Abstract
We report a direct cross-coupling reaction between (het)aryl pivalates/tosylates and di(het)arylzinc species in 2-methyltetrahydrofuran/N-methyl pyrrolidone (1:1), which occurs via C–O bond cleavage under microwave irradiation. The reaction takes place smoothly in short reaction times without the addition of any catalyst or ligand. The reaction is suitable for a broad scope of substrates and exhibits good functional group compatibility, utilizes a simple work-up procedure, and gives the desired products in high purity.

Introduction
Biaryls have been widely applied in the syntheses of natural products, polyaromatic molecules, and pharmaceuticals.1–3 Transition-metal (TM) catalyzed cross-coupling reactions are very powerful tools for constructing such structural units. However, various issues remain, including high costs and the poor stability of many TM catalysts and ligands, and the necessity of disposal of heavy-metal residues. Transition-metal-free (TM-free) cross-coupling methods have attracted significant attention in recent years.4–12 Examples include the reactions of organohalides (R-Hal, including R-I, R-Br, and unreactive R-Cl) with aryl Grignard,13,14 arylzinc,15,16 organoaluminum 17 reagents, and among others.5,18 Different from classical cross-coupling reactions, most TM-free couplings occur as single-electron transfer (SET) processes, and can be regarded as one-electron-catalyzed cross-coupling reactions. 19 The key process in the SET catalytic cycle is the formation of the radical anion R-Hal•−, which undergoes further propagation (with R-M) and electron exchange (with another R-Hal) to form the product and to regenerate itself. 20 The SET mechanisms have been clarified by means of both experimental 21 and theoretical 22 methods.
Phenol derivatives have some advantages: (1) phenol derivatives are easily available and are less expensive than the corresponding halides; (2) the use of halides, which pollute the environment is avoided; and (3) phenol derivatives can exhibit orthogonal reactivity to organohalides. 23 There are many known methods for cross-coupling phenols derivatives. 24 However, no examples utilize simple phenol derivatives (e.g. pivalate esters, sulfonates, carbamates, and sulfamates) for cross-coupling by SET processes. Organozinc reagents have emerged as attractive candidates due to their easy preparation and high functional group tolerance in cross-coupling reactions for the construction of biaryl and aryl–vinyl structural scaffolds.25,26 To the best of our knowledge, phenol derivatives have never been used in couplings with organozinc reagents by SET-catalyzed cross-coupling. Herein, we report the first cross-coupling reactions of phenol derivatives with arylzinc reagents to construct C–C bonds under SET-catalyzed cross-coupling reaction conditions.
Results and discussion
We started our investigation by utilizing phenyl pivalate ester
Optimization of the reaction conditions. a

MW: microwave.
Conditions:
∆ conventional heating.
GC yield using tridecane as an internal standard; isolated yield is given in parentheses.
See Chen et al. 13
No matter if 1 or 2 equiv. of the arylmagnesium bromide is mixed with 1 equiv. of zinc bromide (Table 1, entries 2 and 3), the response was very poor. The reaction yield was improved slightly by adding 1 equiv. of lithium chloride (Table 1, entries 4 and 5); however, the reaction proceeded sparingly or not at all when lithium bromide or magnesium bromide were added (Table 1, entries 6 and 7). The yield reached 46% using 2 equiv. of lithium chloride (Table 1, entry 8), but the yield did not increase significantly when 3 equiv. of lithium chloride were added (Table 1, entry 9).
Under the same conditions as those in entry 9, microwave (MW) heating led to a yield of 68% (Table 1, entry 10). Actually, the arylzinc iodide/lithium chloride complex reported by Knochel 27 gave lower yields of the product, even in the presence of an external magnesium salt (Table 1, entries 11 and 12). The use of N,N-dimethylacetamide (DMA), iPr2O, and 1,4-dioxane as solvents were demonstrated to be of no effect (Table 1, entries 13, 15, and 16). When N,N-dimethylformamide (DMF) was used, a yield of 5% was obtained (Table 1, entry 17). The use of 1-methylpyrrolidin-2-one (NMP), 2-Me-Tetrahydrofuran (THF), diglyme, and toluene led to the desired product, and 2-Me-THF gave the highest yield (Table 1, entries 14 and 18–20). Mixtures (1:1) of 2-Me-THF/toluene, 2-Me-THF/NMP, or 2-Me-THF/diglyme behaved better than a single solvent, with 2-Me-THF/NMP (1:1) leading to the highest yield (Table 1, entries 21–23). The reaction yield did not increase on extending the reaction time, with only 0.5 h being required to obtain the highest yield (Table 1, entries 24–26). When the reaction was run at 80 °C, a 93% yield was achieved. A reaction temperature higher than 80 °C did not further improve the yield, while a temperature lower than 80 °C led to a decrease in the yield (Table 1, entries 25, 27, and 28). However, conventional heating led, under the same conditions, to a yield of only 25% (Table 1, entry 29).
