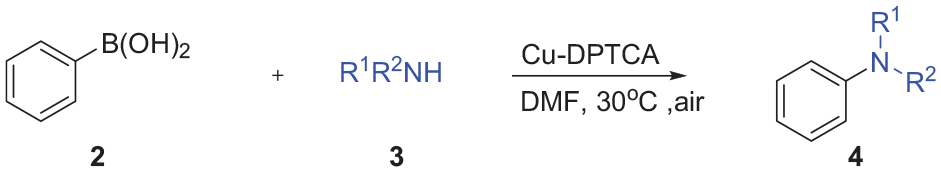
Abstract
A new copper(II) metal-organic framework is constructed as a sustainable copper heterogeneous catalyst. Cu-DPTCA, with high catalytic activity, can effectively promote the Chan–Lam coupling reaction of arylboronic acids and amines without adding any base or additive.

Introduction
Reactions that form C–N bonds are widely used in synthetic chemistry due to the prevalence of this functionality within pharmaceuticals and biologically active natural products.1–4 In recent decades, various effective strategies based on transition-metal catalysis for C–N bond formation have been developed, such as Pd-catalyzed Buchwald–Hartwig coupling5,6 and Ullmann coupling. 7 However, these cases mostly involve the expensive transition-metal palladium and harsh reaction conditions (over 100 °C), which restrict their wide applications in industry. However, the Chan–Lam cross-coupling reaction, the oxidative coupling of amines and arylboronic acids catalyzed by Cu salts, represents one of the most powerful and straightforward tools to construct C–N bonds.8–13 This process is mild, convenient, and cheap, offering an alternative to complementary methods using noble metals. So far, various homogeneous Cu catalysts have been used for the Chan–Lam reaction to access N-arylamine subunits. Nevertheless, the use of an additional base in a stoichiometric amount is required for regeneration of the active Cu species. Furthermore, homogeneous catalytic systems are generally affected by the separation of the products and the metal catalysts, which can be problematic and costly.
In recent years, metal-organic frameworks (MOFs) have emerged as a distinct new option for heterogenization of a metal catalyst for organic synthesis.14–19 In comparison with other existing heterogeneous catalytic systems, the design flexibility and framework tunability resulting from the huge variations of metal nodes and organic linkers allow the introduction of different compositions in a single MOF.20–25 With this in mind, we anticipated that a MOF containing an amine motif in the organic linker and Cu ion nodes should have two benefits. It may eliminate the need for an additional base, and it may facilitate the recovery and separation of the Cu metal catalyst from products, thereby reducing economic and environmental barriers. To achieve this objective, herein we report the synthesis of a new Cu-DPTCA MOF based on diphenylamine tetracarboxylic acid (DPTCA) and Cu metal (Figure 1, left), named [Cu2.5(DPTCA)(OH)(dma)2(H2O)]·2DMF (abbreviated as Cu-DPTCA), (dma = dimethylamine, DMF = N,N-dimethyl-formamide). With a diphenylamine acid as the linker, DPTCA forms a quad-core structure with a high surface area and large pore volume, providing open metal sites accessible in the catalytic process (Figure 1, right). The MOF was characterized by single-crystal and powder X-ray diffraction, infrared spectroscopy, and thermogravimetric analysis (TGA).

DPTCA ligands are presented and assembled with CuII, forming a Cu-based framework.
Results and discussion
The asymmetric unit of Cu-DPTCA contains two and a half CuII ions, one DPTCA ligand molecule, one μ3-hydroxyl group, two coordinated dimethylamines, one coordinated water molecule, and two free DMF molecules. Cu-DPTCA features a non-interpenetrated open three-dimensional (3D) porous framework constructed from a tetranuclear Cu(II) cluster [Cu4(CO2)6(μ3–OH)2] and DPTCA (Figure 1). Six carboxylate groups from six DPTCA ligands and two μ3-hydroxy oxygen atoms link four adjacent Cu(II) ions into tetranuclear clusters. Each DPTCA ligand links four tetranuclear Cu(II) clusters, resulting in the formation of a 3D porous framework. The effective free volume of Cu-DPTCA is calculated by PLATON analysis as 17.1% of the crystal volume including the free DMF molecules. When viewed from the a-axis, Cu-DPTCA has large triangular channels. The structure contains Cu4 cluster units (Figure 2). The crystallographic data of Cu-DPTCA are summarized in Table 1.

