Abstract
The mechanisms of several types of thiol-independent peptide ligation reported over the past 14 years have been reviewed.

Introduction
Ligation in the peptide field has come to be recognised as the linking of a side-chain unprotected N-terminal peptide to a side-chain unprotected C-terminal peptide by a prior tethering process of the two fragments such that the formation of the new peptide link takes place intramolecularly. The first reported example of chemical peptide ligation, dubbed native chemical ligation (NCL), was reported by Dawson et al. 1 in 1994. They used the SH group of an N-terminal cysteine (Cys) of one peptide as the site of tethering and an activated carboxyl residue of a C-terminal amino acid to facilitate its attack by the SH group of the N-terminal cysteine. Specifically, an alkyl thioester (–COSR) of a side-chain unprotected C-terminal peptide was reacted with the SH group of the Cys residue of the side-chain unprotected N-terminal peptide to afford, via a thioester exchange, a (linked) thioester which generated the new peptide linkage via an S to N-acyl transfer. This Cys-based ligation has been widely adopted over the past 25 years or so to synthesise peptides and proteins, some carrying post-translational modifications (e.g. N/O-glycosylation, phosphorylation). The mechanism is well-known and not in dispute; indeed, it can be said that the concept of the tethering methodology was mechanism-based. For this reason, the mechanism of NCL will not be considered in this review.
In response to the low abundance of Cys in proteins (ca. 1.4%), several other methods have been developed over the years, among which β- or γ-thiol-containing unnatural amino acids were used as Cys surrogates. In the pursuit of thiol-independent peptide ligation methods, 16 years after the introduction of Cys-based ligation, Li et al. 2 in 2010 described a prototype of serine/threonine-based ligation (STL) and in 2013 Zhang et al. 3 published a full description. (In 2018, Liu and Li 4 published a comprehensive review of the process.) Potentially, this could extend considerably the scope of chemical ligation, since these amino acids are very abundant in natural proteins (ca. 12.7% in total). In STL, the OH group of an N-terminal serine (Ser) or threonine (Thr) of an unprotected peptide was used as the site of tethering in an analogous way to that used by the Cys-based ligation, but instead of the use of a C-terminal thioester, a C-terminal salicylaldehyde (SAL) ester of an unprotected peptide was used.
Prior to the report of STL, 2 Bode et al. 5 in 2006 reported an important variant in which the ligation was effected by a decarboxylative condensation between chemically modified natural α-amino acids, a C-terminal α-keto-acid and an N-terminal hydroxylamine. This became known as the α-keto-acid-hydroxylamine amide-forming (KAHA) ligation. Here, there is no prior tethering and the success of the selective bimolecular reaction is due to the intrinsic reactivity as a nucleophile of the hydroxylamine which reacts rapidly with the C=O group of the α-keto-acid. There are two closely related processes, one uses an unsubstituted N-terminal hydroxylamine (type I) and the other uses an N-terminal O-benzoyl hydroxylamine (type II).
In 2017, de Figueiredo et al. 6 reported an innovative procedure for dipeptide synthesis that uses an inverse activation strategy (e.g. amine activation) and it occurs with no epimerization. The activation of the amino group is effected by conversion into an N-acylimidazole derivative. It should be said that this method does not compare in scale with the STL and KAHA ligations, since it simply describes dipeptide syntheses and does not use unprotected amino acids. Indeed, de Figueiredo et al. 6 make no claim that the method is a ligation. However, because the mechanism of linking two amino acids is novel and interesting, I have included it, perhaps taking a liberty with the use of ‘ligation’.
