Abstract
This article reports the glass transition temperatures of poly-(
Keywords
Calculation of the glass transition temperature (Tg) from different methods of poly-(

Introduction
Wholly aromatic polyamides (aramids), which contain interlaced rigid aromatic rings and polar amide bonds in their backbones, 1 are often used as high-performance organic materials. Because aramids have acid and alkali resistance, high temperature resistance, ultra-high strength, high modulus, and good mechanical properties over relatively wide temperature ranges, they have high commercial potential. They are widely used in building materials, medical treatment, aerospace, composite materials, in the military industry, and other fields.
Commercialized aramids appeared in the 1960s, but research on aramids was very limited and developed slowly.2,3 Therefore, as the demand for high-performance aramid materials in various fields increased, research on aramids increased significantly.4,5
For amorphous materials such as polyamides, the glass transition temperature (Tg) and mechanical properties are important indicators to characterize the processing and practical applications of such materials,6–9 which determine the form of the materials and the properties after being formed. Thus, Tg is very important in the selection and design of new polymer chemical structures. 10
In recent years, there have been many studies on the simulation of Tg and the mechanical properties of amorphous polymers by using molecular dynamics (MD) simulations and group contribution (GC) method. Han et al.
6
used MD simulations to study the Tg values of several amorphous polymers such as polypropylene, polybutadiene, polystyrene, and polyethylene. The relationship between polymer volume and temperature was obtained and the result was slightly higher than the experimental values. Gharagheizi et al.
11
calculated the Tg of 1,3-dialkylimidazole through GC and compared it with the experimental value. It was found that the predicted result was very close to the experimental data. Yang et al.
12
established a new data set containing 198 polymers to predict Tg∞ (Tg of limit degree of polymerization (DP)) using the improved GC method which showed a good correlation. Karteek et al.
13
created the density functional theory (DFT) forcefield and improved the traditional polymer consistent force field (PCFF) to obtain a wider applicable modified PCFF (m-PCFF), and the Tg values for a number of yet-to-be-synthesized polyhydroxyalkanoates (PHA)-based polymers with a diverse set of functional groups on the polymer side chains were predicted by using this forcefield. Kavitha et al.
14
developed a GC approach and presented property prediction methods for linear polymers. Yang et al.
15
predicted the Tg of polyethylene by the MD method, which was in good agreement with the literature. Fu et al.
16
studied the Tg and mechanical properties of a hydroxyl-terminated polybutadiene (HTPB)/plasticizer by using MD method and their research showed that the addition of a plasticizer reduces Tg and improves the mechanical properties. Yu et al.
17
studied the influence of alkyl side chains on the glass transition behavior of several carbazole trimers (CTs) in the temperature range of 423–183 K through MD. They obtained Tg from the slope intersection point, and it was consistent with the experimental values. Our research group18,19 measured the Tgs of poly(isophthaloyl metaphenylene diamine) (MPDI) and (poly-
In this work, we have designed and studied a series of aramids: poly-(
Molecular modeling
Model building and the simulation method
All molecular simulations were performed by using the Materials Studio software package of Accelrys. GC20,21 using the Synthia module and MD using the Forcite module were applied in this research. The Synthia module of the MS software was used to calculate the Tg of the modeled aramids. This module uses the GC theory from Van Krevelen and Fedors.22,23 The GC method uses the principle that some simple aspects of the structures of chemical components are always the same in many different molecules. So by following this principle, a wide variety of properties of a polymer can be predicted by knowing only its repeat unit. This method is empiric and uses data collected on several polymers that have been extensively studied.
Followed by the single chain of the PPDM, 10 different repeating units were established the by Visualizer module and 20,000 steps were optimized by using geometry optimization of the Forcite module. The obtained repeating unit with the lowest energy and the most stable conformation was used as the repeating unit for later modeling. Taking PPDM-4 as an example, its chemical structure is illustrated in Figure 1. After geometric optimization of the structure model, the molecular weight of the repeat unit was calculated by Synthia at 300 K. Then, based on the repeating unit structure, Synthia was used to set the molecular mass to 1–100 repeating unit masses to calculate the Tg of PPDM-4 with different degrees of polymerization.