(a) The asymmetric unit of Cu-DPTCA, (b) the tetranuclear Cu cluster in Cu-DPTCA, and (c) the cavity in Cu-DPTCA.
Crystallographic data and experimental details.

The thermal stability was verified by TGA. A differential thermal weight loss experiment was performed on Cu-DPTCA under an N2 atmosphere. The temperature range was 30–805 °C, and the temperature ramp rate was 10 °C min−1. The TGA curve (Figure 3) shows that the first mass loss (15%) occurred between 30 and 175 °C due to the loss of coordinated H2O molecules (theoretical value is 11.3%). Between 175 and 300 °C, it can be seen that the frame structure collapsed and was completely dissociated by 500 °C. The above experimental results show that Cu-DPTCA retains its frame structure and is stable below 300 °C.
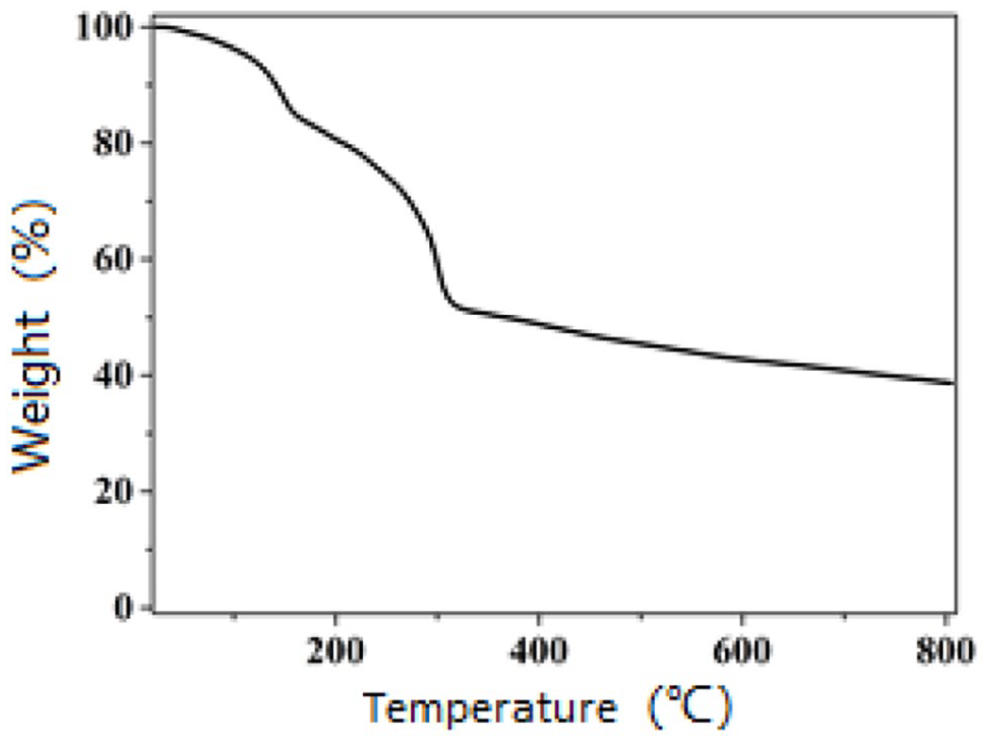
TGA curve of Cu-DPTCA.
To further investigate the porosity of Cu-DPTCA, substrate adsorption experiments were performed with phenylboronic acid (
At the outset of the investigation, we were interested in exploring the catalytic potential of Cu-DPTCA for the Chan–Lam coupling reaction. Before catalytic reactions, pre-treatment is required to activate Cu-DPTCA. Thus, the Cu-DPTCA catalyst was immersed in an acetonitrile solution for 24 h for guest molecular exchange and then dried in a vacuum oven (100 °C, 2 h) to remove acetonitrile molecules and solvent molecules inside the Cu-DPTCA catalyst.
Next, phenylboronic acid (
Optimization of the reaction conditions. a

