Abstract
Lactic acid is an important platform compound used as raw material for the production of lactide and polylactic acid. However, its concentration and composition distribution are not as simple as those of common compounds. In this work, the mass concentration distribution of highly concentrated lactic acid is determined by back titration. The components of highly concentrated lactic acid, crude lactide, and polymer after the reaction are analyzed by HPLC. Different concentrations of lactic acid solution were prepared for the synthesis of lactide and its content in the product was determined by 1H NMR analysis. We found that lactide is more easily produced from high-concentration lactic acid solution with which the condensed water is easier to release. Hence, the removal of condensed water is crucial to the formation of lactide, although it is not directly formed by esterification of two molecules of lactic acid.
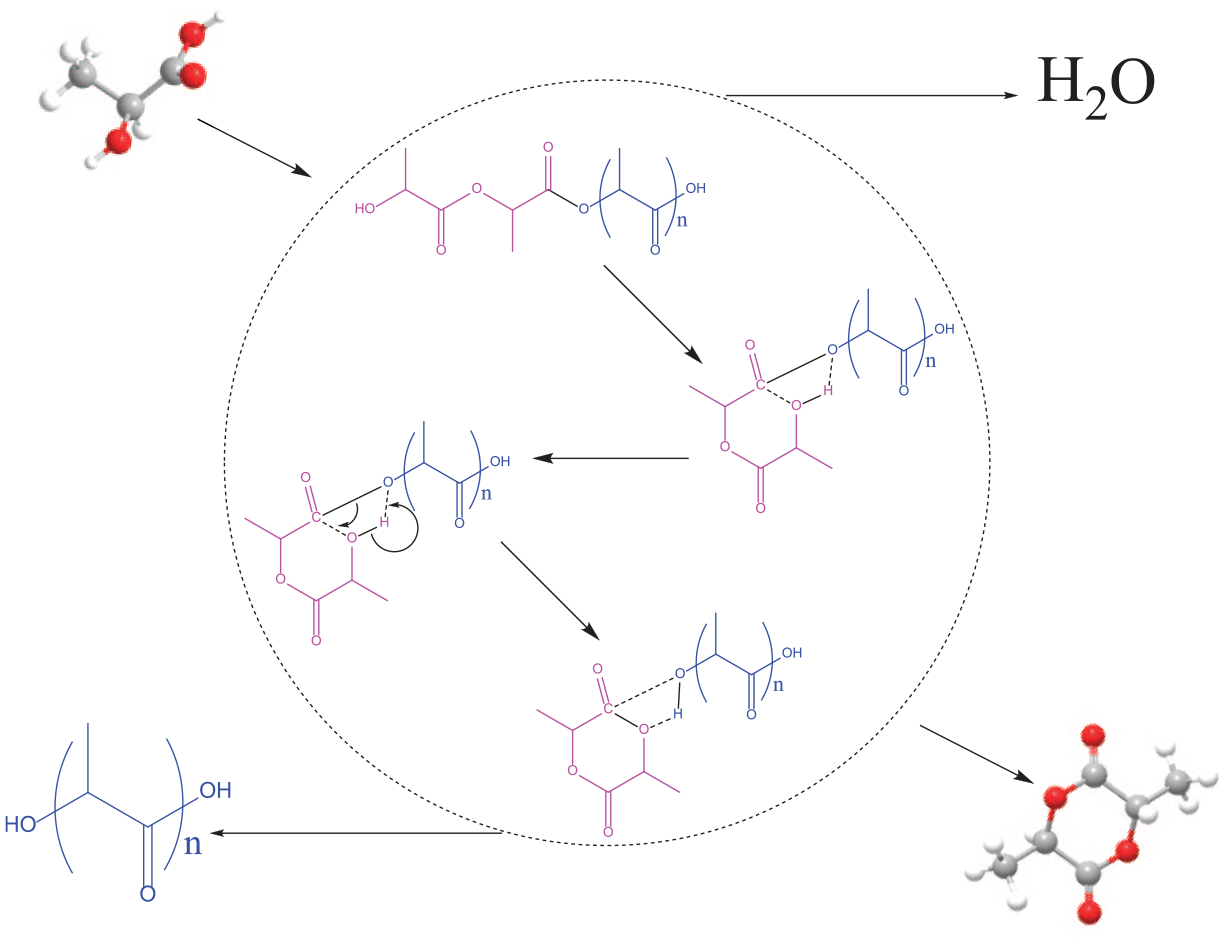
Introduction
Petroleum-based plastics are now used in many fields due to their low cost and great diversity.1,2 However, they are usually very difficult to degrade in nature unlike degradable substances. In addition, the large-scale production of petroleum-based plastics has also resulted in serious pollution because of the lack of effective means and strategies for plastic waste disposal or recycling.3,4
Fortunately, bio-based plastics are expected to overcome these environmental challenges as a substitute for petroleum-based plastics.5,6 Polylactic acid (PLA), a thermoplastic polyester with good biocompatibility and biodegradability, is one of the most well-known bio-based plastics.7,8 Currently, PLA is regarded as an indispensable material that has been used in biomedicine, fishing, forestry, agriculture, and other fields.9–12 In general, there are three methods for the production of PLA with different molecular weights, namely condensation polymerization, melt polycondensation, and ring-opening polymerization.13–16 High-molecular-weight PLA (>105 Da) can be obtained by ring-opening polymerization when lactide is used as the raw material.17–19