With promising results in hand, we next tested the analogous cross-coupling of several other electrophilic partners (Table 2). In addition to the aryl pivalate ester (Table 2, entry 1), the corresponding carbamate and sulfamate were deemed competent substrates (Table 2, entries 2 and 3). Furthermore, sulfonate derivatives of phenol also gave high yields of the coupled product (Table 2, entries 4–6), and aryl sulfonates (in particular, tosylates) were relatively unreactive compared to triflates; however, tosylates were more easily handled, stable and considerably less expensive than aryl triflates. Moreover, the use of a phenyl methyl ether did not lead to the desired product under our optimized conditions (Table 2, entry 7).
Survey of cross-coupling partners. a

Conditions:
GC yield using tridecane as an internal standard.
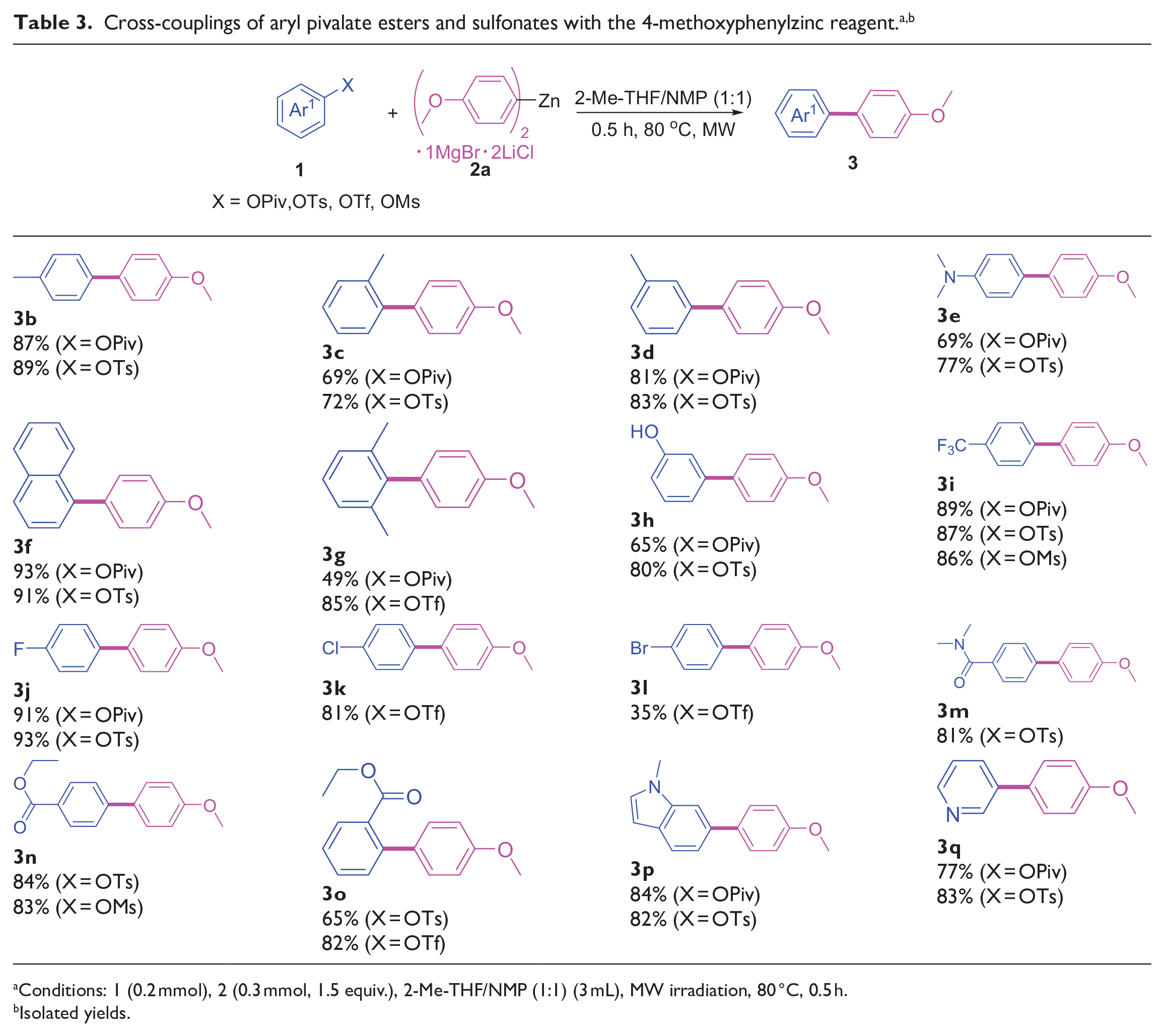
Having identified optimized reaction conditions (Table 1, entry 25), we next investigated the scope of the substrates (Table 3). Aryl pivalates/tosylates containing electron-donating groups were efficiently coupled to provide the corresponding biaryl products in good to excellent yields (Table 3, entries
Conditions: 1 (0.2 mmol), 2 (0.3 mmol, 1.5 equiv.), 2-Me-THF/NMP (1:1) (3 mL), MW irradiation, 80 °C, 0.5 h.