Chemical structural of PPDM-4.
MD is a method that can predict Tg value of polymers, and it is the most convenient method for studying relatively complex polymers. It has been widely used to study the structures and properties of various polymers.24,25 The relationship between polymer density and temperature was obtained through MD simulations to calculate the Tg of the polymer. 26 This method is more accurate than GC, but it takes a longer time to complete.
Followed by the single-chain polymer model with a DP of 30, the repeat unit structure of PPDM was constructed (Figure 2). The molecular chain was optimized over 20,000 steps by using geometry optimization with the Forcite module, and the COMPASS forcefield was used during each stage of the simulations. The COMPASS forcefield is widely used to study condensed state polymers, especially in the study of polyamides.

Amorphous cell construction of PPDM-4.
An amorphous polymer box, with three-dimensional periodic boundary conditions, consisting of polymer chains with 30 monomer units was generated at a density of 0.6 g cm−3 using the amorphous cell module. Excessive density will cause entanglement of molecular chains, resulting in a system energy that is too high to complete the formation of a single-cell model.
In order to eliminate the local unreasonable structure generated during the modeling process and to find the lowest energy and the most stable conformation for the next dynamic simulation at the same time, the amorphous structure was optimized over 20,000 steps by using geometry optimization with the Forcite module. After this stage, subsequently 10 annealing cycles were performed from 300 to 1000 K and back to 300 K at a rate of 25 K μs−1 in a constant-pressure and constant-temperature (NPT) ensemble; temperature and pressure were controlled by employing a Nose–Hoover thermostat and a barostat, respectively. After preliminary optimization, the dynamic simulations of a 100 ps constant-volume and constant-temperature (NVT) ensemble and a 200 ps NPT ensemble were carried out successively by using the Forcite module. The basic properties such as density, bond energy, and non-bonding energy of the structure can be obtained through dynamic simulations. Taking PPDM-4 as an example, the amorphous structure obtained after all optimizations and dynamics simulations is shown in Figure 2.
MD simulations
According to the Tg obtained by the GC method, determine the temperature gradient range of the MD simulation, the gradual cooling method is used for simulation calculations. The temperature range for MD simulation is set from 660 to 340 K with an interval of 20 K, and the equilibrium conformation of the previous stage (higher temperature) is used as the initial conformation of MD of the next stage (lower temperature). The pre-equilibrium of the system at each temperature is attained from molecular simulations at NVT ensembles for 100 ps by MD of the Forcite module. In order to get a reliable average state, molecular simulations were performed by using the NPT ensemble for 200 ps in the same method, and the cutoff distance is set to 18.5 Å.
Based on the data of MD simulations, the density and specific volume are further extracted and discussed. Through analysis and calculation of modulus, the radius of gyration and the cohesive energy of the polymer are obtained. Thus, the Tg and the mechanical properties of PPDM can be obtained via these methods.
The Forcite module offers several methods for the calculation of elastic properties: constant strain, static, and stress fluctuation. The constant strain method is used in this article. This method starts by removing symmetry from the system, followed by an optional re-optimization of the structure. The resulting structure is then optimized, keeping the cell parameters (and hence the strain) fixed to allow for internal relaxation. Each strain pattern represents the strain matrix in Voigt notation. It is converted to the strain matrix,

where
From
A Forcite mechanical properties calculation may be performed on either a single structure or a series of generated structures. Young’s module in each of the Cartesian directions can be calculated from the elastic compliances. Complementary to Young’s modulus is the Poisson ratio, which measures the change in a material at right angles to the uniaxial stress. Formally, it is defined as the ratio of lateral to longitudinal strain under a uniform, uniaxial stress. The bulk modulus can be related directly to Young’s modulus and Poisson’s ratio

where
The mechanical properties of the molecular structure were calculated under the COMPASS forcefield by using the Mechanical Properties in the Forcite module. The electrostatic forces and Van der Waals forces are atom-based and group-based, respectively. The mechanical properties are then calculated as detailed in the classical simulation theory section, averaged over all valid configurations, and reported in the output text document.