Lactide is a six-membered cyclic ester of lactic acid and an important intermediate for the synthesis of PLA. However, lactide has a high price due to its complex production process and low yield.20–23 Several reaction parameters, such as temperature, pressure, the type of catalyst and the molecular weight of lactic acid oligomers, have significant effects on the yield of lactide.24–27 Thus, most research now focuses on the improvement of the lactide production process and the development of various catalysts.
However, the raw lactic acid has a significant influence on the production of lactide, which has not been noted in many studies. Lactic acid (LA) is a hydroxy carboxylic acid that can be mutually soluble with water. Intermolecular esterification can occur in aqueous solutions when the lactic acid concentration is high (>30 wt%).28–30 In addition, the content of monomers and multimers is mainly determined by the lactic acid concentration, which has a direct influence on the formation of oligomers by dehydration condensation. Since only the oligomers with a certain range of molecular weight are amenable for depolymerization into lactide,31–33 the concentration is also a crucial factor for the formation of lactide.
In this work, we analyzed the mass concentration distribution of highly concentrated lactic acid (HCLA), and the existence of some other components was investigated. Meanwhile, lactic acid solutions with different concentrations were prepared for the synthesis of lactide. Herein, we report our observations and discuss the results.
Results and discussion
Concentrations of HCLA
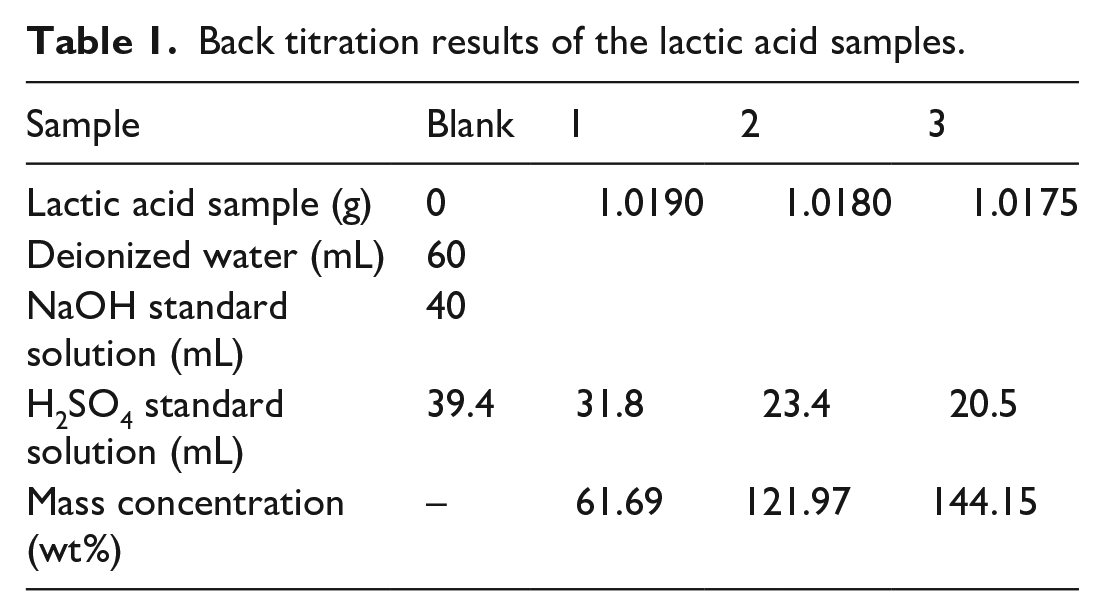