Isolated yields.
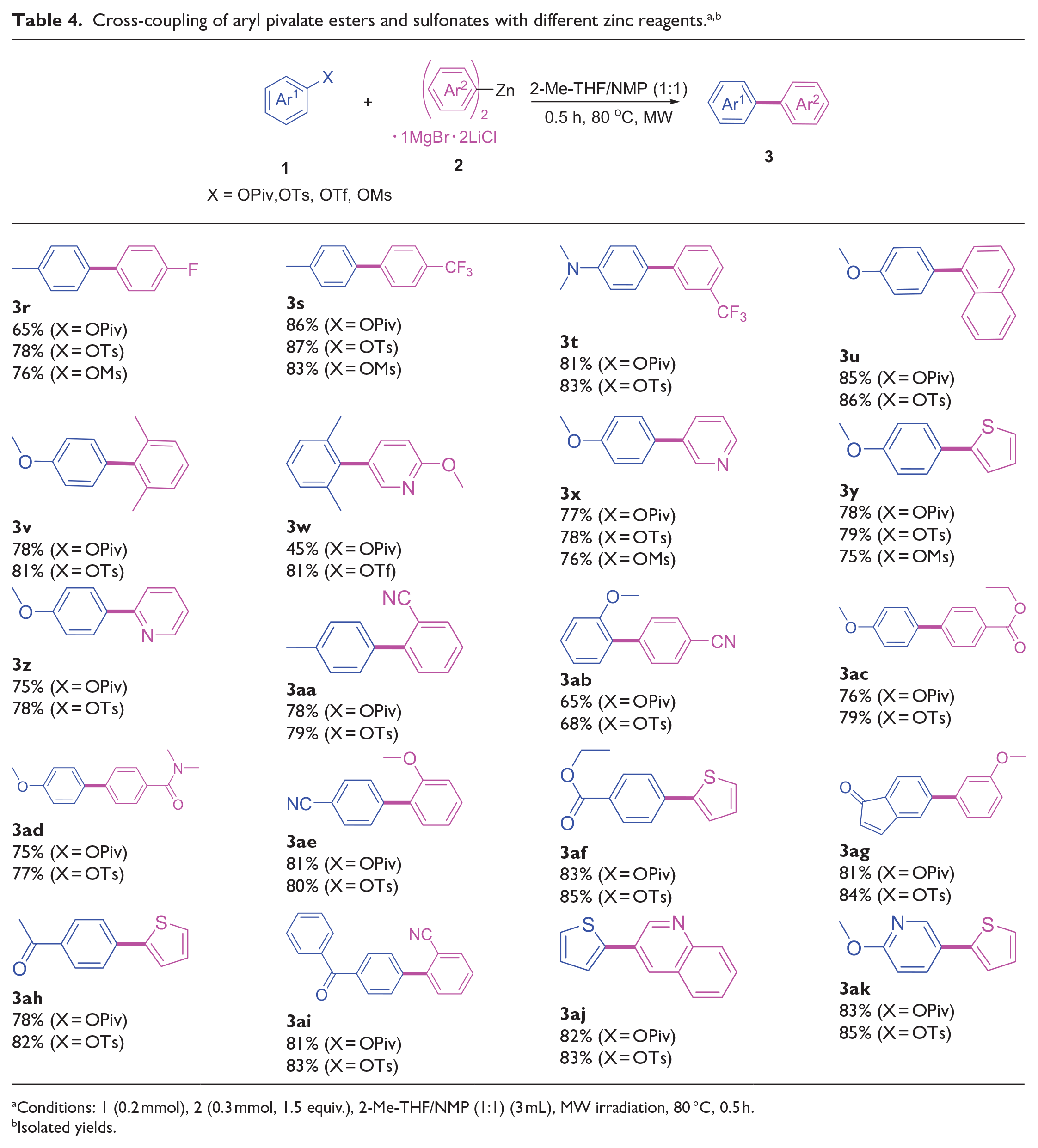
We further inspected the reactivity of different zinc reagents (Table 4). Both electron-rich and electron-deficient (het)aryl zinc reagents (Table 4, entries
Conditions: 1 (0.2 mmol), 2 (0.3 mmol, 1.5 equiv.), 2-Me-THF/NMP (1:1) (3 mL), MW irradiation, 80 °C, 0.5 h.