Results and discussion
Selection and determination of DP
The GC method is based on the additive nature of the molecular properties, and the polymer properties can be predicted according to the quantitative structure of the substance. The relationship between the Tg of the polymer and the DP is shown in Figure 3.
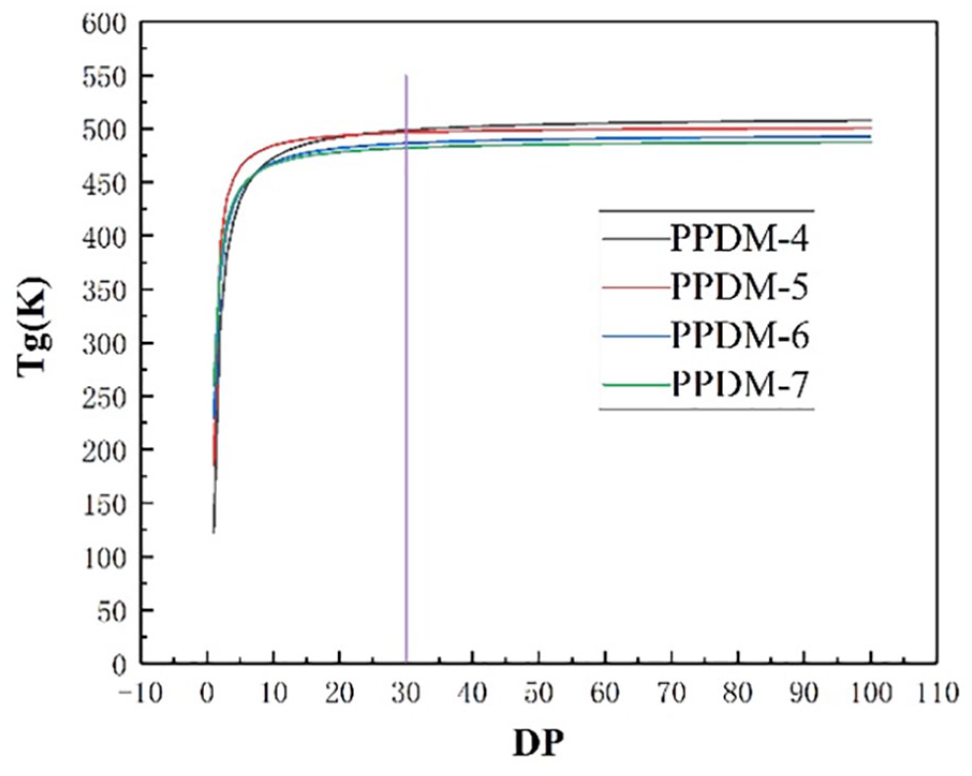
The glass transition temperature (Tg) vs degree of polymerization (DP) for PPDMs.
The figure shows that the Tg of the PPDM increases with the DP, and it tends to be in equilibrium when the DP reaches a certain value. It can be seen from the Figure 3 that the Tg of the polymer when the DP is 30 is very close to the equilibrium value. The simulation time and the complexity of the system increase for the excessive number of molecules because it will increase the difficulty of modeling amorphous structures. Therefore, the PPDM with a polymerization degree of 30 was selected to be simulated. The Tg values of PPDM-4, PPDM-5, PPDM-6, and PPDM-7 calculated using Synthia were 499.4, 491.6, 486.4, and 482.0 K, respectively.