Recently, in 2019, a novel chemoselective, high-yielding α-aminonitrile ligation was reported by Canavelli et al. 7 which exploits only prebiotically plausible molecules – hydrogen sulphide, thioacetate and ferricyanide – to yield α-peptides in water. The ligation, which is carried out at room temperature (RT), is extremely selective for α-aminonitrile coupling and tolerates all of the unprotected 20 proteinogenic amino acid residues. Here, ligation does not have the meaning that it had with STL and KAHA ligation in which large unprotected fragments are linked. Rather, Canavelli et al. 7 describe the ligation as iterative in that unprotected amino acid-derived units are added one at a time.
The mechanisms of the above ligation processes will be discussed below. Several of them have useful variants, but only the basic mechanism of each process will be considered. Each also, to varying extents, has seen many applications with impressive syntheses of large peptides and proteins, but these will not be enumerated here.
Two well-known peptide ligations are not considered here for the reasons given below. Peptide ligation via click chemistry, first reported in 2005 by Franke et al., 8 generates unnatural peptides containing 1,4-substituted [1,2,3]-triazole linkers. They are produced by a Cu-catalysed reaction between peptide fragments terminating in an azide and an alkyne, respectively. This ligation was ruled out because a new peptide bond is not formed. So too was Staudinger peptide ligation which was first reported in 2000 by Nilsson et al. 9 There the original process 9 started from an alkyl thioester of a peptide which was converted by a transthiolation reaction with o-(diphenylphosphino)-benzenethiol into a thioester of a peptide with a C-terminal –CO–S–C6H4–(o-diphenylphosphino) group. This was reacted with a peptide with an N-terminal azido group whereupon reaction between the phosphine group and the azide group in a ‘traceless’ Staudinger process produced, via a rearrangement in which extrusion of the phosphine fragment took place, a new peptide link. The dependence of this process on a thio group exempted it from consideration here.
The purpose of this review is to describe mechanisms of several types of thiol-independent peptide ligation reported over the past 14 years that have not hitherto been brought together and reviewed. These methodologies are becoming important as alternatives to the well-known native peptide ligation introduced in 1994 involving the use of an N-terminal cysteine. Moreover, the wider community of bioorganic chemists would be interested in the often ingenious methods that are being developed to link unprotected peptides together. Moreover, an understanding of the mechanisms of these methods can be very useful in the search for new ligation methodologies. Also a few current drugs and several in development are small peptides and convenient methods for their synthesis are always in demand.
The four ligation processes discussed here can be classified into two main categories, amino acid activation and aminonitrile activation, the former further sub-classified into acid activation, of which there are two variants, and amine activation in which there is just one. The mechanisms of the four ligation processes will be discussed under those headings. Each section will have two self-explanatory sub-headings: Process and Mechanism.
Amino acid activation
Acid activation
STL
Process
As will be seen, the chemoselective STL process, being an O-analogue of Cys-based ligation (NCL), is also mechanism-based. The key to the tethering process is the reaction of a free amino group of a side-chain unprotected N-terminal amino acid with the aldehyde group of a side-chain unprotected C-terminal SAL ester at the C-terminal carboxyl group. The reaction is carried out in a pyridine/acetate buffer and produces an oxazolidine as an intermediate which upon acid treatment (trifluoroacetic acid/water) yields the ligated product (Scheme 1). 3
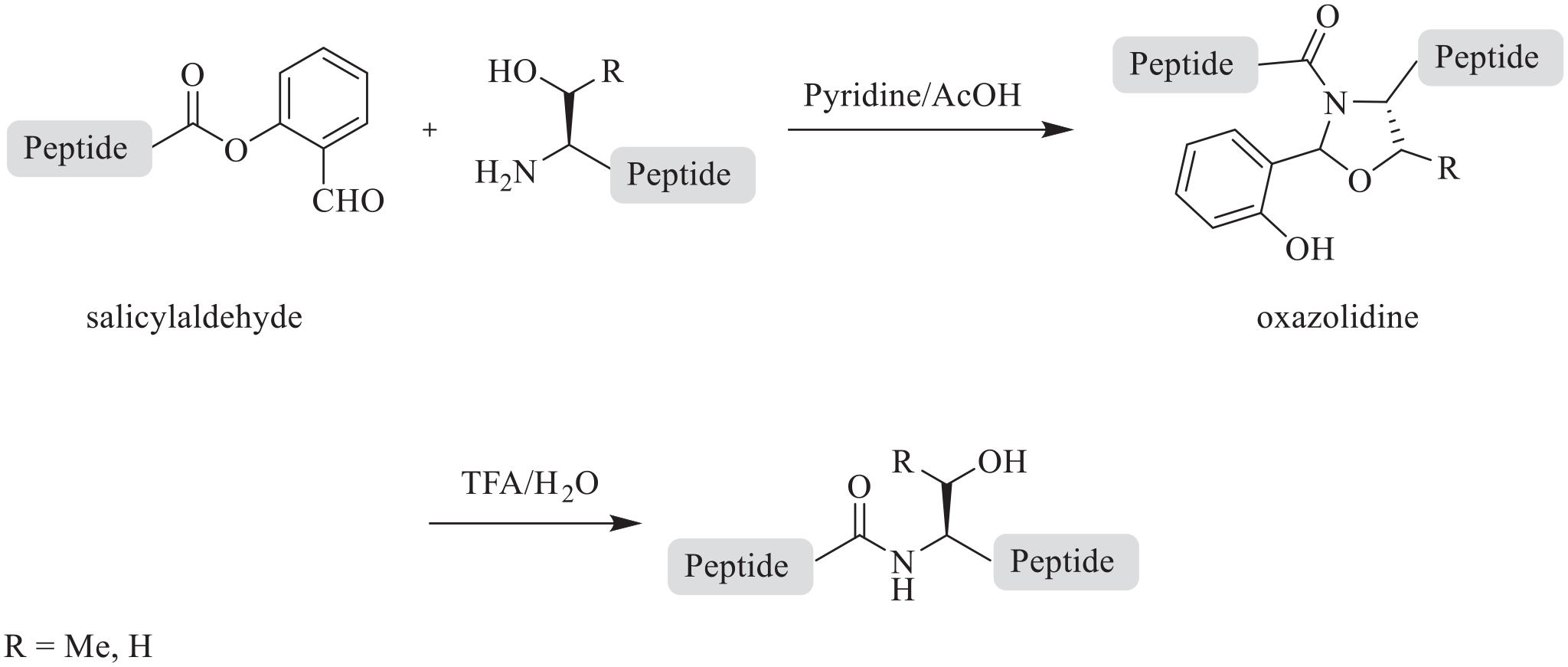
The chemoselective serine/threonine ligation (STL) process.
Mechanism
The detailed mechanism
3
(Scheme 2) involves reaction between the free NH2 group of an N-terminal Ser or Thr of a side-chain unprotected peptide (