Isolated yields.
The SET-catalyzed cross-coupling reactions are more sensitive to steric hindrance. However, the promising results obtained for para- and meta-substituted aryl pivalates/tosylates render this system a new and useful tool in SET-catalyzed cross-coupling reactions. For instance, under these conditions, the selective one-pot synthesis of disubstituted phenol sulfonates gave unsymmetrically substituted terphenyl compounds in satisfactory yields (Scheme 1).

One-pot synthesis of an unsymmetric terphenyl compound.
In Hayashi’s report,
28
the occurrence of SET initiation in the coupling of aryl Grignard reagents with aryl halides was confirmed by the observation that the addition of lithium 4,4-di-tert-butylbiphenylide (LDBB) drastically accelerated the coupling. This result may be rationally understood by inferring that LDBB works as a much more efficient single-electron donor than Grignard reagents in the slow initiation step, and thus, the overall reaction rate is increased. We conducted similar experiments on the arylzinc coupling using LDBB as a single-electron donor. The reactivity of PhOPiv (

Effect of the addition of single-electron donors.
Considering the above result in conjunction with similarities in the intrinsic character between arylzinc and arylmagnesium reagents, the present coupling reaction likely follows a Grignard cross-coupling mechanism2,4,32–34 as shown in Scheme 3, and exemplified by the reaction of PhOPiv

A plausible mechanism.
Conclusion
The reactions of (het)aryl pivalates/tosylates with di(het)arylzincs gave biaryl compounds in short reaction times under microwave irradiation. The reaction is suitable for a broad scope of substrates and exhibits good functional group compatibility. This reaction is applicable to simple hydroxybenzenes that are widely distributed in nature. No catalyst or ligand is required, and a simple work-up procedure is employed. The reaction is believed to occur via a SET mechanism for activation of the aryl pivalates.
Experimental
General information
The reactions were carried out at 50–80 W in a CEM Discover (0–600 W, 2450M) focused microwave reactor equipped with a pressure controller under isothermal conditions. Standard 5 mL glass reaction vessels were used as supplied with the CEM reactor. The reaction mixtures were stirred magnetically. LiCl, ZnBr2, ZnCl2, and LiBr were purchased from Aldrich. MgBr2 was purchased from Alfa Aesar. Other reagents are available commercially and were used without further purification, unless otherwise indicated. All reactions were carried out under an argon atmosphere with dry solvents under anhydrous conditions, unless otherwise noted. THF was dried over alumina under N2 using a Grubbs-type solvent purification system. All arylzinc reagents were prepared from the corresponding arylmagnesium bromides, LiCl and ZnBr2. All aromatic phenols were purchased from Alfa Aesar. Spectroscopic data for known compounds match with the data reported in the corresponding references. Reactions were monitored with Agilent GC Series 6890N and instruments GCMS 7890A. All compounds were characterized by 1H NMR spectroscopy using a Bruker 400 M spectrometer (Bruker Avance III 400 MHz NMR). New compounds are characterized by HRMS (TripleTOF™ 5600+). 1H NMR was recorded in CDCl3 using tetramethylsilane (TMS) and deuterium oxide as the internal standard. Chemical shifts were reported in parts per million. The signal patterns are indicated as follows: s, singlet; d, doublet; t, triplet; dd, doublet of doublets; dt, doublet of triplets; and m, multiplet. Coupling constants, J, are reported in Hertz (Hz). The products were purified by column chromatography on Aladdin silica gel 300–400 mesh under an argon atmosphere.
Experimental procedures
General procedure for the reaction between (het)aryl pivalates
(2,6-Dimethylphenyl)-5-methoxypyridine (3w )
Product
4’-Benzoyl-[1,1’-biphenyl]-2-carbonitrile (3ai )
Product
Supplemental Material
sj-doc-1-chl-10.1177_17475198211026479 – Supplemental material for Organozinc-mediated direct cross-coupling under microwave irradiation
Supplemental material, sj-doc-1-chl-10.1177_17475198211026479 for Organozinc-mediated direct cross-coupling under microwave irradiation by Chun-Jing Li in Journal of Chemical Research
Footnotes
Acknowledgements
The author is thankful to Hebei University of Science & Technology for 1H NMR, 13C NMR, and HRMS (ESI) facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author gratefully acknowledges the financial support from Hebei Chemical & Pharmaceutical College.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.