The equilibrium determination of a dynamic system
The premise of reliable data is that the temperature and energy of the simulated system have reached equilibrium.27–29 Temperature balance requires the standard deviation of temperature change not to exceed 10%; energy balance requires that the energy value fluctuates or is constant above and below the balance value. All the models were conducted at 480 K for 200 ps using the ensembles of the constant number of particles, constant pressure, and constant temperature (NPT). The temperature and energy of PPDM changed with the simulation time, which is illustrated in Figure 4. The temperature and energy of PPDM-4 fluctuate around the equilibrium value, so the kinetic system reaches an equilibrium.

Temperature and energy fluctuation properties vs simulation time of PPDM-4 (480 K, 200 ps NPT).
The glass transition temperature
Calculation of the Tg from the density of PPDM
The density of PPDM at each temperature was calculated from the average specific volume of the system (Figure 5). A steady increase of the density with decreasing temperature and a clear change in the slope of the density curve were observed. The change in the slope of the density curve defines the value of Tg, where PPDM passes from a glassy state to a rubbery state. As the temperature decreases, it is observed that sudden increasing of the rate of density changes at a specific temperature, and the temperature corresponding to the intersection point is obtained by extending the two straight lines. According to the intersection points, the Tg values of PPDM-4, PPDM-5, PPDM-6, and PPDM-7 were 514.9, 504.7, 493.5, and 484.2 K, respectively.

Density vs temperature plots from MD simulations of PPDMs.
Calculation the Tg from the gyration radius of PPDM
According to the theory of free volume, as the temperature increases, the volume of the polymer continuously expands. Therefore, the molecular size and the distance between the atom and the center of gravity of the molecule will gradually increase. The gyration radius of PPDM can be used to predict the Tg. The radius of gyration at different temperatures was obtained by MD simulations (Figure 6). As is shown in the figure, the Tg values of PPDM-4, PPDM-5, PPDM-6, and PPDM-7 obtained from the root mean square radius are 515.0, 506.9, 494.6, and 486.4 K, respectively.
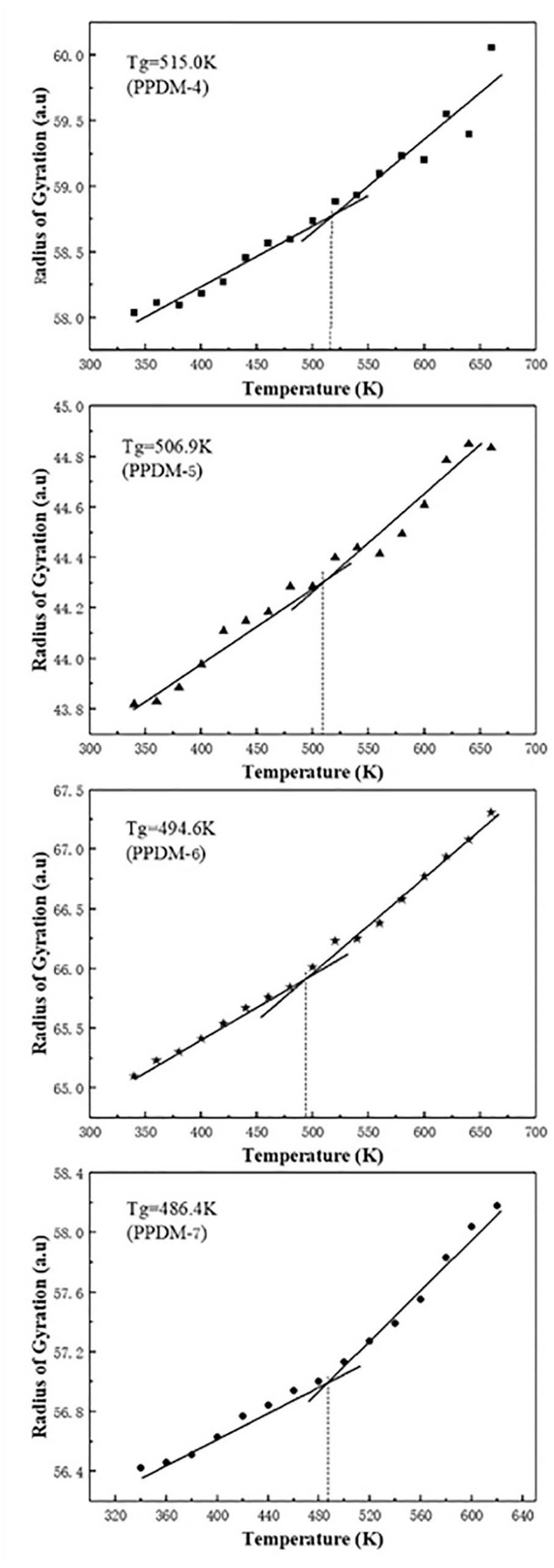
Radius of gyration vs temperature plots from MD simulations of PPDMs.
