Abstract
The dye mixtures formed from three commercial hair colour formers were purified by absorption onto human hair wefts, washed and dried, extracted with dichloromethane:trifluoroacetic acid (75:25) and then analysed by liquid chromatography–mass spectrometry. Only 1–2 dyes were identified from each complex mixture of commercial aromatic amines along with a broad UV absorption mainly consisting of mixtures of quaternary ammonium salts from shampoos and some surfactants. Mecetronium ethyl sulfate and didecyldimethylammonium chloride were the main ammonium salts.
Liquid chromatography–mass spectrometry (LC–MS) chart of Bandrowski’s base (the trimer of p-phenylenediamine) after absorption and extraction from a hair weft.

Introduction
Hair colouration is a popular fashion
1
with different colours sought after.
2
Modern products can be divided into two types, oxidative3–5 and nonoxidative,
6
depending upon the type of chemistry that is used.
1
Permanent or oxidative hair dyeing requires three main components (Figures 1 and 2). The first is an easily oxidised electron rich aromatic amine such as p-phenylenediamine


The primary intermediates and couplers present in some different hair colour formers
Discussion
The dyes formed from three commercial hair colour formers were analysed and a control was performed using pure p-phenylenediamine
Table of the amines present in each hair colouration pack. The colours are taken from the colours on each hair colour former packet. Compounds

LC–MS analysis of the dyes extracted from hair wefts dyed from Honey Blonde, Light Brown Warm and Dark Brown commercial colour formers and a control
The experimental method was much the same as that used with the commercial hair colour former packet and is described in the experimental section. p-Phenylenediamine

LC–MS gradient 1 chart 1 of dye
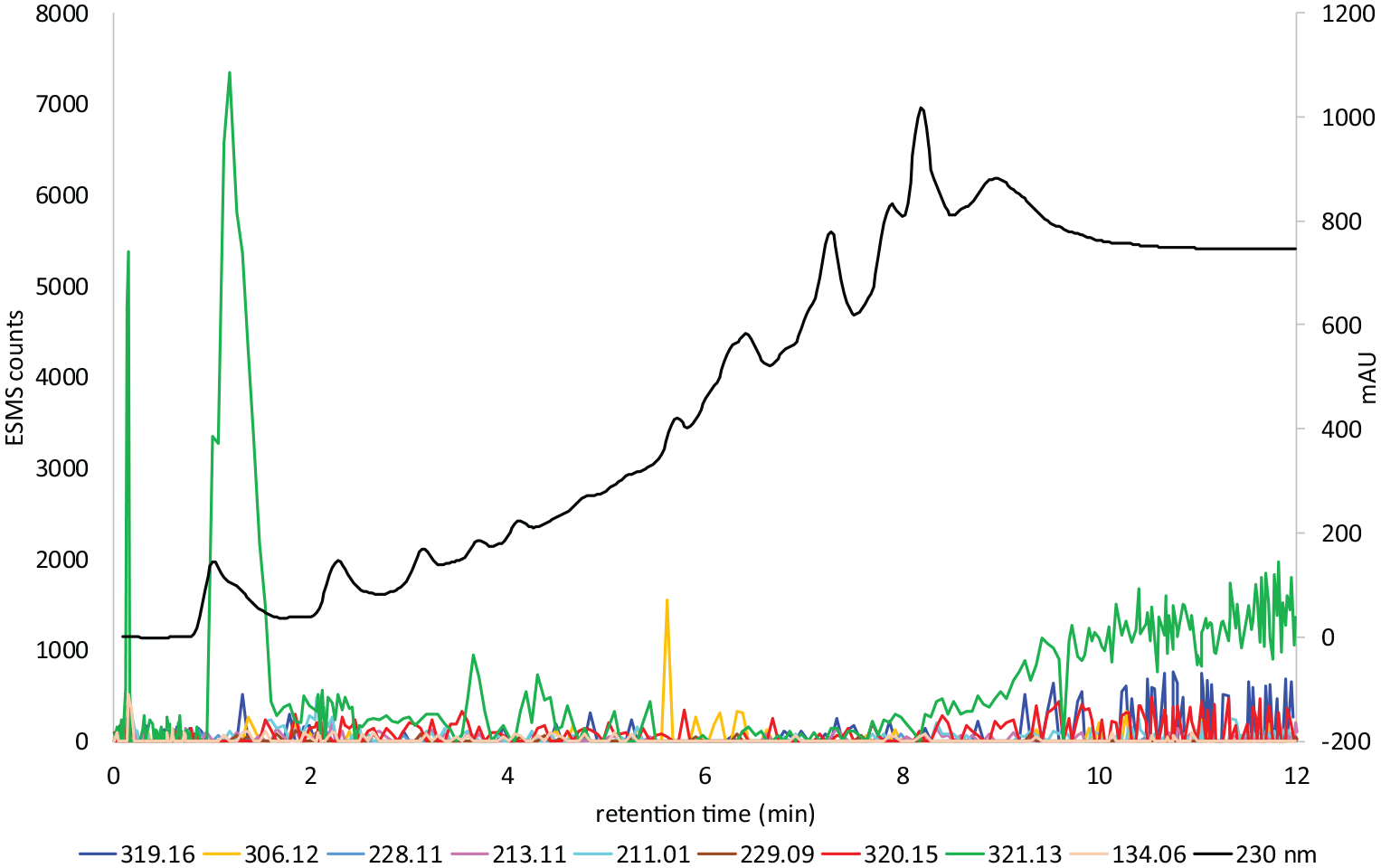
LC–MS gradient 1 chart 1 of dye

LC–MS gradient 1 chart 1 of dyes

LC–MS gradient 1 chart 1 of dyes

Summary of representative dyes proposed to form in hair colour former packets which were extracted after absorption onto hair wefts.