The NaOH (0.9027 mol L−1) and H2SO4 (0.8615 mol L−1) standard solutions were consumed in the sample titration, and the results of the back titration for lactic acid samples 1, 2 and 3 are shown (Table 1). Obviously, the mass concentrations of HCLA, increasing from top to bottom, are different in the presence of crystallization. The mass concentrations of the middle and lower layers, calculated from the back titration equation, were more than 100%. In this case, it was the superficial weight percent of lactic acid, which is defined as the ratio of the weight of the total monomer with the corresponding hydrolyzed water and the total solution weight. The superficial concentration of lactic acid can exceed 100% when multimer content is high enough in solution. The solution near the upper layer contains relatively more water and less lactic acid multimers, so its concentration and density are lower. The part with the higher mass concentration is also denser, so it will settle in the lower layer where the higher concentration also leads to the formation of more lactic acid crystals. This is because the degree of crystallization of lactic acid is determined by the temperature and concentration. Generally, at lower temperature, the concentration is higher and the degree of crystallization is greater. If the solution does not reach the concentration required for crystallization at a certain temperature, the lactic acid crystals will not form.
Back titration results of the lactic acid samples.
Components of HCLA
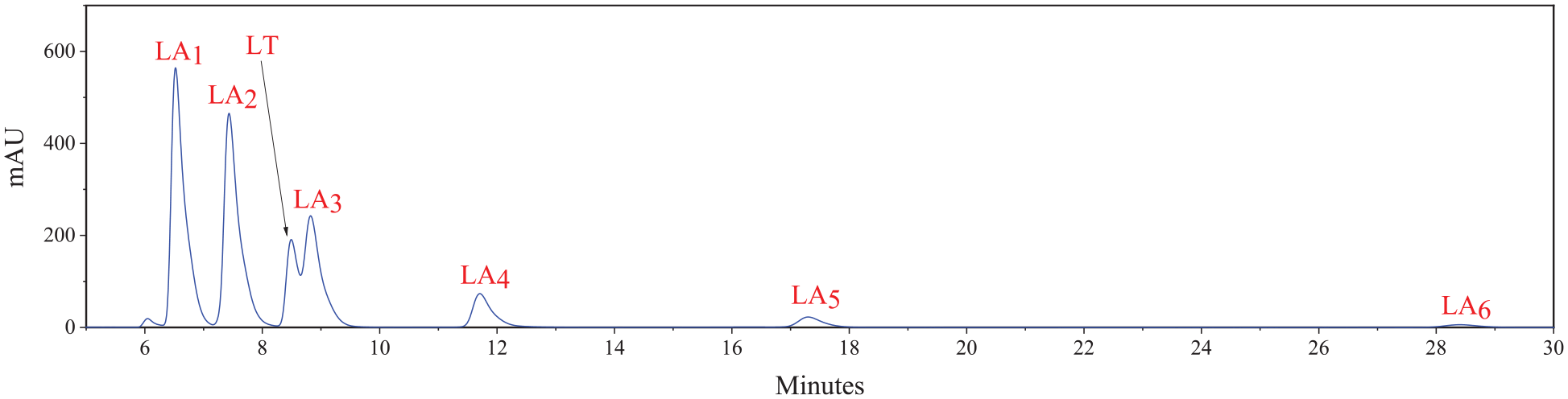
As a reference, 10 wt% lactic acid aqueous solution was also analyzed by HPLC, which showed only one peak in the HPLC chromatograph (Figures 1 and 2). The solution contains only lactic acid monomers (LA or LA1) and no multimers (LAn, n > 1) at a low concentration. Different components of HCLA were separated by such solvent, flow rate, and mobile phase in HPLC analysis, and six peaks occurred in the HPLC chromatography (Figure 3). The peak times of the different components were not the same. This indicates that HCLA consists of not only lactic acid monomers but also multimers. According to the sequence of peaks, the subsequent peaks should be lactic acid multimers, namely LA2, LA3, LA4, LA5, and LA6.

