7.96 (d, J = 8.0 Hz, 2H), 6.93 (t, J = 8.0 Hz, 2H), 3.88 (d, J = 8.0 Hz, 3H), 3.09-3.05 (m, 2H), 1.37-1.33 (m, 3H). 13C NMR (101 MHz, CDCl3): δ 14.90, 23.29, 55.46, 113.71, 129.28, 130.16, 163.66, 190.55. HRMS (EI): m/z [M + H]+ calcd for C10H13O2S: 197.0636; found: 197.0633.
S-ethyl 4-methylbenzothioate (3fa ):
Yield: 112 mg (62%); yellow liquid (lit.14). 1H NMR (400 MHz, CDC l3 ): δ 7.90 (d, J = 12 Hz, 2H), 7.27 (t, J = 8.0 Hz, 2H), 3.12-3.06 (m, 2H), 2.43 (s, 3H), 1.37 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 14.83, 21.67, 23.34, 127.23, 129.23, 134.75, 144.05, 191.74 HRMS (EI) m/z [M + H]+ calcd for C10H13OS 181.0687; found: 181.0685.
S-propyl 4-methoxybenzothioate (3eb ):
Yield: 147 mg (70%); yellowish liquid (lit.13). 1H NMR (400 MHz, CDCl3): δ 7.98 (d, J = 8.0 Hz, 2H), 6.95 (d, J = 8.0 Hz, 2H), 3.88 (s, 3H), 3.06 (t, J = 8.0 Hz, 2H), 1.74-1.69 (m, 2H), 1.05 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.47, 23.12, 30.81, 55.49, 113.71, 129.33, 130.20, 163.65, 190.65. HRMS (EI): m/z [M + H]+ calcd for C11H15O2S 211.0793; found: 211.0787.
S-propyl 4-(trifluoromethyl)benzothioate (3gb ):
Yield: 104 mg (42%); yellowish liquid. 1H NMR (400 MHz, CDCl3): δ 8.10 (d, J = 8.0 Hz, 2H), 7.74 (d, J = 8.0 Hz, 2H), 3.11 (t, J = 8.0 Hz, 2H), 1.77-1.72 (m, 2H), 1.06 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.41, 22.84, 31.22, 122.21 (t, JC-F = 271 Hz), 125.71 (d, JC-F = 8.0 Hz), 134.68 (d, JC-F = 32 Hz), 140.00, 191.21. HRMS (EI): m/z [M + H]+ calcd for C11H12F3OS: 249.0561; found: 249.0558.
S-propyl 3-chloro-2-methylbenzothioate (3hb ):
Yield: 103 mg (45%); colorless liquid. 1H NMR (400 MHz, CDCl3): δ 7.56 (d, J = 8.0 Hz, 1H), 7.50 (d, J = 8.0 Hz, 1H), 7.20 (t, J = 8.0 Hz, 1H), 3.06 (t, J = 8.0 Hz, 2H), 2.47 (s, 3H), 1.77-1.72 (m, 2H), 1.07 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.40, 16.76, 22.91, 31.82, 126.14, 126.51, 131.83, 133.88, 140.71, 194.74. HRMS (EI): m/z [M + H]+ calcd for C11H14ClOS: 229.0454; found: 229.0450.
S-(4-fluoro-3-methylphenyl)butanethioate (3db ):
Yield: 106 mg (50%); yellowish liquid. 1H NMR (400 MHz, CDCl3): δ 7.87-7.82 (m, 2H), 7.10 (t, J = 8.0 Hz, 1H), 3.07 (t, J = 8.0 Hz, 2H), 2.35 (d, J = 4.0 Hz, 3H), 1.75-1.70 (m, 2H), 1.06 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.42, 14.53 (d, JC-F = 3.0 Hz), 22.99, 31.02, 115.35 (d, JC-F = 23 Hz), 125.49 (d, JC-F = 18 Hz), 127.06, 130.80, 133.32, 163.19, 165.70, 190.82. HRMS (EI): m/z [M + H]+ calcd for C11H14FOS: 213.0749; found: 213.0745.
S-propyl 2-methoxybenzothioate (3ib ):
Yield: 69 mg (33%); yellowish liquid. 1H NMR (400 MHz, CDCl3): δ 7.80-7.78 (m, 1H), 7.49-7.46 (m, 1H), 7.04-6.99 (m, 2H), 3.94 (s, 3H), 3.04 (t, J = 8.0 Hz, 2H), 1.75-1.70 (m, 2H), 1.06 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.56, 22.82, 31.41, 55.84, 112.00, 120.35, 127.41, 129.65, 133.37, 157.59, 191.42. HRMS (EI): m/z [M + H]+ calcd for C11H15O2S: 211.0793; found: 211.0790.
