Abstract
An amino-functionalized three-dimensional cadmium metal–organic framework, [Cd1.5(L)(DMF)]·2H2O (complex
Keywords
A Cd-MOF was obtained based on an amino-functionalized ligand and exhibited excellent fluorescence sensing performance for Fe3+.
Introduction
Metal organic frameworks (MOFs) constructed from inorganic metal centers and bridged organic ligands have attracted considerable attention in recent years.1–3 As a new porous material different from zeolites and molecular sieves, MOFs possess a high specific surface area, a large aperture, structural functional diversity, and unsaturated metal sites.4–7 They offer opportunities for applications such as in gas storage and separation, in catalytic processes, as fluorescent sensors, and in drug delivery.8–11 In terms of structural features, the design of functional MOFs is a prerequisite for many applications, which can be regulated according to adjustable structures and modified skeletons.12–14
As is well known, heavy metal ions are harmful to humans and organisms, and thus finding sensitive and selective detection methods is an important scientific goal.15–17 At present, the most reported fluorescent MOF sensors, transition MOFs, and lanthanide MOFs contain important groups such as
Herein, we have modified and synthesized the amino-modified organic ligand 2′-amino-[1,1′:3′,1″-terphenyl]-4,4″,5′-tricarboxylic acid (H3TTCA-NH2). Taking into consideration the luminescence performance of transition metal MOFs, an amino-functionalized cadmium (Cd)-MOF, [Cd1.5(L)(DMF)]·2H2O (complex
Results and discussion
The amino-functionalized H3TTCA-NH2 was synthesized according to a literature method, and the solvothermal reaction of H3TTCA-NH2 and Cd(NO3)2 in DMF-EtOH-H2O (5/2/1, v/v/v) afforded light yellow, block-shaped crystals of complex
Crystal structure
The crystallographic data of complex
Crystal data and structure refinement of complex

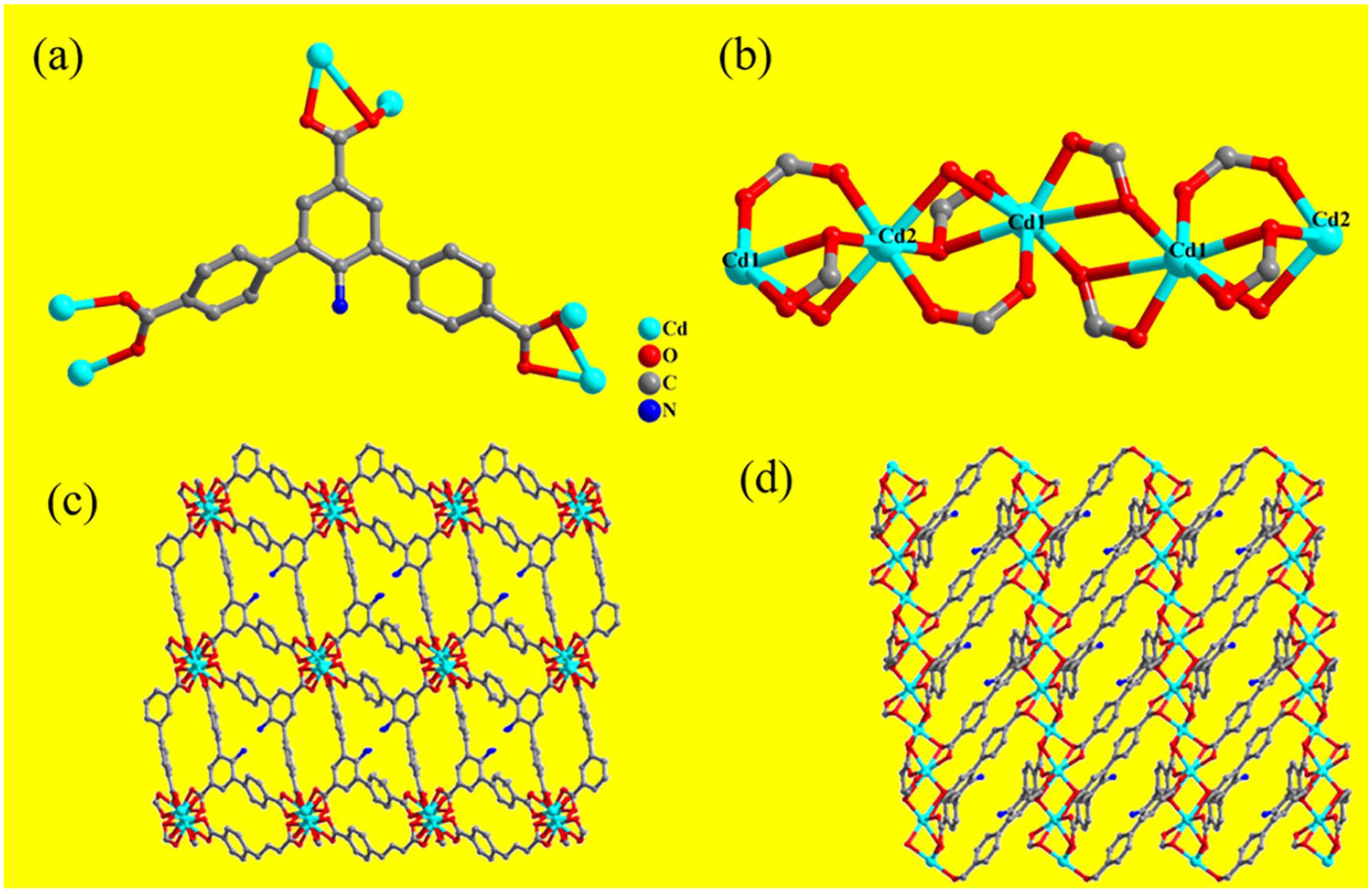
(a) The coordination environment of the ligand (TTCA-NH2)3−. (b) The one-dimensional chain SBUs. (c, d) The three-dimensional framework structure viewed from the b and c axes.
Characterization of powder diffraction, thermogravimetry, and surface area
The purity of the synthesized complex was evaluated by powder X-ray diffraction (PXRD). The PXRD figure shows that the as-synthesized crystal pattern is consistent with that simulated from SCXRD data (Figure 2(a)), indicating the pure phase of complex
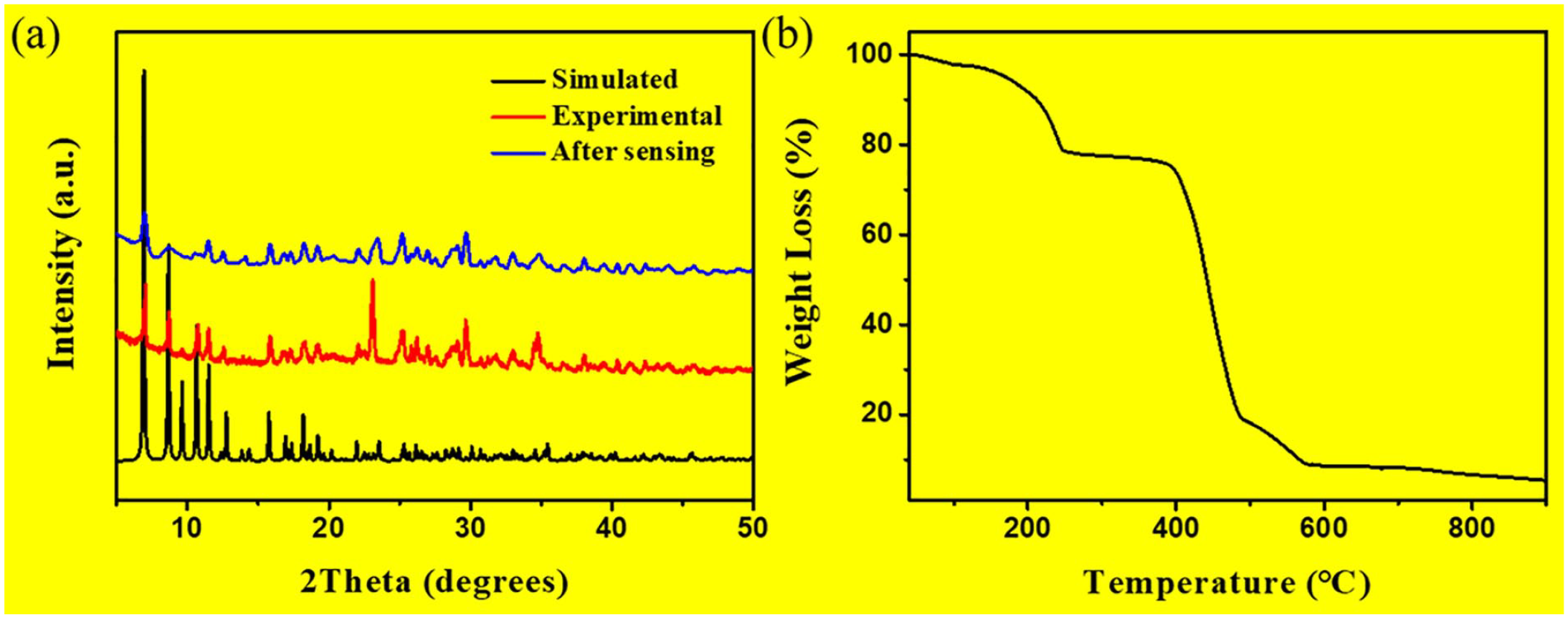
(a, b) The PXRD and TGA patterns of complex
Fluorescence studies
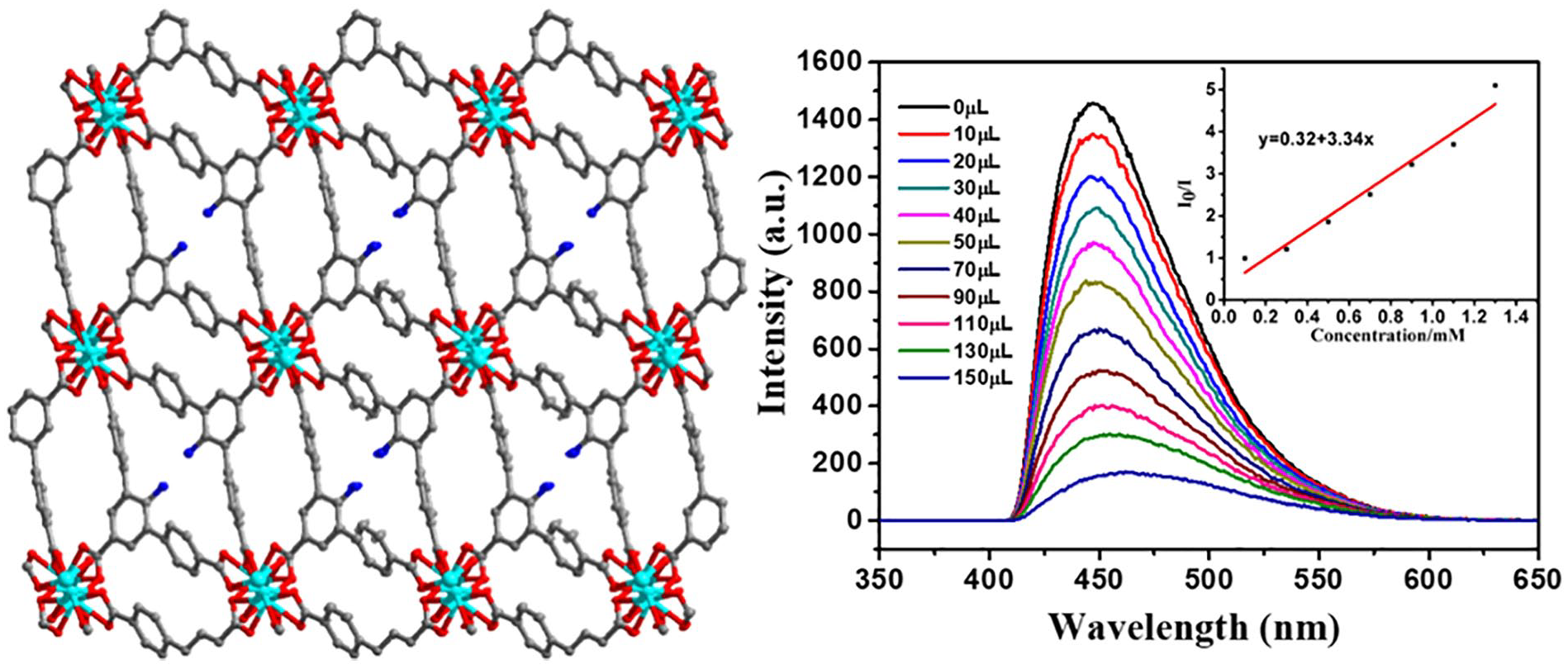