The mechanism of the chemoselective serine/threonine ligation (STL) process.
α-Ketoacid-hydroxylamine amide-forming (KAHA) ligation
Process
In 2006, Bode et al.
5
described a novel ligation process that involves the coupling of hydroxylamines and α-ketoacids to form amides which proceeds in the presence of reactive functional groups, requires no reagents or catalysts and produces only water and CO2 as by-products There are two variants of the KAHA ligation reaction, each requiring no reagent or a catalyst (Scheme 3). The type I KAHA ligation (a) relies on the reaction in DMSO or MeOH at 40 °C of an α-ketoacid (

The two variants, (a) type I and (b) type II of the α-ketoacid-hydroxylamine amide-forming (KAHA) ligation reaction.
Mechanism
In 2012, Pusterla and Bode 10 reported their investigations into the mechanism of KAHA ligation. They carried out three 18O-labelling experiments using 18O-phenalkylhydroxylamines and 18O-phenalkyl-α-ketoacids, the results of which (Table 1) ruled out some mechanisms for both type I and type II. Surprisingly, in the type I process, the oxygen of the amide product originated from the hydroxylamine (entry 2) rather than from the ketonic C=O group of the α-ketoacid (entry 1). However, in the type II process, the oxygen of the amide product did originate from the ketonic C=O group of the α-ketoacid (entry 3).
The results of three 18O-labelling experiments using 18O-phenalkylhydroxylamines and 18O-phenalkyl-α-ketoacids carried out in studies of the mechanism of the type I and type II α-ketoacid-hydroxylamine amide-forming (KAHA) ligation reaction.