Figure 4 shows the LC–MS analysis of the hair extract formed from the in situ oxidation of the commercial colour former Honey Blonde with aqH2O2. A strong absorption occurs at m/z 321.1347 (M + H) (calculated m/z 321.1346). This is assigned as the known compound
Figure 5 shows the LC–MS analysis of the hair extract formed from the in situ oxidation of the commercial colour former Light Brown Warm with aqH2O2. A strong absorption occurs at m/z 320.1507 (M + H) (calculated m/z 320.1506) and m/z 319.1666 (M + H) (calculated m/z 319.1666). These are assigned as the known compounds
Figure 6 shows the LC–MS analysis of the hair extract formed from the in situ oxidation of the commercial colour former Dark Brown with aqH2O2. A strong absorption occurs at m/z 321.1347 (M + H) (calculated m/z 321.1346) and m/z 320.1507 (M + H) (calculated m/z 320.1506). These are assigned as the known compounds
The dyes observed were weaker in intensity than expected and were swamped out by much stronger chromophores, hence the need to analyse the data in stages. The number of dyes observed, three in total, was much less than expected and many more must surely form. These are the known compounds
Figures 8–16 show the gradient 1 LC–MS charts of Honey Blonde, Light Brown Warm and Dark Brown colour former with more information added in stages about the UV absorbing material. File 1 Supplemental Figures 17S–19S gradient 1 and Files 2–5 Supplemental Figures 20S–35S gradient 2 are in the Supplemental. Table 2, compounds

