Abstract
Coordination complexes constructed from the nitronyl nitroxide radical NIT-Pyra-3-Isobu and MII(hfac)2(H2O)2 as building blocks (NIT-Pyra-3-Isobu = 2-(3-isobutyl-pyrazole)-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide, hfac = hexafluoroacetylacetonato, and M = Mn or Co) are successfully synthesized. The crystal structures of the coordination compounds reveal the usefulness of the functionalized radical in providing discrete or extended architectures. Single-crystal X-ray diffraction analyses indicated that the ligand is coordinated through the oxygen atom of NO• and the N atom of the pyrazole linkage to the metal, and the metals adopt an octahedral geometry leading to one-dimensional zig-zag chain systems. The magnetic behavior of the manganese and cobalt shows overall antiferromagnetic interactions between the metal center and the organic radical.

Introduction
Studies on metal−organic radical complexes have attracted more and more attention in recent decades due to the intriguing magnetic interactions between metal ions and organic spin carriers.1–12 Among the commonly used organic radicals, nitronyl nitroxide radicals (NITRs) are particularly attracting because they are stable, easy to synthesize, and participate in numerous coupling pathways that favor the onset of bulk magnetic properties in the solid state.13–18 However, the popularity of nitronyl nitroxides mainly stems from their coordination properties. Their metal complexes are characterized by strong direct exchange couplings between organic and metal unpaired electrons, and their properties are highly dependent on the structure of the free radical and on the nature of the metal ion. In addition, modification of the substituted R groups of NITR with functional coordination groups can generate various metal–radical complexes with diverse magnetic properties. For example, three-dimensional (3D) metal–NITR complexes with different R groups can form, such as mono- and poly-nuclear complexes, as well as one-dimensional (1D) and higher dimensional architectures.19–27
Herein, we have synthesized the nitronyl nitroxide ligand 2-(3-isobutyl-pyrazole)-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide (NIT-Pyra-3-Isobu) (Scheme 1). And then, we report the syntheses, crystal structures, and magnetic properties of two complexes which were constructed from NIT-Pyra-3-Isobu and MII(hfac)2(H2O)2 as building blocks, where M = Mn or Co.