Their proposed mechanism for the type I KAHA ligation that took account of those results involved lactone and oxiridine intermediates. That mechanism will not be reproduced here, since a subsequent theoretical study showed that it was energetically less favoured than an alternative pathway. The labelling pattern obtained with the type II process indicated a quite different mechanism which was not delineated at that time.
Some 5 years later in 2017, Patil
11
reported his computational investigations of the mechanisms of both types of KAHA ligation. For the type I process, the energetically favoured mechanism that he proposed differed markedly from that of Pusterla and Bode,
10
yet still took account of the results of their 18O-labelling experiments (Table 1). The model compounds used in the calculations for the type I process were phenylpyruvic acid (
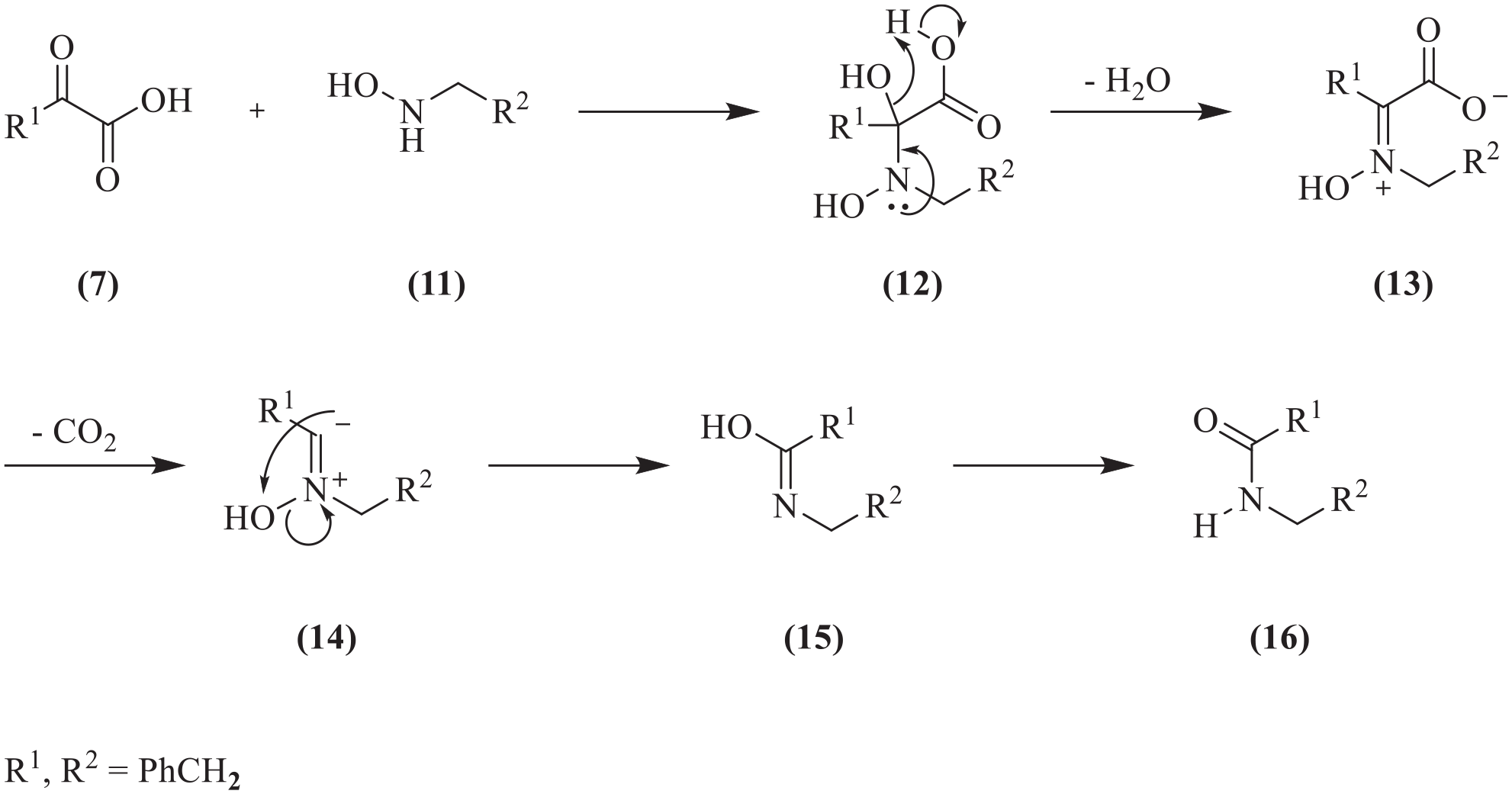
The mechanism of the type I α-ketoacid-hydroxylamine amide-forming (KAHA) ligation reaction.

The mechanism of the type II α-ketoacid-hydroxylamine amide-forming (KAHA) ligation reaction.
Amine activation
N,Nʹ-carbonyldiimidazole (CDI)-based ligation
Process
Classical amide synthesis methods involve the activation of the carboxylic acid to a reactive acyl derivative in order to allow facile reaction with an amine, and the foregoing methods all apply this methodology for the synthesis of peptides. Now, a recently reported innovative procedure for peptide synthesis goes through an inverse activation strategy (e.g. amine activation) and it occurs with no epimerization. 6 The activation of the amine is by conversion to an N-acylimidazole derivative.
Mechanism
In a typical dipeptide synthesis (Scheme 6), the activation of the amino group of an α-amino ester (

The process and mechanism of a dipeptide synthesis using the N,Nʹ-carbonyl-diimidazole (CDI)-based ligation.
Aminonitrile activation/ligation
Process
Addition of an amino acid to a peptide (

The α-aminonitrile ligation process.
Mechanism
In early model experiments reported by Canavelli et al.,
7
a key step in the efficiency of the process was identified as the conversion of an α-amino acid nitrile (AA-CN) (

(a) The conversion of an α-aminonitrile (AA-CN) into an α-amidonitrile (Ac-AA-CN) via oxidative acetylation with potassium ferricyanide/thioacetic acid and (b) the mechanism of thiolysis of an α-amidonitrile (Ac-AA-CN) to an α-amidothioamide (Ac-AA-SNH2) and of its hydrolysis to an α-amidothioacid (Ac-AA-SH).