Calculation of Tg from the energy of PPDM
According to the principles of molecular mechanics,
30
the total energy of the system (

The relationship between the energy and temperature is shown in Figure 7. As is shown, the bond energy, the bond angle energy, and the torsional energy of the polymer increase in a straight line when the temperature goes up. However, there is no inflection point of the bond energy bond, the angle energy, and the torsional energy at Tg, so they are not the root cause of the Tg.
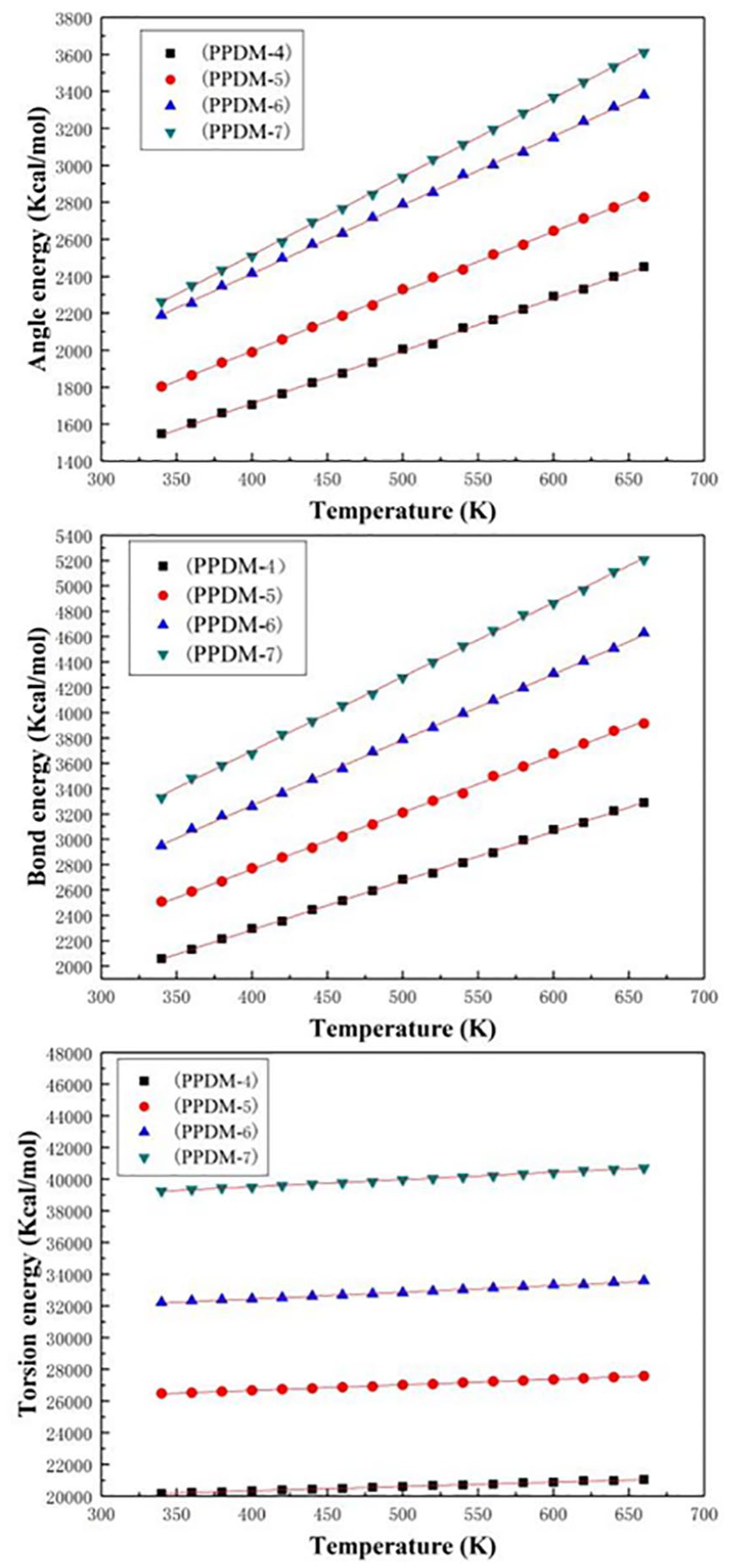
Plots of bond, angle, and torsion energy vs temperature for MD simulations of PPDMs.
As can be seen in Figure 8, the non-bonding energy changes suddenly at the glass transition point. Due to temperature limitations, the non-bonding energy increases slowly when the temperature is lower than Tg. The increase in the rate of the non-bonding energy increases significantly when the temperature is higher than the Tg, which means that the non-bonding energy may be the root cause of the glass transition of the polymer. It is found that the Tg values of PPDM-4, PPDM-5, PPDM-6, and PPDM-7 are 513.0, 505.8, 494.6, and 486.3 K, respectively.

Plots of non-bonding energy item vs temperature from MD simulations of PPDMs.
Calculation of the Tg of PPDMs by different methods