HPLC chromatograph of 10 wt% lactic acid/CH3OH solution.

HPLC chromatograph of 10 wt% lactic acid/CH3CN solution.

HPLC chromatograph of HCLA/CH3OH solution.
In the HPLC chromatograph of (HCLA + LT)/CH3OH solution (Figure 4), an additional peak occurred between LA2 and LA3 compared with that of the HCLA/CH3OH solution. The additional peak time was 8.493 min, which was the same as that of lactide standard (Figure 5) and distinguished from that of other components, while the peak times of LA1~LA6 remained basically unchanged (Table 2). This means that a new component appeared after the lactide standard was added to the HCLA/CH3OH solution. This component must be the added lactide standard, which is not originally present in highly concentrated lactic acid.
HPLC chromatograph of (HCLA + LT)/CH3OH solution.

HPLC chromatograph of lactide standard/CH3OH solution.
Peak times (min) of different components in the HPLC chromatograph.

LA: Lactic acid; HCLA: highly concentrated lactic acid.
To more accurately detect whether the structure of the ring dimer exists in highly concentrated lactic acid, 1H NMR technique was also used to analyze the HCLA sample. In the 1H NMR spectrum of the HCLA sample (Figure 6), there is a single peak at δ = 7.29 which is the

1H NMR spectrum of a highly concentrated lactic acid sample.
Many studies have also demonstrated that lactide is difficult to directly synthesize from the esterification of lactic acid, while the condensation of two molecules of lactic acid is more likely to form a linear dimer. Lactide is usually produced by depolymerization of lactic acid oligomers at high temperature.
Synthesis of lactide
After the reaction, the lactic acid solution was separated into two parts, namely the product and distillate. Oligomers formed when condensed water was released by heating lactic acid at high temperature, and then, some of these with certain molecular weight depolymerized to give lactide. Therefore, the product is mainly lactic acid polymer, but also contains lactide. The polymer was dissolved in acetonitrile and analyzed by HPLC. Polymers with different molecular weights were separated according to the solvent, flow rate, and mobile phase (Figure 7). We found that the number of components was even higher than that of highly concentrated lactic acid before the reaction.
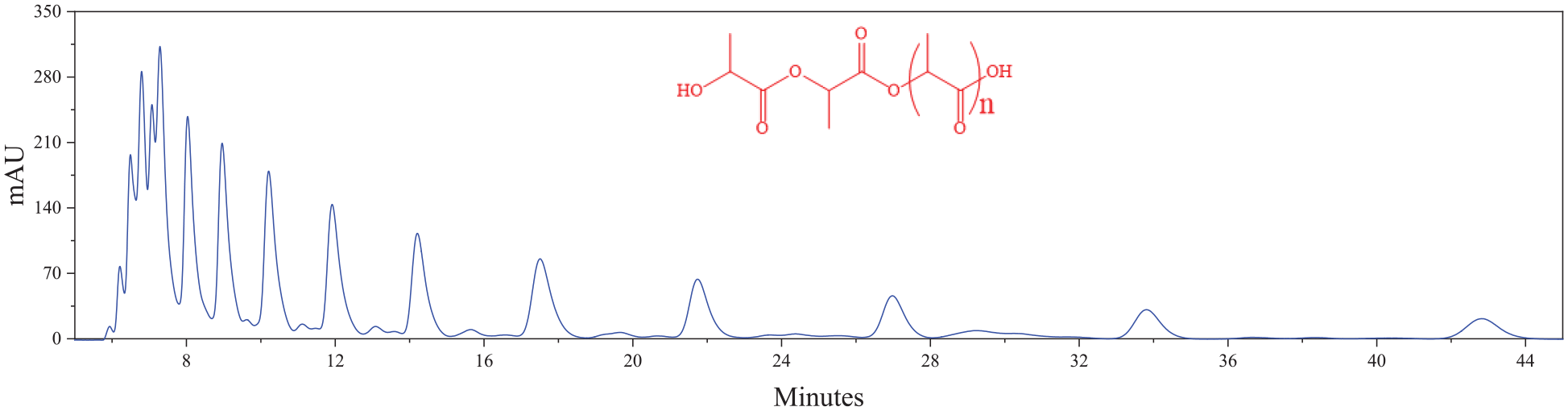
HPLC chromatograph of polymer/CH3CN solution.