LC–MS gradient 1 chart 2 of Honey Blonde colour former.

LC–MS gradient 1 chart 3 of Honey Blonde colour former.

LC–MS gradient 1 chart 4 of Honey Blonde colour former.
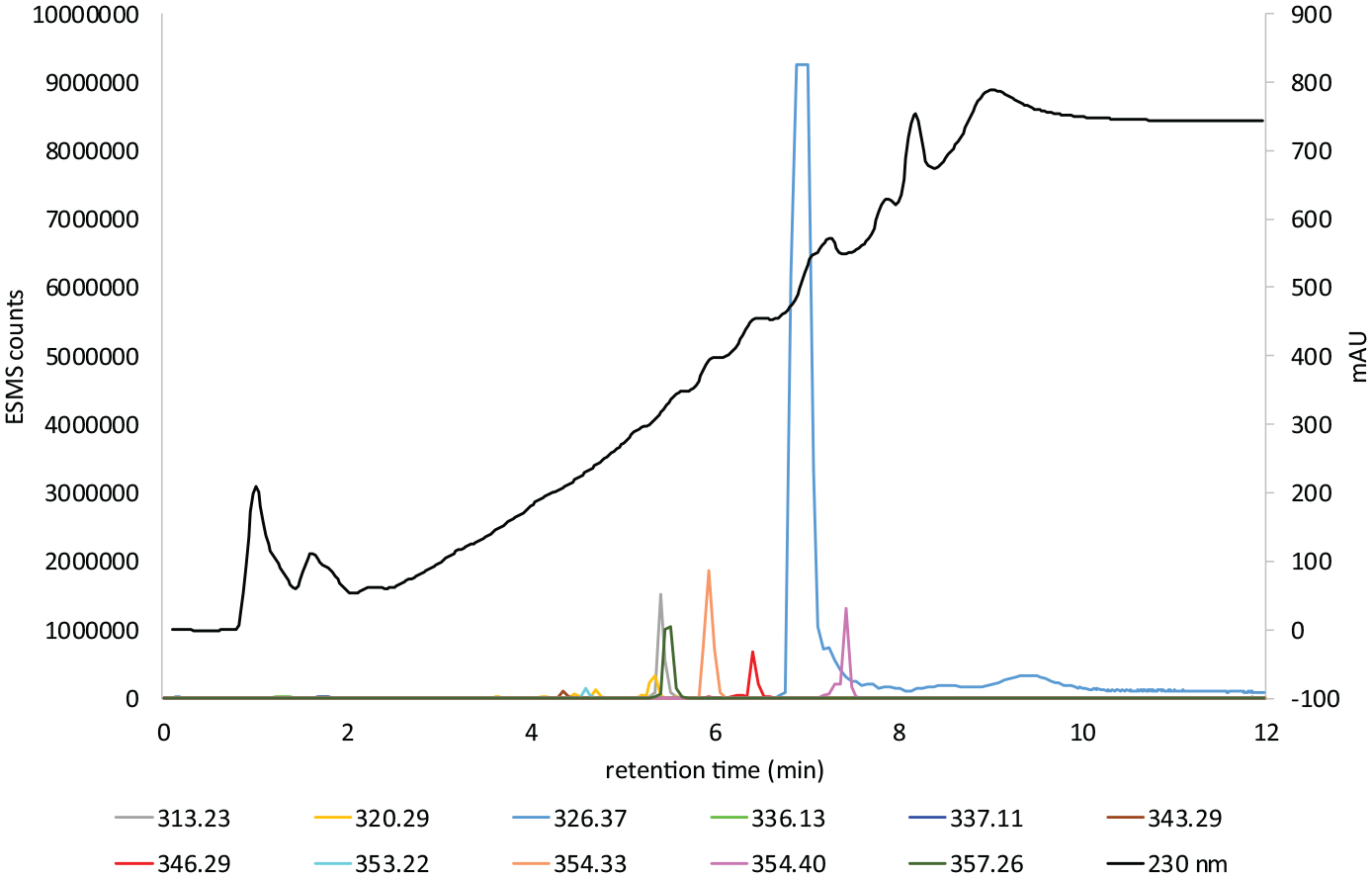
LC–MS gradient 1 chart 2 of Light Brown Warm colour former.

LC–MS gradient 1 chart 3 of Light Brown Warm colour former.

LC–MS gradient 1 chart 4 of Light Brown Warm colour former.
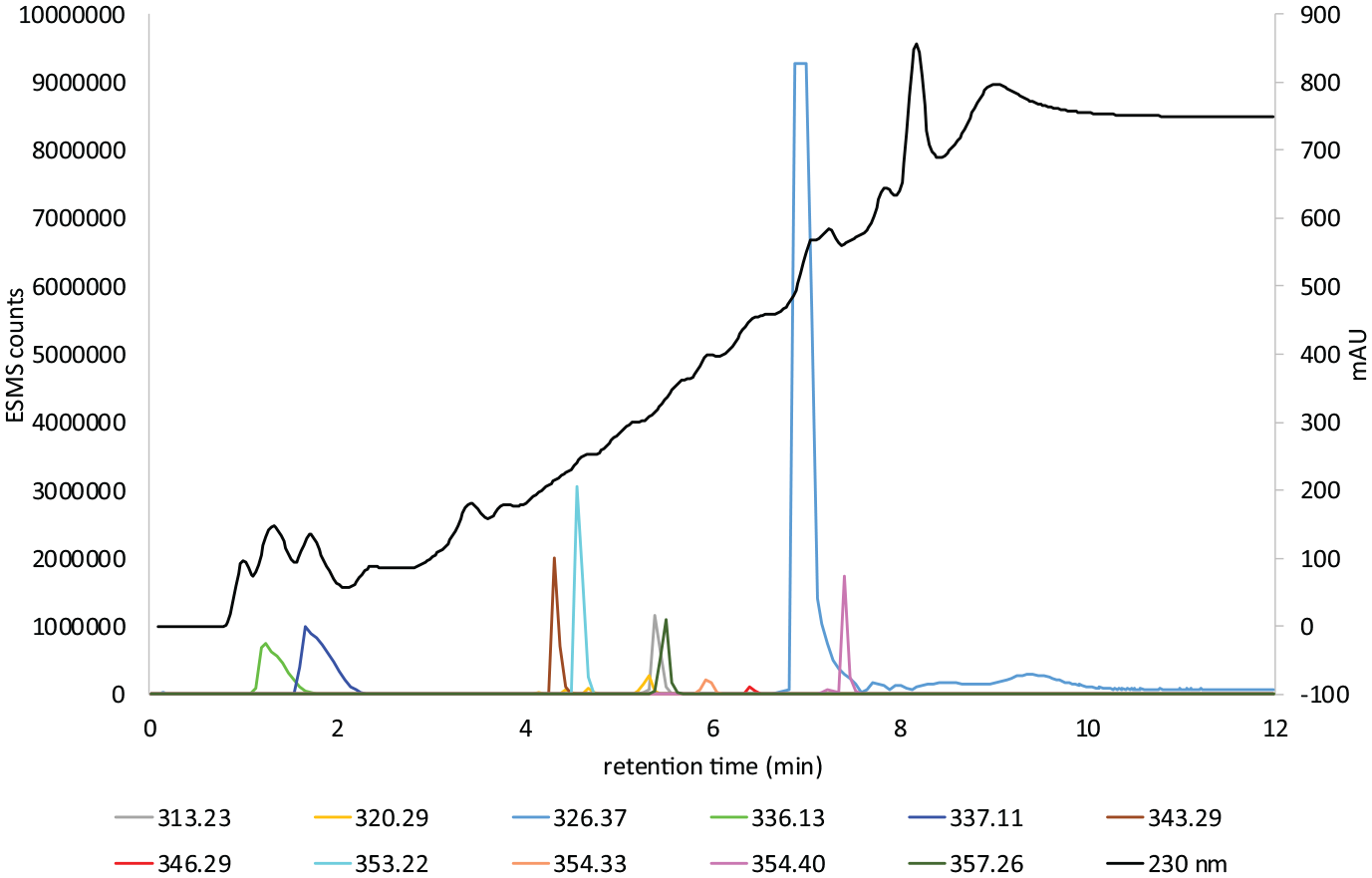
