Abstract
A label-free fluorescence assay for hyaluronidase (HAase) activity based on self-assembly of quantum dots is developed. A cationic polymer (polycation) can induce aggregation of the negatively charged quantum dots through electrostatic interactions and the fluorescence of the quantum dots is quenched. When the polycation is mixed with hyaluronic acid (HA), intense binding of HA to the polycation makes the quantum dots free and recovery of the fluorescence of the quantum dots is observed. However, in the presence of HAase, HA is hydrolyzed into small fragments and the polycation induces reaggregation of the quantum dots. A simple and rapid fluorescence sensor with high sensitivity and selectivity for HAase activity detection is therefore successfully established with a detection limit of 0.01 U/mL. Moreover, we have demonstrated an assay that can be applied to detect HAase activity in a complex mixture sample including 1% human serum.
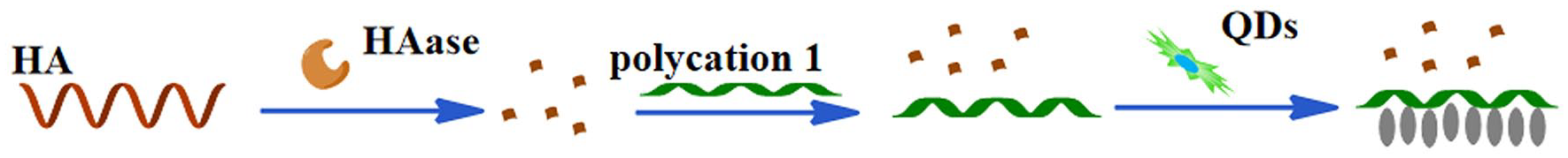
Introduction
Hyaluronic acid (HA) with repeating D-glucuronic acid and N-acetyl-D-glucosamine disaccharide units is a negatively charged linear glycosaminoglycan.1–4 It exists widely in the extracellular matrix and is closely related to various biological processes, such as cell proliferation, differentiation, and migration.4–6 As its specific enzyme, hyaluronidase (HAase) is involved in many important physiological and pathological processes.7–11 The overexpression of HAase has a relationship with many malignant tumors, such as bladder, brain, neck, and colorectal cancers.12–19 HAase is considered as a type of tumor marker. Therefore, the development of simple and sensitive detection techniques or methods for the analysis of HAase activity is of great importance and valuable for diagnosis and therapy of cancer at its early stages.
A number of techniques for HAase activity detection have been developed based on viscosimetry,20,21 turbidimetry, 22 colorimetry,23–25 zymography, 26 and fluorometry.27–31 Among them, fluorescence methods are more popular owing to their high sensitivity, convenience, and rapidity. Many advanced materials have been successfully used for fluorescence analysis of hyaluronidase activity, including gold nanoparticles,32,33 organic fluorescent dyes,34–36 conjugated polyelectrolytes (CPEs), 37 quantum dots (QDs), 38 and an upconversion luminescence material. 39 However, these assays based on covalent labeling can be complicated, time-consuming and might affect the activity of HAase. Recently, some label-free fluorescent methods have been developed to monitor the bioactivity of HAase.40–43 For instance, Yang et al. 41 developed a modification-free fluorescence assay for HAase detection via the electrostatic interaction between HA and positively charged fluorescent components. Liu et al. 43 have established a label-free fluorescence-sensing platform for HAase activity detection based on the IFE (inner filter effect) between AuNPs and CS-Au/AgNCs. Compared with these HAase detection methods using fluorophore-labeled HA, the label-free assay is simple and flexible. Thus, the development of simple and label-free fluorescent HAase assays with high stability, selectivity, and in particular, sensitivity, is still needed for early diagnosis of diseases.
Quasi-zero-dimensional nano-sized particles with quantum confinement effects are well known as quantum dots. Due to their high fluorescence quantum yields, high photostability, and excellent color tunability, QDs have been widely used for sensing various biomarkers such as DNA, aptamers, antibodies, and specific binding proteins. 44 However, QD-based methods, especially methods based on FRET (Förster resonance energy transfer), require complicated and expensive labeled peptides or antibodies and a modification of the QDs. Recently, some label-free fluorescent sensors based on QDs were developed. For instance, Xu et al. 45 developed the label-free fluorescent detection of protein kinase activity based on the aggregation behavior of unmodified quantum dots. Hu et al. 46 established a label-free fluorescence method for the detection of alkaline phosphatase (ALP) based on nucleic acid controlled aggregation of QDs.
Here, we report a label-free hyaluronidase activity assay based on electrostatic-controlled self-assembly of CdTe QDs. Our method is simple, fast, and highly sensitive. Our design principle is illustrated in Scheme 1. It can be explained as follows: (1) the polycation

