Abstract
A highly efficient heterogeneous palladium-catalyzed transfer semihydrogenation of internal alkynes was achieved in dimethylformamide at 145 ºC by using an MCM-41-immobilized bidentate nitrogen palladium(II) complex [MCM-41-2N-Pd(OAc)2] as catalyst and dimethylformamide/KOH as hydrogen source, yielding a variety of cis-alkenes in good to high yields with excellent stereoselectivity. This supported palladium catalyst can be easily recovered by filtration of the reaction solution and recycled up to seven times with almost consistent activity.

Introduction
Alkenes are highly useful building blocks in organic synthesis because of their versatile further functionalization. They are commonly used as substrates in a variety of organic reactions including Markovnikoff’s addition, epoxidation, ozonolysis, hydroboration, hydrogenation, hydroformylation reactions, and so on.1–3 This diversity in application makes them important intermediates in the synthesis of drug molecules, natural products, and biologically active compounds.4–6 In addition, well-defined (E)- or (Z)-alkenes constitute an integral part of many biologically active molecules. 4 As a result, many synthetic methods such as the Wittig reaction, 7 Horner–Emmons–Wadsworth reaction, 8 Peterson reaction, 9 Julia–Kocienski reaction, 10 Takai olefination, 11 olefin metathesis, 12 and cross-coupling reactions 13 have been developed to construct C–C double bond. Besides, the reduction of alkynes is an attractive alternative route to alkenes due to its high atom economy.4,14
The selective semihydrogenation of internal alkynes to cis-alkenes is a highly useful reaction in organic synthesis, and there are mainly two types of catalytic processes to complete this transformation. One process involves hydrogenation using hydrogen gas (H2) in the presence of Lindlar’s catalyst,4,15 or other heterogeneous palladium catalysts.16–18 The other is transfer hydrogenation with hydrogen donors. 19 The latter has obvious advantages such as safer operation and easy control of chemoselectivity of alkenes. Recently, transition metal-catalyzed transfer semihydrogenation of internal alkynes has been shown to be a highly efficient method for the synthesis of cis-alkenes and various transition metal catalysts such as palladium,20–24 ruthenium,25,26 gold, 27 nickel, 28 and copper 29 based catalytic systems have been developed for this transformation. However, the use of expensive palladium, ruthenium, and gold as well as difficult recovery and non-recyclability of the metal catalysts make these methods of limited synthetic utility from economic and environmental points of view. Catalyst recycle is one of the most important features for many green synthetic methods. The heterogenization of the existing homogeneous metal catalysts appears to be a logical solution to this problem. There has been considerable interest in the development of heterogeneous transition metal catalytic systems that can be facilely recycled for several times with almost consistent activity. However, to the best of our knowledge, no examples of supported palladium complexes–catalyzed transfer semihydrogenation of internal alkynes to cis-alkenes have been described until now, despite the practical benefits of heterogeneous catalysis.
Mesoporous MCM-41 materials have recently emerged as powerful supports for immobilization of homogeneous catalysts owing to their outstanding properties.30–32 Recently, we reported the synthesis of MCM-41-immobilized bidentate nitrogen palladium(II) complex [MCM-41-2N-Pd(OAc)2] and found that it was a highly efficient and recyclable catalyst for the Suzuki–Miyaura reaction of aryl bromides, 33 the cross-coupling of acyl chlorides with terminal alkynes, 34 and the carbonylative cross-coupling of triarylbismuths with aryl iodides. 35 In continuation of our effort to develop economical and eco-friendly synthetic pathways for organic transformations,33–35 herein, we reported a heterogeneous palladium-catalyzed transfer semihydrogenation of internal alkynes by using the MCM-41-2N-Pd(OAc)2 complex as catalyst with dimethylformamide (DMF)/KOH as hydrogen source to provide cis-alkenes in good to high yields with excellent stereoselectivity (Scheme 1).
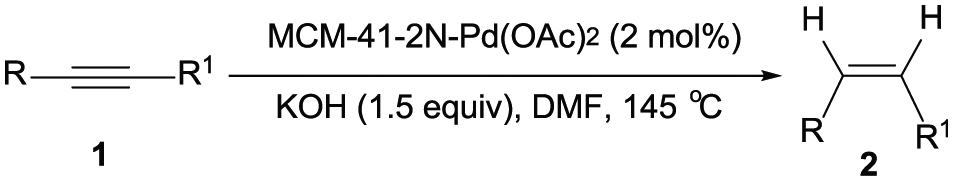
Heterogeneous palladium-catalyzed transfer semihydrogenation of internal alkynes.
Results and discussion
The MCM-41-immobilized bidentate nitrogen palladium(II) complex [MCM-41-2N-Pd(OAc)2] could be conveniently prepared via a simple two-step procedure as shown in Scheme 2. 34 First, the mesoporous MCM-41 was condensed with commercially readily available and inexpensive 3-(2-aminoethylamino)propyltrimethoxysilane in toluene under reflux for 24 h, followed by the silylation with Me3SiCl at room temperature for 24 h to produce the 3-(2-aminoethylamino)propyl-functionalized MCM-41 (MCM-41-2N). Then the latter was reacted with Pd(OAc)2 in acetone under reflux for 72 h to afford the MCM-41-immobilized bidentate nitrogen palladium(II) complex [MCM-41-2N-Pd(OAc)2] as a pale yellow powder. The palladium content of this heterogeneous palladium complex was determined to be 0.29 mmol g−1 by inductively coupled plasma atomic emission spectroscopy (ICP-AES) analysis.