Structures of the organic radical NIT-Pyra-3-Isobu and the inorganic complexes MII(hfac)2: M = Mn or Co.
Results and discussion
Crystal structures of complexes 1 and 2
The molecular structures of
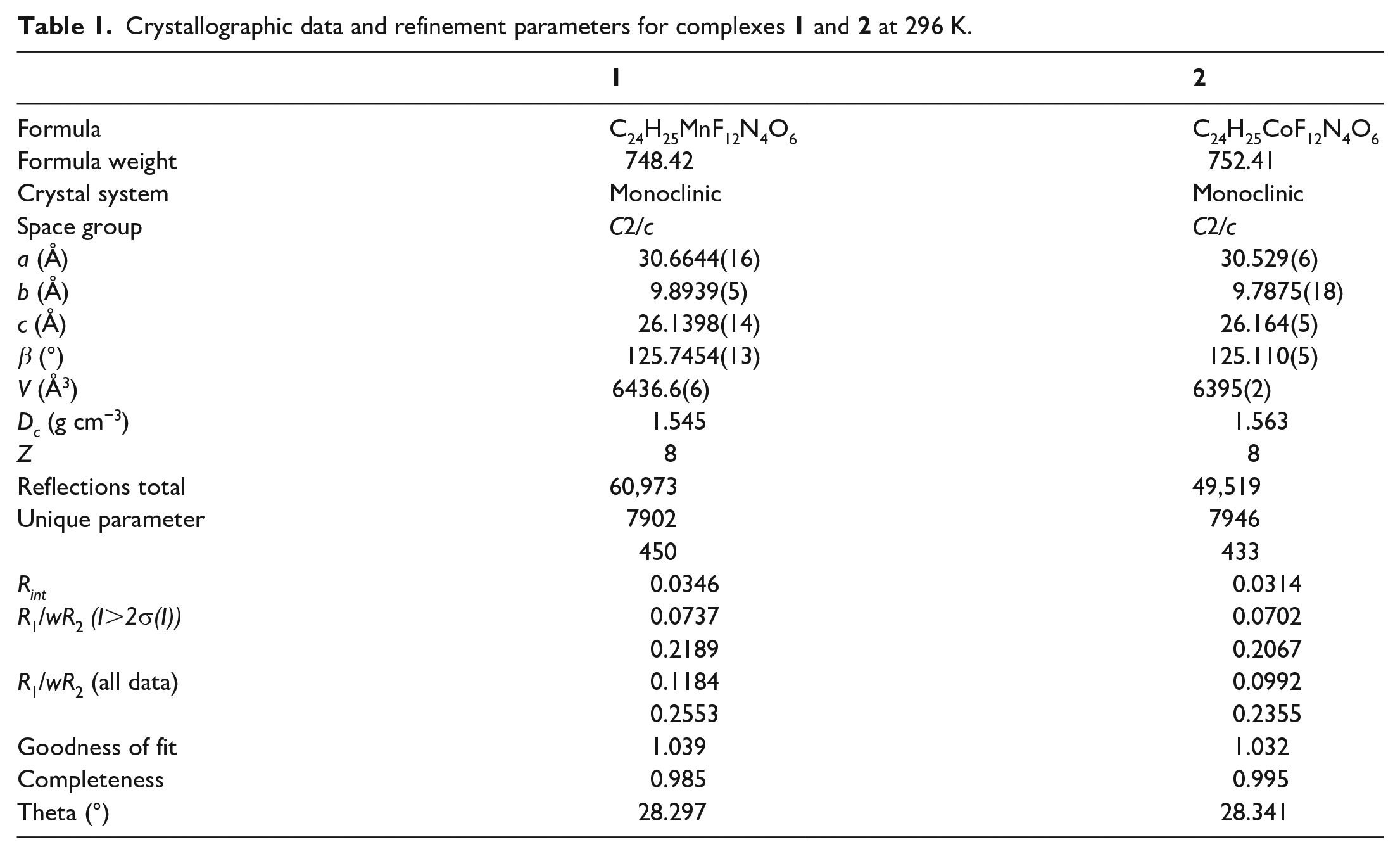
Crystallographic data and refinement parameters for complexes

The X-ray crystal structure complex

The crystal packing details of compound

Details of the crystal packing of complex
For complex
Magnetic properties
The temperature-dependent magnetic susceptibilities of the complexes were measured in the temperature range 2–300 K in an external magnetic field of 5000 Oe, and the data are corrected for the diamagnetism of the components.
The thermal dependence of the χm (square) and χmT (circle) product for complex
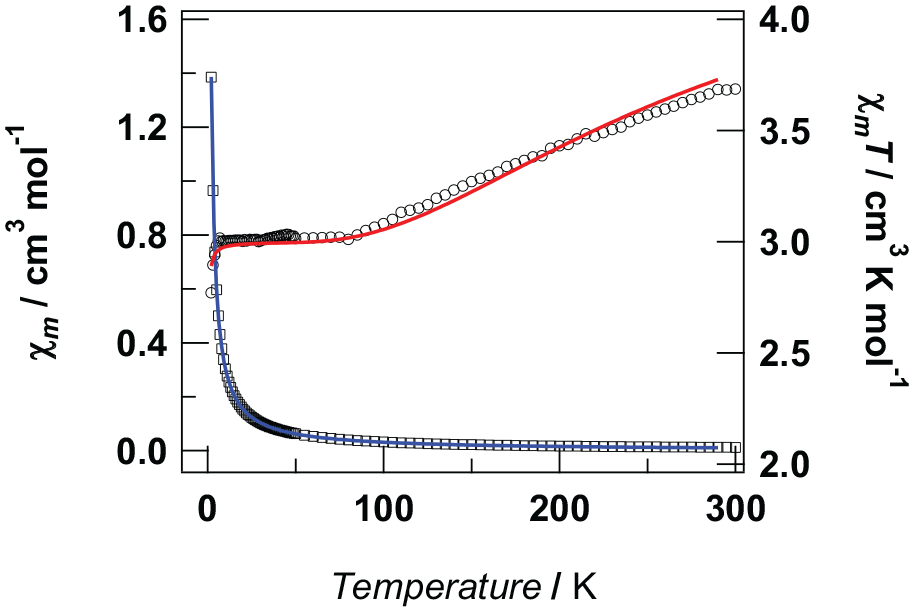
Temperature dependence of χm (squares) and χmT (circles) for complex