LC–MS gradient 1 chart 2 of Dark Brown colour former.
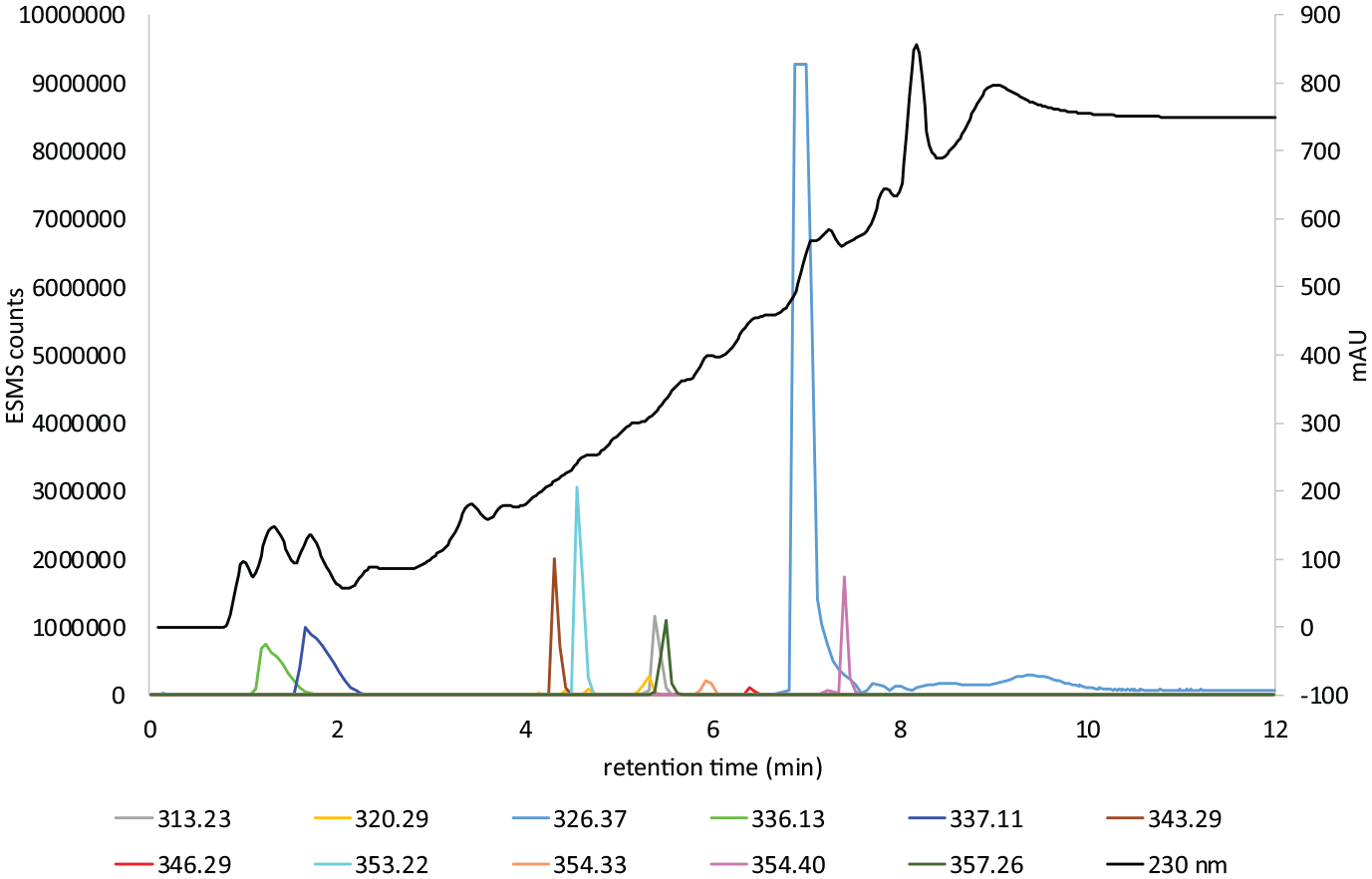
LC–MS gradient 1 chart 3 of Dark Brown colour former.
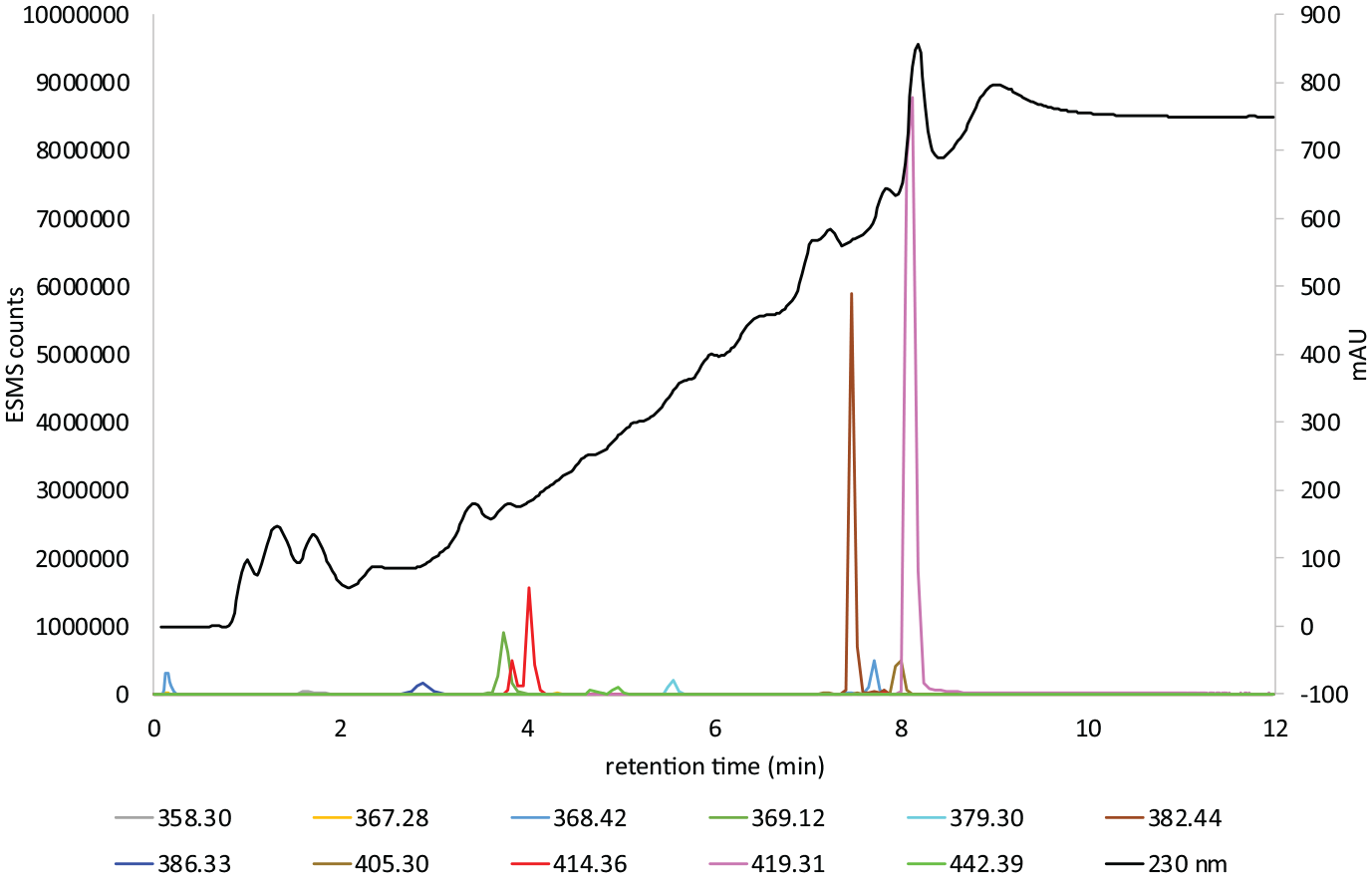
LC–MS gradient 1 chart 4 of Dark Brown colour former.
Quaternary ammonium salts identified by LC–MS analysis of coloured hair wefts extracted with dichloromethane:trifluoroacetic acid (75:25).
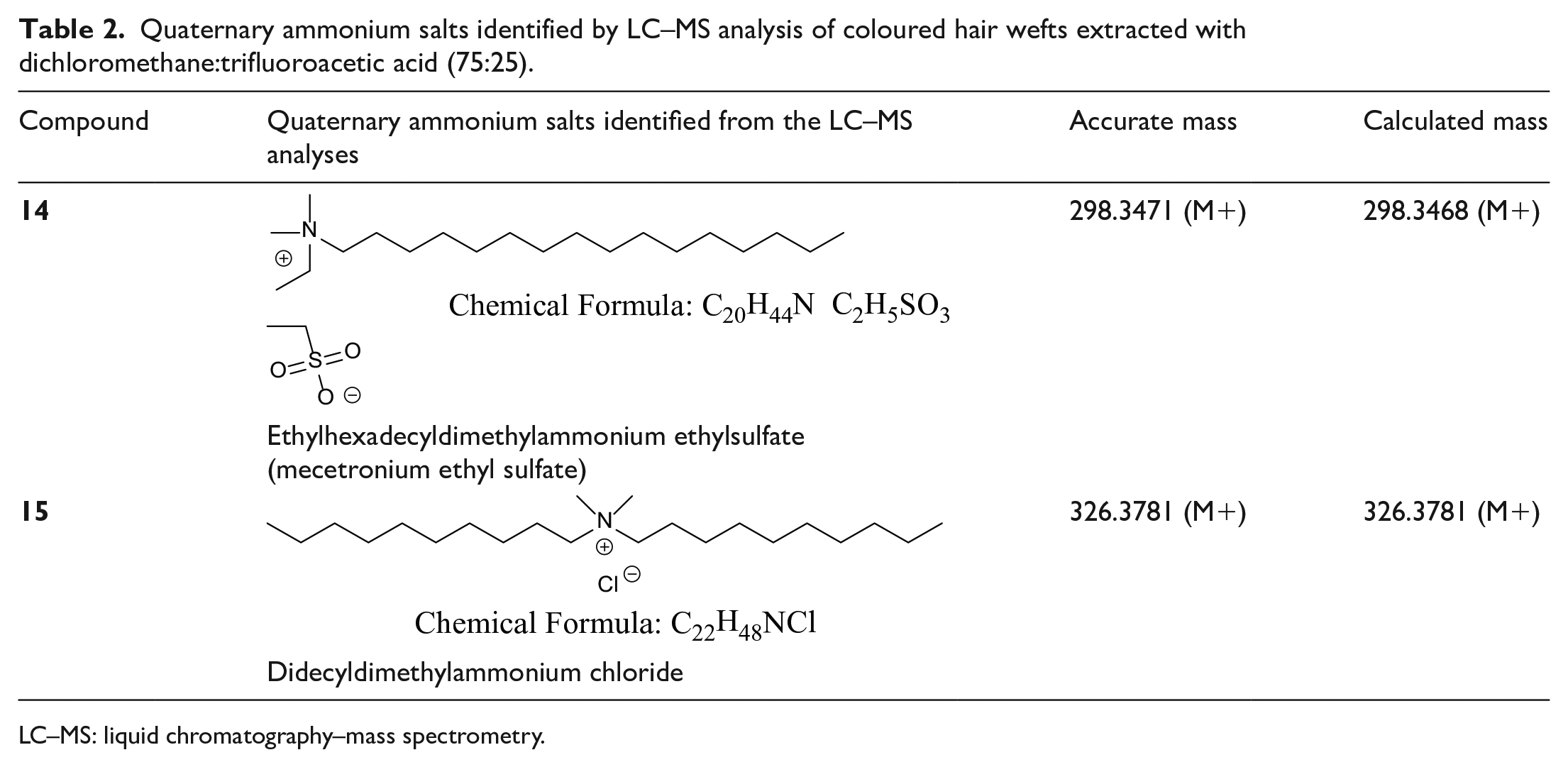
LC–MS: liquid chromatography–mass spectrometry.