The distillate is mostly water, sometimes containing very little crude lactide. The components of crude lactide can all dissolve in acetonitrile and be separated during HPLC analysis. There are three peaks in the HPLC chromatograph of crude lactide/CH3CN solution (Figure 8), which indicates that crude lactide also contains other components besides lactide. The highest peak that is the main component of crude lactide is lactide based on the peak time. The first peak time is obviously that of lactic acid monomer, so there is no doubt that the component is lactic acid. According to the peak time sequence of the components, the lactic acid dimer appears later than the monomer and earlier than the cyclic dimer. Thus, the second peak should be lactic acid dimer, since its peak time is also closer to this component in the HPLC chromatogram of (HCLA + LT)/CH3OH solution (Table 3).

HPLC chromatograph of crude lactide/CH3CN solution.
Peak times (min) of different components in the HPLC chromatograph.

LA: lactic acid.

1H NMR spectrum of the product synthesized from 61.46 wt% lactic acid solution.
It is feasible to use 1H NMR to determine the presence and content of lactide in the product. Taking the 1H NMR spectrum of product synthesized from 61.46 wt% lactic acid solution as an example (Figure 9), the presence of methylene (–CH) and methyl (–CH3) in the target product lactide molecules was detected. As can be seen, the signal at δ = 5.07 is a quater peak with an intensity ratio of 1:3:3:1 and a peak area of 3.43, indicating that it is due to –CH; the signal at δ = 1.65 shows a double peak with an intensity ratio of 1:1 and a peak area of 11.48, indicating that it is due to –CH3, respectively. Taking the single peak (δ = 7.284) area of 1 2 H of CDCl3 as the benchmark 1, the lactide concentration in the tested sample showed a linear relationship with the peak areas corresponding to –CH and –CH3.
Obviously, the results with different concentrations of lactic acid solution as reactants to produce lactide varied. When the concentrations were 78.53 wt% and 90.11 wt%, a small amount of lactide was observed in the distillate. More lactide appeared in the distillate when 90.11 wt% lactic acid solution was used, so the lactide content at the 90.11 wt% of lactic acid is smaller than at 78.53 wt% (Table 4). In this case, the gaseous lactide from the product can directly vaporize with the water under the low vacuum conditions (5~10 Kpa) due to the relatively high content. A 50.36 wt% lactic acid solution was also used to synthesize lactide, but 1H NMR analysis did not detect the presence of the lactide structure, which meant there was almost no lactide in the product. When the lactic acid concentration was low (<70 wt%), less condensed water was released, although the total water evaporation was higher. This can be explained by the presence of more free water, which is obstructive to esterification and condensation of lactic acid, and by the fact that the equilibrium of the reaction is hard to move toward formation of oligomers.
Results of lactide synthesized from lactic acid solution.