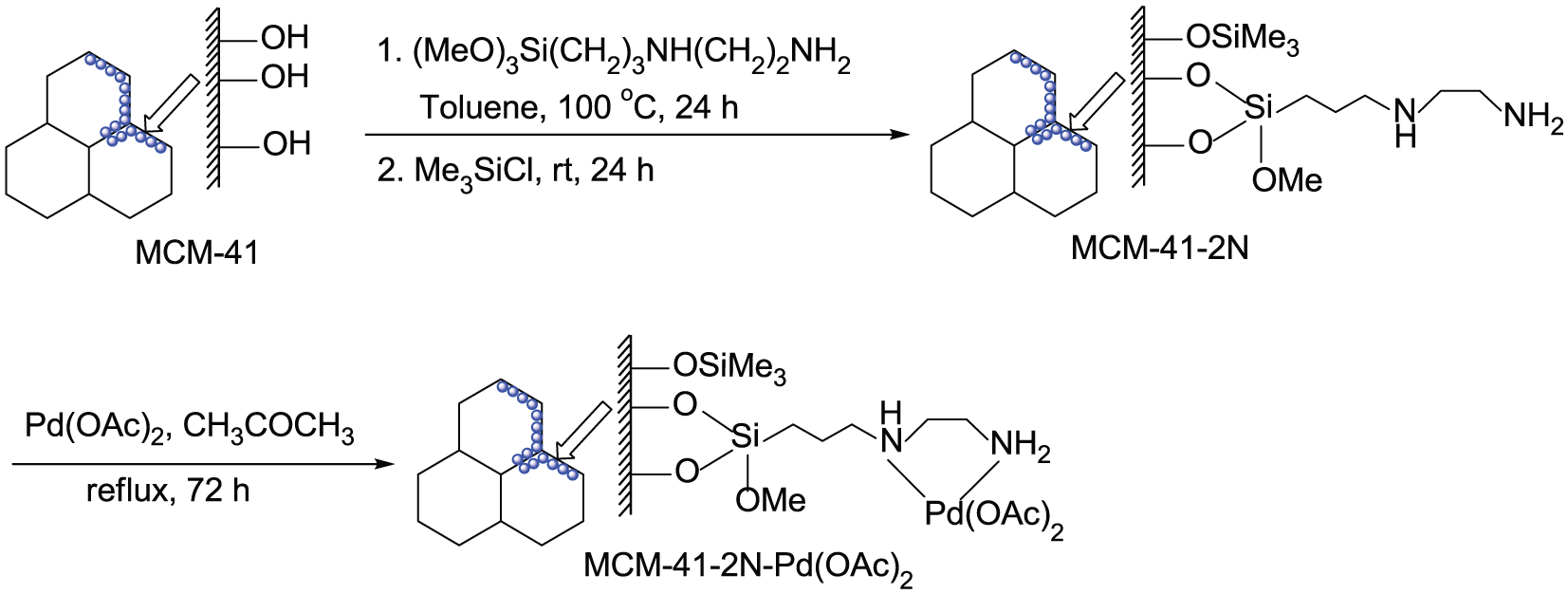
Preparation of MCM-41-2N-Pd(OAc)2.
In continuation of our research to employ this heterogeneous palladium system for other organic transformations, we commenced our studies by investigating the transfer semihydrogenation of 1,2-diphenylacetylene
Heterogeneous Pd-catalyzed semireduction of 1,2-diphenylacetylene

GC: gas chromatography; DMF: dimethylformamide.
Reaction conditions:
The number in parentheses is isolated yield.
Determined by GC analysis of reaction mixture.
The reaction was carried out for 24 h.
The reaction was performed at 100 ºC for 24 h.
The reaction was carried out under air.
The reaction was performed for 12 h.
The cis-stilbene skeleton is found in many natural products that exhibit a variety of biological activities,
38
thus, development of a general, efficient, and practical method for construction of cis-stilbenes by heterogeneous palladium-catalyzed transfer semihydrogenation of diarylacetylenes with high stereoselectivity is highly desirable. With the optimized conditions in hand, we next investigated heterogeneous palladium-catalyzed semireduction of a variety of internal alkynes and the results are summarized in Table 2. In general, all the reactions proceeded smoothly under the standard reaction conditions to afford the cis-alkenes in good to high yields with excellent stereoselectivity. Diarylacetylenes bearing various substituents
Heterogeneous Pd-catalyzed semireduction of internal alkynes. a

DMF: dimethylformamide; GC: gas chromatography.
Reaction conditions:
Isolated yield.
Determined by GC analysis of the reaction mixture.
For 9 h.
To verify whether the observed catalysis was due to the heterogeneous catalyst MCM-41-2N-Pd(OAc)2 or to a leached palladium species in solution, we conducted the hot filtration test.