Decomposition products of quaternary ammonium salts identified by LC–MS analysis of coloured hair wefts extracted with dichloromethane:trifluoroacetic acid (75:25).
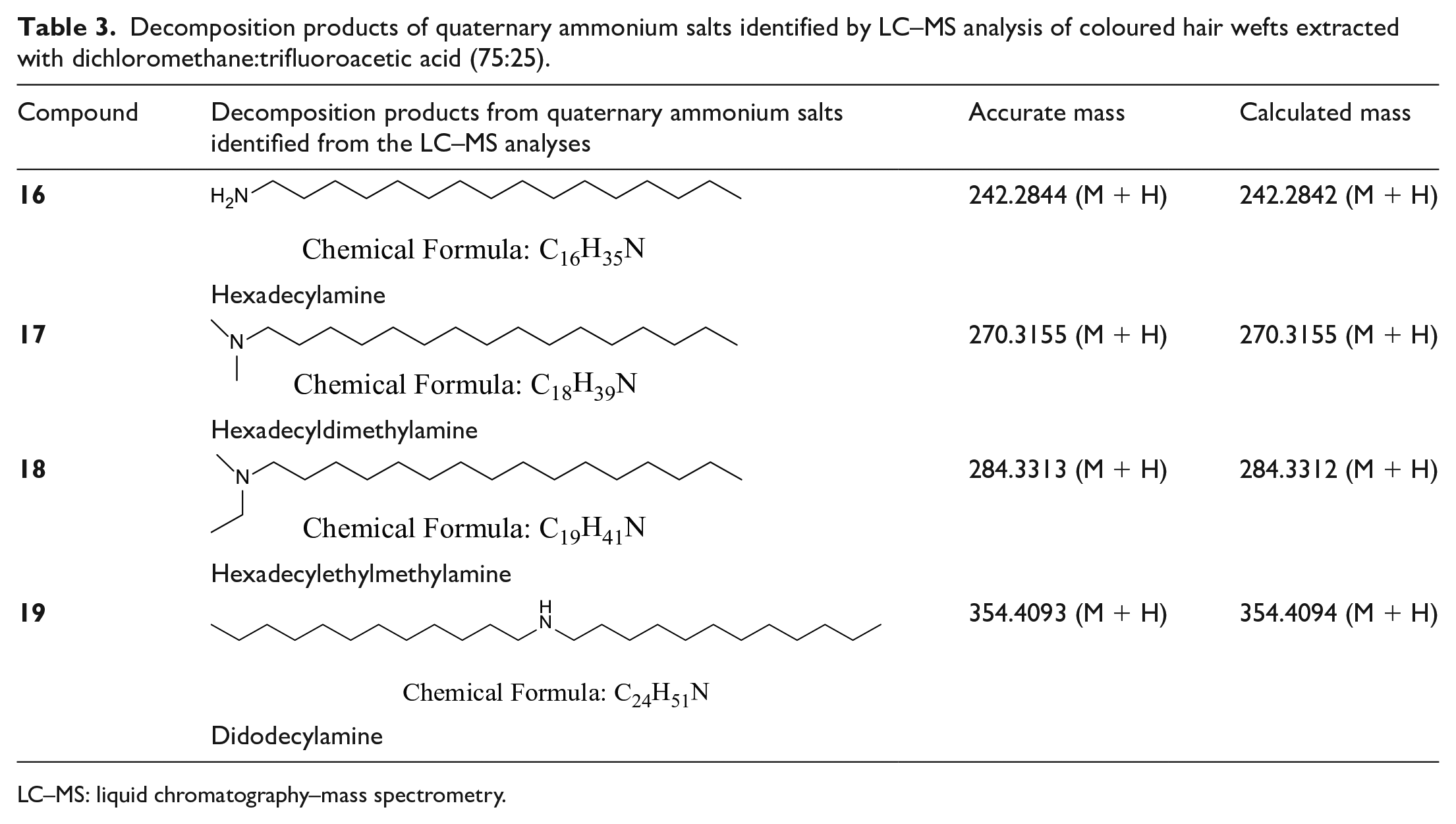
LC–MS: liquid chromatography–mass spectrometry.