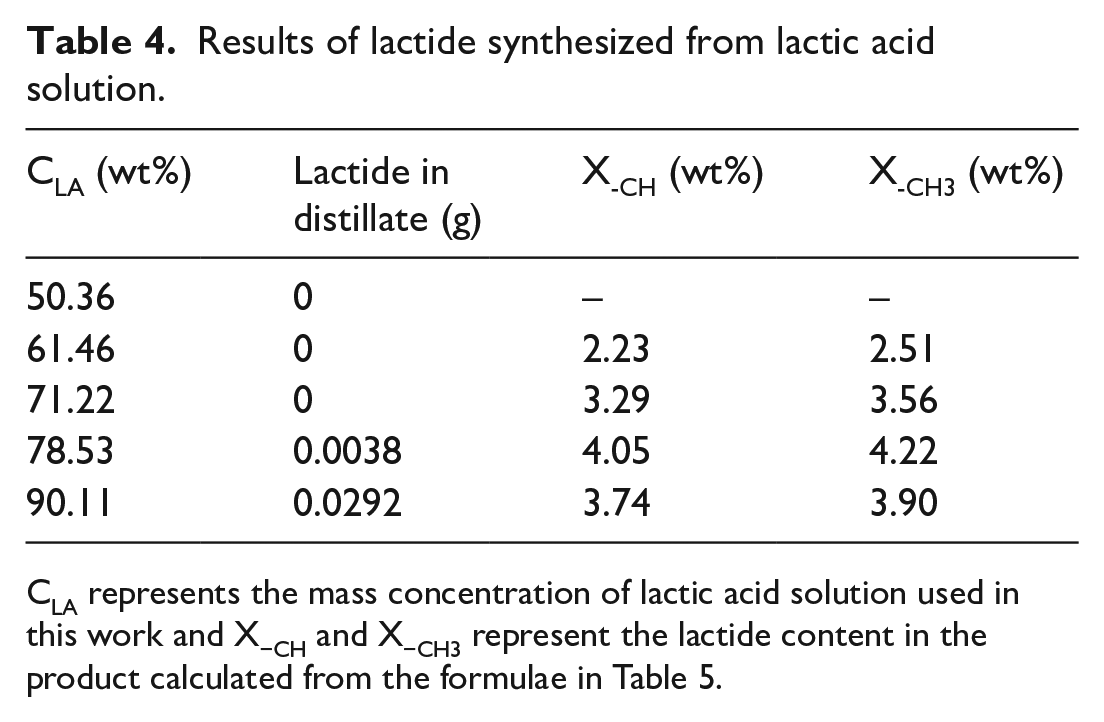
CLA represents the mass concentration of lactic acid solution used in this work and X−CH and X−CH3 represent the lactide content in the product calculated from the formulae in Table 5.
The water in the distillate consists of free water from the solution and condensed water from the polymerization of lactic acid. Free water can more easily evaporate from the reaction system when heated at 210 °C. The quantity of condensed water released depends on the degree of polymerization, which is also related to the concentration of lactic acid. For the equal mass of lactic acid, high-concentration lactic acid solution can release more condensed water. The results of the lactide content in the products calculated according to the formulae in Table 5 are reliable, although there is a small difference that is within the acceptable range. The lactide content also improves with an increase of lactic acid concentration. The variation in trend of the lactide content in the product is consistent with that of the amount of condensed water released by a unit mass lactic acid (Figure 10). It can be seen that the key to the formation of lactide is the release of intermolecular condensed water from lactic acid. Although lactide is not formed by esterification of two molecules of lactic acid, the depolymerization of oligomers to lactide is equivalent to removing two molecules of water for every two molecules of lactic acid. This is evidence that the final reaction is independent of the path. Thus, the release of condensed water is essential for the synthesis of lactide at any lactic acid concentration. By comparison, high-concentration lactic acid, which is easier to esterify and condense to form oligomers, is more likely to produce lactide.
Formulae obtained by fitting the data of the concentration and peak area.

A represents the peak area corresponding to the hydrogenation chemical shift; C represents the lactide concentration in the sample, g L−1.