Schematic illustration of hyaluronidase activity assay based on the self-assembly of QDs.
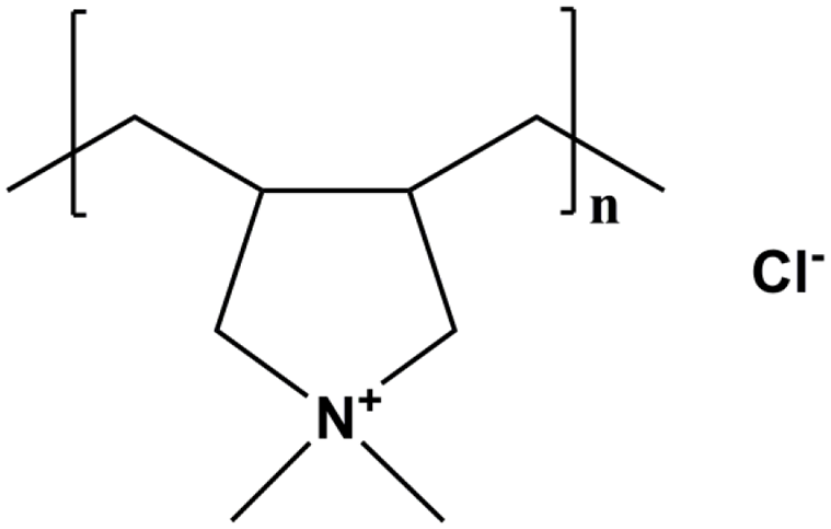
The structure of polycation
Results and discussion
Polycation-induced CdTe QDs fluorescence quenching
The quantum dots were prepared according to the literature description. 47 In a buffer solution (5 mM Tris-HCl, pH = 7.4), the CdTe QDs emit intense blue luminescence and the maximum fluorescence emission is at 534 nm (Figure 2).

Fluorescence emission spectra of the CdTe QDs (50 nM).
Polycation

(a) Emission spectra of the CdTe QDs at 534 nm against polycation
Disaggregation of the QDs in the presence of HA and fluorescence recovery
Figure 4 shows how the fluorescence spectrum and emission intensities of the CdTe QDs change with increasing amounts of HA, indicating that competitive binding of the negatively charged HA to polycation

(a) Emission spectra of the QDs against HA concentration (0, 0.005, 0.2, 1, 4, and 10 nM, respectively). (b) Plot of the emission intensity of the CdTe QDs versus HA concentration (0, 0.005, 0.2, 1, 4, and 10 nM, respectively).
The scanning electron microscope (SEM) images of the CdTe QDs sample only, of the CdTe QDs mixed with the polycation

SEM images of (a) CdTe QDs, (b) CdTe QDs + polycation
Hyaluronidase activity assay
Figure 6 shows that the CdTe QDs emission intensity gradually decreases with increasing the hyaluronidase enzymatic reaction time (0–50 min) in the presence of 0.25 U/mL HAase. This result indicates that HA is gradually degraded into fragments at an enzymatic reaction time of 50 min and the released polycation

Emission intensity of the QDs in the presence of 0.25 U/mL hyaluronidase at different enzymatic reaction times (0–50 min).
Figure 7 shows that the fluorescence spectrum and fluorescence emission intensity of the CdTe QDs decreases with HAase concentration added to the samples at 0–0.8 U/mL. The inset shows a linear relationship between the fluorescence emission intensity and HAase concentration in the range of 0–0.1 U/mL, with a calibration curve of y = 136.510 − 491.32x. The detection limit is as low as 0.01 U/mL (3σ/slope, correlation coefficient R2 = 0.989). Compared with previously reported methods,36–43 our assay method is highly sensitive.