The Tg values of PPDMs obtained by different methods are shown in Table 1. The Tg obtained by the GC method is lower than the MD method, but they are all within the allowable temperature range. In the MD simulation, the Tg values of the PPDMs are measured with density, specific volume, radius of gyration, and non-bonding energy, and the results obtained by the four methods were basically consistent. With an increase in the number of benzene rings on the side groups of the main chain, the Tg showed a downward trend, and each additional benzene ring decreased Tg by an average of 10 K. Usually, the Tg could be observed along with a steady increase of the volume of the PPDM for a clear increase of the number of benzene rings and the molecular chain rigidity. However, the experimental results are contrary to the theoretical analysis. With the increase of side groups, the intermolecular distance is expanded and the interactions weakened. This means that “internal plasticization” occurs, so the Tg decreases relatively.
The glass transition temperatures of PPDMs determined via different methods.

PPDM:
Regardless of the GC method or MD simulation, the Tg of the PPDM is higher than 480 K, which indicates that these polymers have good heat resistance and can be used as a high-performance type of polyamide fibers.
Mechanical performance analysis
In order to study the effect of the increase in the number of benzene rings on the side chain of PPDM on the mechanical properties, MD was used to simulate the bulk modulus, shear modulus, and compressibility coefficient of the PPDMs at 298 K. The data obtained of the bulk modulus, shear modulus, Young’s modulus, and Poisson’s ratio are shown in the Figure 9 and Table 2, respectively.