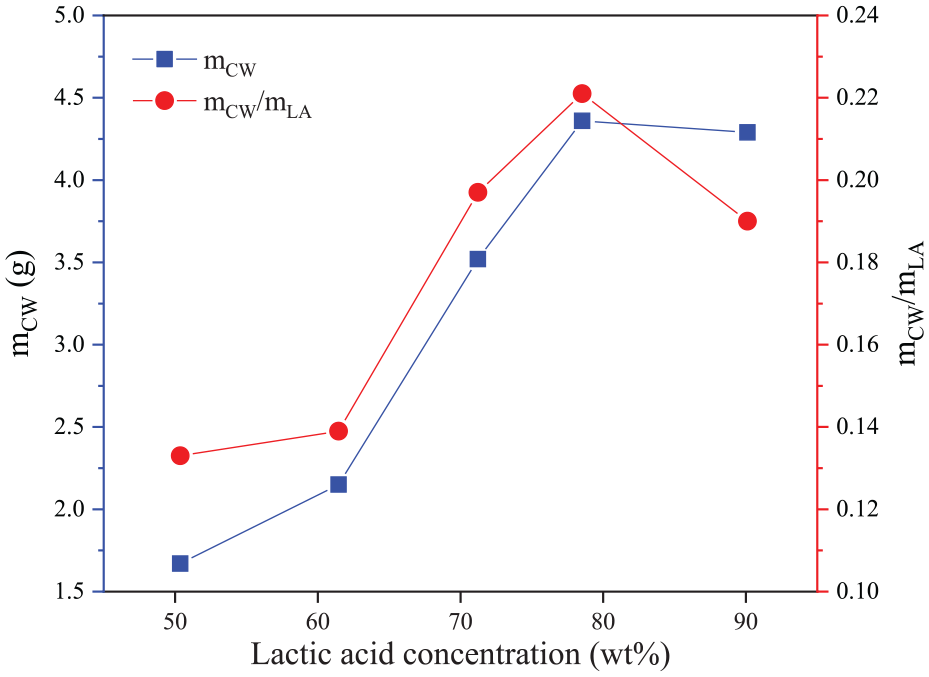
The amount of condensed water in different concentrations of lactic acid solution. (mCW represents the mass of condensed water; mLA represents the mass of lactic acid in solution).
Conclusion
In this study, we have found that the concentration distribution in highly concentrated lactic acid is uneven. A concentrated solution of lactic acid also contains lactic acid multimers, but does not contain ring dimers. Our main work has focused on the synthesis of lactide by using different concentrations of lactic acid solution, and we have found that the characteristics of the reactions for synthesizing lactide, that is, the removal of condensed water are crucial to the formation of lactide. This also provides an important explanation for the reaction mechanism of lactide synthesis. The content of free water in lactic acid solution has a direct impact on the formation of oligomers and its depolymerization into lactide. As a consequence, lactide synthesis from different concentrations of lactic acid give different results. The production of lactide from a lactic acid solution with a low concentration is difficult, while a high-concentration solution is more suitable as a raw material. This is significant for the industrial production of lactide.
Experimental
Materials
Highly concentrated lactic acid (HCLA) was purchased from Henan Jindan Lactic Acid Technology Co., Ltd. Lactide standard (LT, AR, 99%) was purchased from Sigma-Aldrich, Co., Ltd. Sodium hydroxide (NaOH, AR, ⩾96.0%), sulfuric acid (H2SO4, AR, 98.0%), and phosphoric acid (H3PO4, AR, ⩾85.0%) were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Methanol (CH3OH, HPLC grade, 99.9%) was purchased from Sinopharm Chemical Reagent Co., Ltd. Acetonitrile (CH3CN, HPLC grade, 99.9%) was purchased from Tianjin Guangfu Fine Chemical Research Institute. Chloroform-D (CDCl3, AR, 99.8%) was purchased from Cambridge Isotope Laboratories, Inc.
Methods and instruments
Back titration
About 1.0 g lactic acid sample was accurately weighed and dissolved in 60 mL of deionized water, and then 40 mL of NaOH standard solution (about 1 mol L−1) was added. The multimers of lactic acid were completely hydrolyzed by boiling for 5 min. Three drops of phenolphthalein were added as a visual indicator to titrate the sample with H2SO4 standard solution (about 1 mol L−1) while hot. At the same time, a blank titration using 40 mL of NaOH standard solution was also carried out. The total mass concentration was calculated according to following equation

where cH+ is the H+ concentration of the H2SO4 standard solution, mol L−1; Vblank, NaOH and Vsample, are the volumes of the H2SO4 standard solution consumed by titrating the sample and the blank 40 mL NaOH standard solution, respectively, mL; and m is the weight of the sample, g.