(a) Emission spectra of the QDs against hyaluronidase concentration (0, 1, 5, 10, 15, 25, 50, and 80 mU/mL, respectively). (b) Plot of the QDs emission intensity at 534 nm versus hyaluronidase concentration at a reaction time of 40 min. Inset: the expanded linear region.
Selectivity studies
The selectivity of our method was studied. Several potential interfering enzymes such as collagenase, ALP, trypsin, and lysozyme were selected to perform the control experiment. Each enzyme was tested under the same experimental conditions as mentioned above. From Figure 8, none of these enzymes brought about the obvious fluorescence signal change of the QDs, which indicated that these enzymes did not interfere with our assay. Thus, our assay is highly selective for HAase.

Selectivity study. The QDs emission intensity at 543 nm after the addition of different enzymes. Enzyme concentration: 0.25 U/mL.
Assay in biological fluid
Our assay was also tested in a complex mixture sample including 1% human serum. The assay was carried out under the same experimental conditions. Figure 9 shows that the QDs emission intensity decreased gradually with HAase concentration (0, 0.03, 0.15, 0.25, 0.4, and 0.6 U/mL, respectively). This result shows that our method could be applied in a complex biological fluid.

The QDs emission intensities in the presence of different concentrations of hyaluronidase in 1% human serum added to the assay solution. Columns 0–5: with the addition of 0, 0.03, 0.15, 0.25, 0.4, or 0.6 U/mL HAase, respectively.
Conclusion
In summary, we successfully developed a sensitive, simple, and rapid fluorescence method for HAase activity detection based on the self-assembly of QDs. Polycation
Experimental section
Apparatus
A Fluoromax-4 spectrofluorometer (Horiba Jobin Yvon Inc., USA) was used for the fluorescence emission spectra measurements. The excitation wavelength was 350 nm. Excitation and emission slit widths of 3 nm were selected. Quartz cuvettes with a path length of 10 mm were used for fluorescence emission measurements. A Sigma 300 field-emission electron microscope (Carl Zeiss, Jena) was used to acquire SEM images. Unless otherwise specified, all spectra were taken in 5 mM Tris-HCl buffer solution (pH = 7.4).
Materials
Cadmium(II) chloride (CdCl2·5H2O) was provided by Beijing Chemical Works (Beijing, China). Tellurium powder and sodium borohydride (NaBH4) were purchased from the Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). 3-Mercaptoacetic acid was purchased from the Sinopharm Chemical Reagent Co., Ltd. Poly(diallyldimethylammonium chloride) (polycation
Polycation 1-induced CdTe QDs aggregation and fluorescence quenching
Different amounts of polycation
Fluorescence recovery of the CdTe QDs in the presence of HA
Different amounts of HA and 4 µL of polycation
Assay for hyaluronidase activity
A total volume of 200 µL of buffer solutions containing different concentrations of HAase (0, 0.01, 0.05, 0.1, 0.15, 0.25, 0.5, and 0.8 U/mL) and 4 µL of HA (100 nM) were left standing at 37 °C for 40 min and 4 µL of polycation
Selectivity studies
Collagenase, ALP, trypsin, lysozyme, and HAase (0.25 U/mL, each) were added to the buffer solutions containing 4 µL of HA (100 nM), respectively. The sample solutions were kept at 37 °C for 40 min and 4 µL of polycation
Assay in biological fluid
A total volume of 200 µL of the buffer solutions containing different concentrations of HAase (0, 0.03, 0.15, 0.25, 0.4, and 0.6 U/mL), 4 µL of HA (100 nM), and 1% human serum were prepared. The solutions were kept at 37 °C for 40 min and 4 µL of polycation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Doctoral Scientific Research Foundation of Yulin University (no.16GK12) and the Natural Science Foundation for Basic Research of Shaanxi Province of China (no. 2018JQ2041).