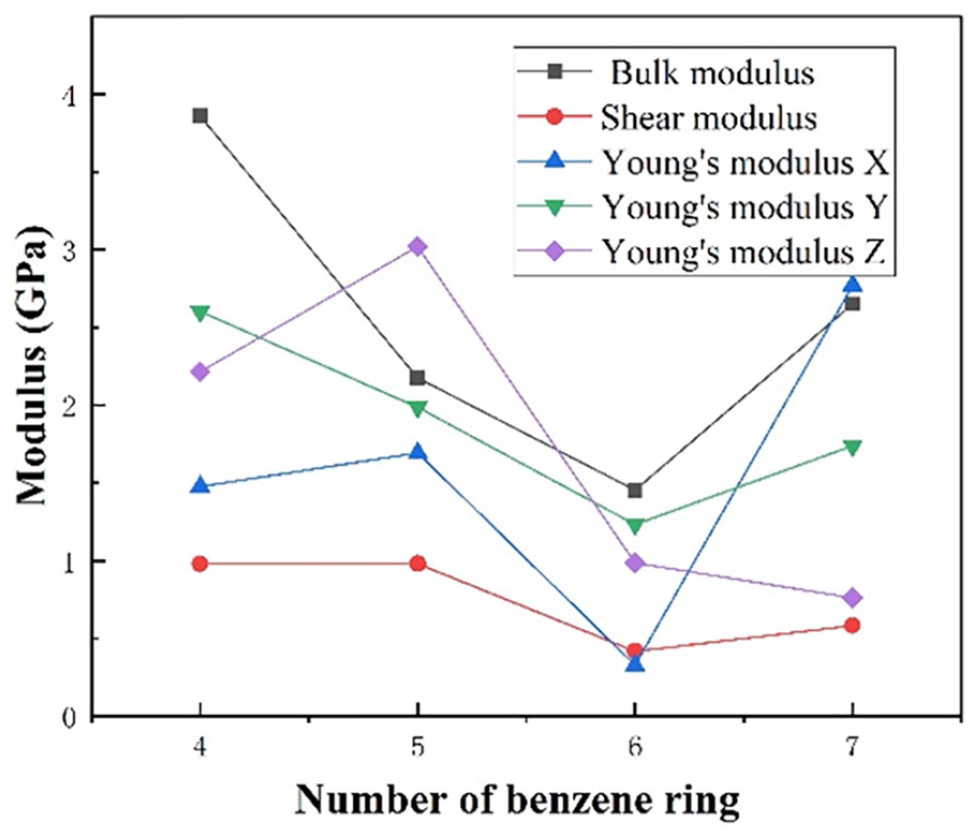
Modulus of the PPDM vs the number of benzene rings of the branch.
Poisson’s ratios of the PPDMs.

PPDM:
With an increase of the number of benzene rings on the side chain, the Poisson’s ratio has no obvious regular change. However, a negative Poisson’s ratio can be obtained on the Vxy and Vyx planes of PPDM-7, which means PPDM-7 can be used as a special high-performance material. The density of a series of PPDMs showed a downward trend and they were 1.200, 1.194, 1.185, and 1.182 g cm−3, respectively.
It can be clearly found that when the number of benzene rings on the side chain exceeds 6, the modulus does not continue to decrease as expected. This is because too many benzene rings increase the steric hindrance between molecules such that the rigidity of the polymer is also affected.
Conclusions
The Tg values of PPDMs have been studied by both MD simulations and the GC method. The temperatures obtained by the two methods are similar, and they are higher than 480 K. Hence, PPDMs are expected to be used as high-temperature-resistant polymeric materials.
With the increase in the number of benzene rings on the side groups of the main chain, the Tg showed a downward trend, and each additional benzene ring decreased the values by an average of 10 K. The steric hindrance of PPDM molecules increased with an increase of the number of benzene rings. With an increase of the volume of the side groups, the distance between the molecules expanded and the interaction weakened. This indicates that “internal plasticization” occurs, so the Tg decreases relatively.
With the addition of benzene rings on the side chain of the polymer, the Poisson’s ratio in each direction changes irregularly, but the bulk modulus, shear modulus, and Young’s modulus decrease first. When the number of benzene rings on the branch exceeds six, the modulus increases due to an increase in the intermolecular steric hindrance. It was found that the modulus of the molecule was also affected by steric hindrance.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (No. 21104031). We are grateful for the software support from Donghua University.