A schematic showing the sequential thiolysis and hydrolysis of an α-aminonitrile (AA-CN) and of an N-acetyl-α-aminonitrile (Ac-AA-CN) in which only the latter yields a unique hydrolysis product, an N-acetylated thioacid (Ac-AA-SH).

The general mechanism of the conversion of a compound containing an amino group into its N-acyl derivative via oxidative acylation with potassium ferricyanide/thioacetic acid.
The quantitative nature of these reactions was demonstrated by following the four-step conversion of Gly-CN (50 mM) to Ac-Gly2-CN (Scheme 11) by 1H NMR (600 MHz, H2O:D2O::98:2, 25 °C). 7 In the first step, Gly-CN was converted into Ac-Gly-CN by quantitative oxidative acetylation using thioacetic acid/potassium ferricyanide at RT. Thiolysis (H2S) of Ac-Gly-CN at pH 9 in water at RT for 10 min to Ac-Gly-SNH2 was also quantitative, but hydrolysis in water at pH 9 at 60 °C to Ac-Gly-SH was only 80% complete after 24 h. The crucial chain-lengthening process in which Ac-Gly-SH is converted to Ac-Gly2-CN using Gly-CN/potassium ferricyanide at RT was quantitative after 20 min.

The demonstration of the quantitative nature of the α-aminonitrile homologation by following the four-step conversion of Gly-CN to Ac-Gly2-CN by 1H NMR.
Impressively and importantly, the iterative ligation cycle in which an N-acetyl-α-amino acid nitrile (
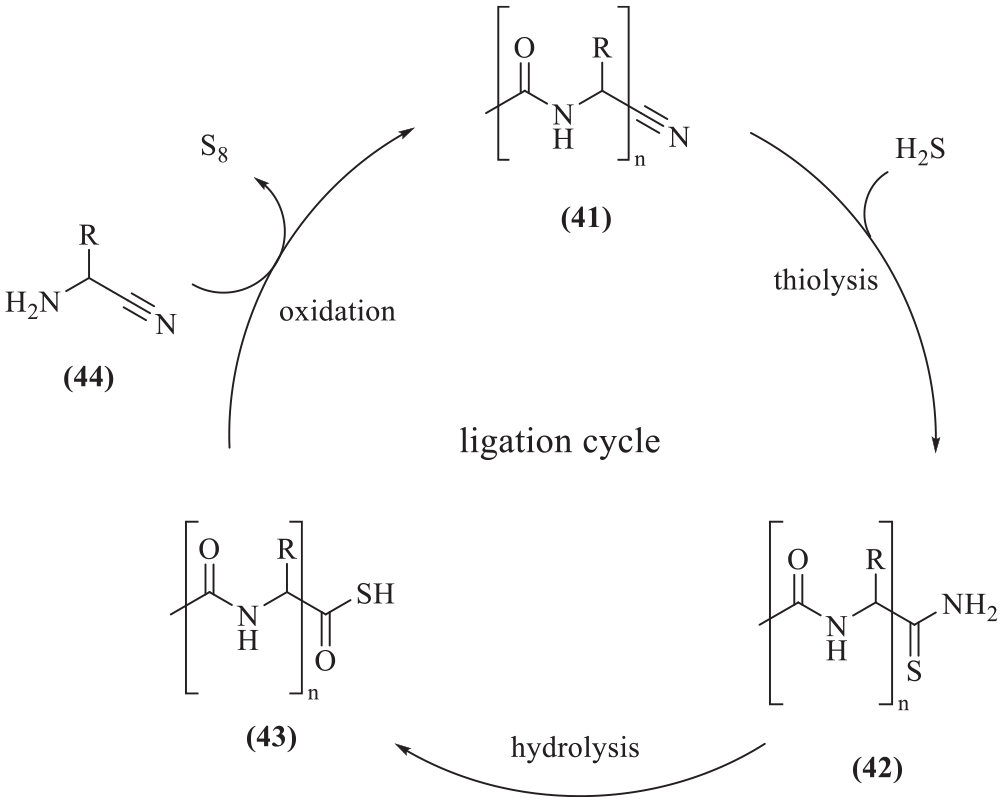
The iterative ligation cycle in which an N-acetyl-α-amino acid nitrile is converted to a higher homologue.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