The thermal dependence of the χm (square) and χmT (circle) product for complex

Isothermal magnetization at 2 K of complexes

Temperature dependence of χm (squares) and χmT (circles) for complex
Conclusion
1D chain complexes {(NIT-Pyra-3-Isobu)[Mn(hfac)2]}n and {(NIT-Pyra-3-Isobu)[Mn(hfac)2]}n based on metal(II) and NITRs have been successfully prepared. The single-crystal structures show that both compounds exhibit a 1D chain structure in which Co or Mn ions are linked by the oxygen atoms of NO• groups and the N atoms of the pyrazole rings. The magnetic studies revealed antiferromagnetic interactions for both compounds and the magnetic data were fitted using simplified models through metal–radical pairs. Their magneto-structural correlations have been explained based on the crystal structures.
Experimental
Materials and methods
All reagents were obtained from commercial sources and used without further purification. The nitroxide radical was prepared as reported previously with only minor modifications (Scheme 2).24,37–39

Synthesis of the NIT-Pyra-3-Isobu radical.
General characterizations
Infrared spectra were recorded on a JASCO FT/IR-660 PLUS spectrometer by transmission through KBr pellets in the range 400–4000 cm−1. Elemental analyses for C, H, and N were carried out using a PerkinElmer series II CHNS/O Analyzer 2400. The magnetization measurements on the crystalline solids were carried out using Quantum Design MPMS-5S and MPMS-2 SQUID magnetometers. The magnetic field was varied from −50 to 50 kOe and the temperature from 2 to 300 K. The data are corrected for sample diamagnetism using Pascal’s constants.40,41
1-Isobutyl-1H-pyrazole-4-carboxaldehyde
1-Isobutylpyrazole (12.52 g, 0.1 mol) was dissolved in DMF (36 g, 0.5 mol), and POCl3 was added dropwise (30.66 g, 0.2 mol). The reaction mixture was stirred for 5 h at 70–75 °C and then poured in to cold water (200 mL). The pH of the solution was brought to 7 or 8 with saturated NaHCO3 solution. The precipitate was filtered of and dried in air. The yield is 8.87 g (58%).
Preparation of NIT-Pyra-3-Isobu
A solution of 2,3-bis-(hydroxyamino)-2,3-dimethylbutane sulfate hydrate (3.11 g, 11.7 mmol) in water (50 mL) and a mixture of 1-isobutyl-1H-pyrazole-4-carboxaldehyde (1.39 g, 10.0 mmol) in Et2O (15 mL) was stirred for 4 h at room temperature and then treated with NaHCO3 until CO2 ceased to evolve. The resulting precipitate was filtered off, washed with water and acetone, and dried in a vacuum desiccator. The obtained adduct (1.32 g, 4.9 mmol) was stirred in CH3OH (7 mL) in the presence of MnO2 (6.6 g, 75.9 mmol) for 1.5 h. The reaction mixture was filtered and the precipitate washed with CH3OH. The filtrate was evaporated and the residue dissolved in a minimum amount of ethyl acetate. The resulting solution was filtered through a silica gel layer (1.5 cm × 15 cm) and evaporated. Yield: 1.17 g (84%). FTIR (KBr, cm−1): ν = 3418 (m), 3124 (w), 2962 (s), 2875 (s), 1678 (s), 1585 (s), 1541 (m), 1510 (m), 1455 (s), 1396 (s), 1358 (s), 1321 (m), 1221 (m), 1173 (s), 1141 (s), 1058 (w), 1012 (w), 962 (w), 871 (m), 830 (m), 791 (w), 761 (w), 656 (w), 541 (m).
Preparation of Mn(hfac)2·2H2O