Transition-metal MOFs have specific luminescence advantages; therefore, solid state fluorescence and liquid fluorescence tests were performed using a Hitachi F-7000 fluorescence spectrophotometer. When the excitation wavelength was 330 nm, complex

(a) The solid state fluorescence spectra of complex

(a) The relative fluorescence intensity after adding eight different metal ions relative to complex
Conclusion
In summary, on the basis of designing the amino functional group–modified organic ligand (H3TTCA-NH2), an amino-functionalized 3D Cd-MOF (complex
Experimental section
Materials and method
All chemical reagents were purchased from chemical vendors and were used without further purification. The PXRD diffractograms were obtained on a PANalytical X-Pert PRO diffractometer with Cu-Kα radiation. Elemental analyses (C, H, N) were performed using a CE instruments EA 1110 elemental analyzer. IR was accomplished on a Nicolet 330 FTIR Spectrometer within the 4000–400 cm−1 region. TGA measurements were carried out on a Mettler Toledo TGA instrument under an N2 atmosphere with a heating rate of 10 °C/min in the range of 40 °C–900 °C. Fluorescence spectra were recorded with a Hitachi F-7000 Fluorescence Spectrophotometer.
Synthesis of 2′-amino-[1,1′:3′,1″-terphenyl]-4,4″,5′-tricarboxylic acid