Phthalates identified by LC–MS analysis of coloured hair wefts extracted with dichloromethane:trifluoroacetic acid (75:25).

LC–MS: liquid chromatography–mass spectrometry.
Miscellaneous compounds identified by LC–MS analysis of coloured hair wefts extracted with dichloromethane:trifluoroacetic acid (75:25).

LC–MS: liquid chromatography–mass spectrometry.
The strong affinity of quaternary ammonium salts for hair can be exploited for making new colour extenders.18,19 The study here verifies that the method has precedent and that a wide variety of quaternary ammonium salts and dyes might be exploited by covalent attachment together. The oxidative in situ formation of dyes, a commercial method which dominates supermarket shelves, remains a method with some toxicity owing to the toxicity of aromatic amines and the many by-products that form. Some people experience an unpleasant inflammatory reaction, so alternative cleaner and less toxic methods of fixing dyes to hair are desirable.
The common decomposition products
Phthalates were also identified as some of the components which can also be used in shampoos. Isononylphthalate
Miscellaneous compounds 1-hexadecyl-1,2-dihydropyridin-2-one
Conclusion
The dyes formed from three commercial hair colour formers have been purified by selective absorption on long human hair wefts. After washing and drying, the dyes were extracted with a non-nucleophilic and volatile solvent mixture of dichloromethane and trifluoroacetic acid. After ambient evaporation of the extractant, the extract was taken up in MeOH with no further treatment and analysed by LC–MS. Only one or two dyes, compounds
Methods
For analytical separation, an Agilent 1290 Infinity high-performance liquid chromatography (HPLC) system consisting of a quaternary HPLC pump, cooled auto sampler compartment, column compartment and diode array UV-Vis detector was used. An F5-Kintex C-18 column (2.1 mm × 150 mm, Thermo Scientific, UK) was used for the separation with an acetonitrile/methanol gradient (both 0.1% v/v formic acid) from 40% acetonitrile to 100% in 10 min with 2 min hold and then reset (gradient 1); from 5% acetonitrile to 100% in 10 min with 2 min hold and then reset (gradient 2). Data are shown in the Supplemental section (Supplemental Figures S18–S36). The flow rate was 0.5 mL min−1, the column temperature was 40 °C, and the sample volume was 5 μL. The mass spectrometer (ES-MS) used was a MaXis II UHR-TOF LC–MS System (Bruker UK Ltd, UK) with electrospray ionisation (ESI) source connected to the UV-Vis detector by a short length of PEEK tubing. The ES-MS was operated in positive ion mode with a capillary voltage of 4.5 kV using sodium formate clusters for calibration and methyl stearate as lock mass. Mass spectra were recorded automatically. Long blonde human hair wefts (15 inches) from China were purchased from Remy and were used for dyeing in accordance with the relevant guidelines and regulations of the University of Aberdeen Health and Safety committee.
LC–MS analysis of the dyes extracted from hair wefts dyed from Honey Blonde, Light Brown Warm and Dark Brown commercial colour former packets and a control
The experimental method was much the same as that used with the commercial hair colour former packet. An equal volume of aqH2O2 (5 mL), provided, was mixed with an equal volume of the mixture of primary intermediates and couplers (5 mL) in a beaker. For the control, pure p-phenylenediamine
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820978621 – Supplemental material for Liquid chromatography–mass spectrometry analysis of dyes formed by in situ oxidative methods then purified by absorption and extraction from hair wefts
Supplemental material, sj-pdf-1-chl-10.1177_1747519820978621 for Liquid chromatography–mass spectrometry analysis of dyes formed by in situ oxidative methods then purified by absorption and extraction from hair wefts by M John Plater and Andrea Raab in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are grateful to the Development Trust of the University of Aberdeen for funding.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Development Trust of the University of Aberdeen.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