39
We focused on the semihydrogenation of diphenylacetylene
A plausible mechanism for this heterogeneous palladium-catalyzed transfer semihydrogenation of internal alkynes is illustrated in Scheme 3.
23
First, oxidative addition of HCOOH, generated in situ by the hydrolysis of DMF in the presence of KOH, to MCM-41-2N-Pd(0) provides an MCM-41-anchored HCOO-Pd(II)-H complex intermediate

Proposed catalytic cycle.
For a supported precious metal catalyst, it is important to evaluate its ease of separation, recoverability, and reusability. We next investigated the recyclability of MCM-41-2N-Pd(OAc)2 by using the transfer semihydrogenation reaction of diphenylacetylene
In conclusion, we have developed an efficient and practical heterogeneous palladium-catalyzed transfer semihydrogenation of internal alkynes leading to cis-alkenes by using an MCM-41-supported bidentate nitrogen palladium(II) complex [MCM-41-2N-Pd(OAc)2] as catalyst and DMF/KOH as the hydrogen source. The reactions generated a wide variety of cis-alkenes in good to excellent yields with excellent stereoselectivity and were applicable to a range of diarylacetylenes and alkyl-substituted internal alkynes. Importantly, this heterogeneous palladium catalyst can be easily prepared via a simple procedure from commercially readily available reagents, and recovered by filtration of the reaction solution and recycled up to seven times with almost consistent activity, thus making this approach economically and environmentally more acceptable.
Experimental
All reagents were analytical grade and used as received without further purification. The MCM-41-immobilized bidentate nitrogen palladium(II) complex [MCM-41-2N-Pd(OAc)2] was prepared according to our previous method, 34 the palladium content was determined to be 0.29 mmol g−1 by ICP-AES analysis. DMF was chemical pure and used as received without drying. All reactions were carried out under Ar in oven-dried glassware with magnetic stirring. The products were purified by flash chromatography on silica gel using cyclohexane as eluent. All products were characterized by comparison of their spectra and physical data with authentic samples. The gas chromatography (GC) analysis of the reaction mixture was performed using Perkin Elmer Clarus 400 GC equipped with flame ionization detector with a capillary column and n-octadecane was used as an internal standard. 1H NMR spectra were recorded on a Bruker Avance 400 (400 MHz) spectrometer with tetramethylsilane (TMS) as an internal standard using CDCl3 as the solvent. 13C NMR spectra were recorded on a Bruker Avance 400 (100 MHz) spectrometer using CDCl3 as the solvent. Palladium content was determined with inductively coupled plasma atom emission Atomscan16 (ICP-AES, TJA Corporation).
General procedure for the heterogeneous palladium-catalyzed transfer semihydrogenation of internal alkynes
Under an argon atmosphere, a thick-walled reaction tube was charged with KOH (84 mg, 1.5 mmol) and DMF (1.5 mL). The mixture was stirred at 145 °C under Ar for 0.5 h. After being cooled to room temperature, MCM-41-2N-Pd(OAc)2 (69 mg, 0.02 mmol) and internal alkyne (1.0 mmol) were added to the tube and the resulting mixture was stirred at 145 °C under Ar for 6–9 h. After completion of the reaction, the reaction mixture was cooled to room temperature, diluted with ethyl acetate (15 mL), and filtered. The MCM-41-2N-Pd(OAc)2 catalyst was washed with distilled water (2 × 5 mL) and acetone (2 × 5 mL), and reused in the next run. The filtrate was concentrated under reduced pressure and the residue was purified by flash column chromatography on silica gel (eluent: cyclohexane) to provide the desired product
Cautionary note. the base-induced thermal decomposition of DMF with base generates CO2 and Me2NH, both gases at the temperatures involved. It is likely that significant pressure build up would occur at larger scales of reaction.
cis-Stilbene (
cis-4,4′-Dimethylstilbene (
cis-4-Methyl-4′-methoxystilbene (
cis-1-(4-Methoxyphenyl)-2-phenylethene (
cis-1-(2-Methoxyphenyl)-2-phenylethene (
cis-1-(4-Fluorophenyl)-2-phenylethene (
cis-1-(4-Chlorophenyl)-2-phenylethene (
cis-1-Phenyl-2-(2-trifluoromethylphenyl)ethene (
4-(cis-Styryl)biphenyl (
cis-1-(1-Naphthyl)-2-phenylethene (
cis-1-(2-Naphthyl)-2-phenylethene (
cis-2-Styrylthiophene (
cis-Pent-1-ene-1,5-diyldibenzene (
cis-1-Ethyl-2-phenylethene (
cis-1-Phenyl-1-hexene (
cis-Oct-1-en-1-ylbenzene (
cis-1-(2-Cyclopropylvinyl)naphthalene (
cis-Decene (
Footnotes
Acknowledgements
The authors thank the National Natural Science Foundation of China (No. 21462021), the Natural Science Foundation of Jiangxi Province of China (No. 20161BAB203086), and Key Laboratory of Functional Small Organic Molecule, Ministry of Education (No. KLFS-KF-201704) for financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