The synthesis procedure of the H3TTCA-NH2 ligand is shown in Scheme 1, according to the literature method. 24

Synthesis procedures of the H3TTCA-NH2 ligand.
Synthesis of [Cd1.5(L)(DMF)]·2H2O (complex 1 )
Cd(NO3)2·6H2O (0.096 g, 0.28 mmol) and H3TTCA-NH2 (0.05 g, 0.13 mmol) in 10 mL of DMF-EtOH-H2O (v/v/v = 5:2:1) were sealed in a 10-mL Teflon reactor and heated to 100 °C over 40 min. The mixture was then heated at 100 °C for 3000 min and cooled to room temperature slowly at a rate of 0.1 °C/min. Light yellow, block-shaped crystals were obtained and washed with DMF and dried in the air (yield: 85% based on cadmium). Elemental analysis calcd (%) for complex
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211018981 – Supplemental material for An amino-functionalized three-dimensional cadmium metal–organic framework: Synthesis, characterization and excellent fluorescence sensing of Fe3+
Supplemental material, sj-pdf-1-chl-10.1177_17475198211018981 for An amino-functionalized three-dimensional cadmium metal–organic framework: Synthesis, characterization and excellent fluorescence sensing of Fe3+ by Jifa Yin, Youjie Han, Hongmei Zhu and Ye Tian in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Jinan Motor Vehicle Pollution Prevention and Control Center.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