hfac (1 g, 7.1 mmol) was added to a suspension of Mn(CH3COO)2·H2O (0.68 g, 3.55 mmol) in H2O (17 mL) at room temperature and then NaHCO3 (0.071 g, 0.8 mmol) was added with stirring. After 10 min, a light blue precipitate was obtained which was washed three times with n-hexane. The light blue solid was dried at 50 °C for 3 days to give Mn(hfac)2·2H2O (0.74 g) was obtained. FTIR 42 (KBr, cm−1): 3398 (s), 1618 (s), 1561 (s), 1534 (s), 1497 (s), 1250 (s), 1212 (s), 800 (s), 767 (w), 743 (w), 667 (s), 590 (s).
Preparation of Co(hfac)2·2H2O
The procedure for the synthesis of Co(hfac)2·2H2O was the same as that used for except that Co(CH3COO)2·H2O. FTIR 42 (KBr, cm−1): 3367 (s), 1645 (s), 1563 (s), 1536 (s), 1488 (s), 1236 (s), 1144 (s), 806 (s), 746 (w), 676 (s), 590 (s).
Preparation of {(NIT-Pyra-3-Isobu)[Mn(hfac)2]}n (1 )
MnII(hfac)2·2(H2O) (1 mmol, 51.5 mg) was dissolved in hot n-heptane 10 mL and then the solution was cooled to about 50 °C. NIT-Pyra-3-Isobu (1 mmol, 28.1 mg), dissolved in CHCl3 (2 mL), was added with constant stirring. The solution was stirred for ca. 10 min and then cooled to room temperature. The filtrate was kept under an N2 stream at room temperature until a solid appeared. A few drops of CHCl3 were added until the solid disappeared and then the flask was sealed and placed in a refrigerator. After 2–3 days, dark blue crystals were obtained. Yield 81%. Anal. calcd for C24H25MnN4O6 are: C, 38.48; H, 3.34; N, 7.48; found: C, 38.04; H, 3.21; N, 7.51. FTIR (KBr, cm−1): ν = 2969 (m), 1648 (s), 1595 (m), 1555 (m), 1532 (m), 1482 (s), 1399 (w), 1358 (m), 1263 (s), 1212 (s), 1148 (s), 872 (w), 850 (w), 800 (s), 769 (w), 747 (w), 676 (s), 532 (m).
Preparation of {(NIT-Pyra-3-Isobu)[Co(hfac)2]}n (2 )
The method for the synthesis was similar to used for preparation of {(NIT-Pyra-3-Bu)[Mn(hfac)2]}n. Dark blue crystals in ca. 82% yield were obtained under similar conditions. Anal. calcd for C24H25CoN4O6 are: C, 38.28; H, 3.32; N, 7.44; found: C, 38.24; H, 3.26; N, 7.57. FTIR (KBr, cm−1): ν = 2969 (m), 1645 (s), 1596 (m), 1559 (m), 1531 (m), 1482 (s), 1398 (w), 1358 (m), 1266 (s), 1211 (s), 1148 (s), 872 (w), 851 (w), 800 (s), 766 (w), 746 (w), 676 (s), 535 (m).
Single-crystal X-ray diffraction studies
X-ray diffraction intensities were collected for the selected single crystals individually mounted on glass fibers at room temperature using Bruker diffractometers: SMART-APEX II with a CCD and D8-QUEST with a CMOS area detector. Both employed graphite-monochromated MoKα (λ = 0.71073 Å) radiation. Data reduction was made using SAINT, and the intensities were corrected for absorption by SADABS.43,44 The structures were solved by direct methods and refined by full-matrix least-squares against F 2 using ShelXL. 45 Part of the hydrogen atoms was located in the difference Fourier maps, and those not found were added at theoretical positions using the riding model. The details can be obtained from the cif files deposited at the Cambridge Crystallographic Data Centre. The crystal and refinement data are summarized in Table 1.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Program of Shaanxi Province (nos 2017GY-131 and 2018GY-086), the Doctoral Scientific Research Foundation of Yulin University (no. 18GK24), the Shaanxi Provincial Science and Technology Department Innovation Talents Promotion Plan Project of China (no. 2018KJXX-078), the Shaanxi Provincial Science and Technology Resources Open Sharing Platform Project (no. 2019PT-18), and the Project of Production and Study of Yulin City (no. 2016CXY-01).