S-propyl (1,1’-biphenyl)-4-carbothioate (3jb ):
Yield: 118 mg (46%); yellow solid. 1H NMR (400 MHz, CDCl3): δ 8.08 (d, J = 8.0 Hz, 2H), 7.70-7.64 (m, 4H), 7.51-7.42 (m, 3H), 3.11 (t, J = 8.0 Hz, 2H), 1.78-1.73 (m, 2H), 1.08 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.47, 23.03, 30.97, 127.21, 127.27, 127.75, 128.23, 128.96, 136.02, 139.89, 145.98, 191.67. HRMS (EI): m/z [M + H]+ calcd for C16H17OS: 257.1000; found: 257.0997.
S-propyl thiophene-3-carbothioate (3kb ):
Yield: 78 mg (46%); yellow liquid. 1H NMR (400 MHz, CDCl3): δ 8.13-8.12 (m, 1H), 7.57-7.55 (m, 1H), 7.36-7.28 (m, 1H), 3.06 (t, J = 8.0 Hz, 2H), 1.75-1.69 (m, 2H), 1.05 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.42, 23.08, 30.76, 126.04, 126.32, 130.26, 141.18, 185.77. HRMS (EI): m/z [M + H]+ calcd for C8H11OS2: 187.0251; found: 187.0250.
S-(2-fluoroethyl) 4-methoxybenzothioate (3ec ):
Yield: 84 mg (39%); yellow liquid. 1H NMR (400 MHz, CDCl3): δ 7.99 (d, J = 8.0 Hz, 2H), 6.97 (d, J = 8.0 Hz, 2H), 4.67 (t, J = 8.0 Hz, 1H), 4.55 (t, J = 8.0 Hz, 1H), 3.90 (s, 3H), 3.44-3.36 (m, 2H). 13C NMR (101 MHz, CDCl3): δ 28.71, 28.93, 55.55, 82.87 (d JC-F = 170 Hz), 113.88, 129.46, 129.55, 164.04, 189.32. HRMS (EI): m/z [M + H]+ calcd for C10H12FO2S: 215.0542; found: 215.0537.
S-(furan-2-ylmethyl) 4-methoxybenzothioate (3ed ):
Yield: 139 mg (56%); yellow liquid. 1H NMR (400 MHz, CDCl3): δ 7.98 (d, J = 8.0 Hz, 2H), 7.37 (d, J = 8.0 Hz, 1H), 6.96-6.93 (m, 2H), 6.34-6.31 (m, 2H), 4.36 (s, 2H), 3.89 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 25.63, 55.54, 108.01, 110.65, 113.83, 129.54, 142.23, 150.73, 163.92, 189.20. HRMS (EI): m/z [M + H]+ calcd for C13H13O3S: 249.0585; found: 249.0583.
S-(2-methylfuran-3-yl) 4-methoxybenzothioate (3ee ):
Yield: 112 mg (45%); yellowish liquid. 1H NMR (400 MHz, CDCl3): δ 8.04 (d, J = 8.0 Hz, 2H), 7.44 (d, J = 4.0 Hz, 1H), 6.99 (d, J = 8.0 Hz, 2H), 6.43 (d, J = 4.0 Hz, 1H), 3.91 (s, 3H), 2.33 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 12.02, 55.56, 103.66, 113.93, 115.28, 129.29, 129.76, 141.09, 156.68, 164.00, 188.01. HRMS (EI): m/z [M + H]+ calcd for C13H13O3S: 249.0585; found: 249.0580.
S-butyl 3,5-dimethylbenzothioate (3lf ):
Yield: 140 mg (63%); colorless liquid. 1H NMR (400 MHz, CDCl3): δ 7.60 (s, 2H), 7.21 (s, 1H), 3.08 (t, J = 8.0 Hz, 2H), 2.39 (s, 6H), 1.67-1.64 (m, 2H), 1.51-1.45 (m, 2H), 0.97 (t, J = 8.0 Hz, 3H). 13C NMR (101 MHz, CDCl3): δ 13.63, 21.19, 22.05, 28.70, 31.64, 124.92, 134.85, 137.37, 138.27, 192.45. HRMS (EI): m/z [M + H]+ calcd for C13H19OS: 223.1157; found: 223.1155.
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211028114 – Supplemental material for Nickel-catalyzed carbonylation of thioacetates with aryl iodides via CO insertion and C–S bond cleavage
Supplemental material, sj-pdf-1-chl-10.1177_17475198211028114 for Nickel-catalyzed carbonylation of thioacetates with aryl iodides via CO insertion and C–S bond cleavage by Wen-Peng Mai, Hong-Da Sui, Ming-Xiu Lv and Kui Lu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors greatly acknowledge financial support from the Henan University of Engineering for the Youth Doctoral Funding (no. 2015017) and the Henan Province Key Research and Development and Promotion Special (212102310258).
Supplemental material
Supplemental material for this article is available online.