HPLC analysis
An Agilent Technologies 1200 Series liquid chromatograph equipped with an automatic sampler, a gradient flow pump, an oven and a Hitachi-L400h UV detector (set at 210 nm) were used. A Venusil C18 column (4.6 mm × 250 mm) installed in the column oven was maintained at 30 °C. The sample was analyzed at a flow rate of 5 mL min−1. 1 L of solvent (500 mL H2O + 500 mL CH3OH) was prepared as the mobile phase, and 1 mL of H3PO4 was added to adjust the pH value of the mobile phase to 2.5.
Each component of the sample has a particular peak time, so it is feasible to determine what the specific component is according to the peak time. The peak time of known substances need be measured as the identification basis. 10 wt% of lactic acid aqueous solution and lactide standards were dissolved in methanol and acetonitrile, respectively, and then analyzed by HPLC to obtain the peak time of the lactic acid monomer and cyclic dimer (Figures 1, 2, 5, and 11). Their peak time is very slightly different in the two solvents. (LA represents the lactic acid monomer; LT represents the lactic acid cyclic dimer).

HPLC chromatograph of lactide standard/CH3CN solution.
1H NMR analysis
1H NMR spectroscopy was used to analyze tested samples. According to the obtained spectrum (Figure 12), the peak area of the hydrogen chemical shift of the solvent CDCl3 (δ = 7.26) was taken as the benchmark 1 and that of –CH (δ = 5.0–5.1) and –CH3 (δ = 1.68–1.72) were recorded. Standard LT/CDCl3 solutions with concentrations of 5.0, 10.0, 15.0, 20.0, 25.0, and 30.0 g L−1 were prepared. The peak areas corresponding to the hydrogen shifts of the –CH and –CH3 groups were linearly related to the lactide concentration. After fitting the data, the lactide concentration in the sample could be calculated according to the formulae shown in Table 5.

1H NMR spectrum of lactide standard.
Titration of concentrations
Highly concentrated lactic acid contains very little water. When standing, the upper layer is a clear colorless liquid, while the lower layer is mostly milky white crystals. Samples were taken from different layers of HCLA, denoted as samples 1, 2, and 3, and their appearance characteristics were as follows:
Sample 1: taken from the surface layer of HCLA, colorless transparent liquid, no lactic acid crystals.
Sample 2: taken from the middle layer of HCLA, with certain fluidity, containing a small amount of lactic acid crystals.
Sample 3: taken from the bottom layer of HCLA, with a large amount of white lactic acid crystals.
The mass concentrations of samples 1, 2, and 3 were measured by back titration.
Analysis of the components
In order to investigate whether lactide is present in highly concentrated lactic acid, solutions of HCLA/CH3OH and (HCLA + LT)/CH3OH were prepared by the standard addition method. About 1 g of the HCLA sample was weighed and dissolved in 20 mL of CH3OH; 10 mL of this solution was removed and 0.05 g of lactide standard was added for dissolution. Samples (0.5 mL) were taken from the two solutions respectively and analyzed by HPLC.
Synthesis of lactide with lactic acid solution
The HCLA was heated in a 60–70 °C water bath to melt all the crystals and shaken to mix the lactic acid evenly. Deionized water was added to prepare lactic acid solutions with concentrations of about 50, 60, 70, 80, and 90 wt%. Their mass concentrations were measured by back titration.
25 g lactic acid solutions were placed into a two-necked 100-mL round-bottom flask, and a high-temperature resistant rotor was added to enable stirring. The reaction vessel was vacuum-pumped and decompressed to 5~10 KPa. The reactants were heated from room temperature to 210 °C in the oil bath. The heating process lasted for about 1 h based on the heating rate of the oil bath and heating was kept at 210 °C for 20 min. At the end of the reaction, the products and distillates were collected separately.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