Reaction conditions: amine (1.5 mmol), arylboronic acid (1 mmol), catalyst Cu-DPTCA (8 μmol based on a paddlewheel unit), DMF (4 mL), and air.
Under the optimized conditions, the substrate scope of the amines was further investigated, and a number of structurally different amines were selected. Primary aliphatic amines, such as
Reaction conditions: amine (1.5 mmol), arylboronic acid (1 mmol), catalyst Cu-DPTCA (8 μmol based on a paddlewheel unit), DMF (4 mL), rt, and air.
Isolated yields.
The advantage of the Cu-DPTCA catalyst over its homogeneous counterparts lies in its facile recovery and reusability. To this end, the Cu-DPTCA catalyst was recovered and used under the optimized conditions for C–N coupling of phenylboronic acid (
Conclusion
In the present work, a new Cu(II)-carboxylate-based MOF [Cu2.5(DPTCA)(dma)2(DMF)2(H2O)]n has been synthesized by a solvothermal method and characterized by single-crystal and powder X-ray diffraction, infrared spectroscopy, and TGA. Cu-DPTCA has good triangular channels, and the presence of coordinated, unsaturated open metal sites allows it to be used for the Chan–Lam coupling reaction, affording various amine products. It is worth mentioning that this eco-friendly process does not require an additional basic additive and the Cu-DPTCA can be easily recovered from the product, and thus the sustainability is significantly improved.
Experimental
Materials and measurements
All solvents and chemical materials used in the synthesis were purchased from commercial sources and were used without further purification. X-ray powder diffraction (PXRD) was recorded with CuKα radiation (λ = 1.54056 Å) using a Bruker AXS D8 advanced diffractometer at an angle of 2293 = 5–50° at 293 K.
Crystallography
The single-crystal structure was obtained using Bruker d8 Venture SMART-CCD single-crystal diffractometer with MoKα radiation, wavelength (λ) = 0.71073 Å. Diffraction data were collected using the SAMRT and SAINT programs. The test temperature was maintained near 200 K with liquid nitrogen. The crystal structure was solved by direct methods using SHELXTL-97. The full-matrix least-square method was used to refine F2 (CCDC: 2047623).
Synthesis of Cu-DPTCA
DPTCA (13.8 mg, 0.04 mmol) and copper nitrate hexahydrate (22.0 mg, 0.12 mmol) were placed in a 10-mL glass bottle, and DMF (4 mL) and distilled water (0.5 mL) were added. The resulting mixture was sonicated for 30 min. The bottle was placed in a 25-mL polytetrafluoroethylene-lined autoclave, sealed and placed in a program-controlled oven. The oven was heated to 115 °C for 5.5 h and held for a further 70 h. The oven was cooled to room temperature over 15 h and colorless transparent square crystals were obtained with the chemical formula C26H35Cu2.5N5O11. The crystals were filtered and washed with the mother liquor. The yield was 52%.
Cham–Lam reaction
N-arylation of phenylboronic acid and amine, in a typical experimental procedure, Cu-DPTCA (8 μmol) was added to the amine (1.5 mmol) and phenylboronic acid (1 mmol) in DMF (4 mL) at 30 °C, and the mixture was stirred at room temperature for 12 h under an air atmosphere. The progress of the reaction was monitored by TLC, and when the reaction was completed, the mixture was filtered to obtain a crude product. The crude product was purified by silica gel column chromatography (petroleum ether–EtOAc, 10: 1) to give coupling products
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211026506 – Supplemental material for A base-free Chan–Lam reaction catalyzed by an easily assembled Cu(II)-carboxylate metal-organic framework
Supplemental material, sj-pdf-1-chl-10.1177_17475198211026506 for A base-free Chan–Lam reaction catalyzed by an easily assembled Cu(II)-carboxylate metal-organic framework by Xinhai Zhang, Jianhua Qin, Ruixuan Ma and Lei Shi in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Project of Science and Technology of Henan Province (no. 202102210233) and the Open Research Fund of the School of Chemistry and Chemical Engineering, Henan Normal University (no. 2020YB03).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.