Abstract
Monolaurin is a naturally occurring compound widely utilized in food and cosmetics. In this paper, we present a new method for the synthesis of monolaurin by esterification between lauric acid and glycerol catalyzed by Novozym® 435 using a microreactor. The conversion of lauric acid is 87.04% in 20 min, compared with 70.54% via the batch approach in 5 h. Using an optimized solvent system consisting of

Introduction
Glycerol monolaurate, 1 also known as monolaurin or 1-lauroyl-glycerol (Figure 1), is a naturally occurring fatty acid that is widely utilized in food and cosmetics. It is most commonly used as a surfactant/emulsifier in food and cosmetics. It is also widely used as a dietary supplement, a food ingredient, and a feed additive because of its nutritional function, and is generally recognized as safe (GRAS) by the Food and Drug Administration. Also, monolaurin has antibacterial, antiviral, and other antimicrobial effects in vitro, and can be used as a preservative in the above products. 2
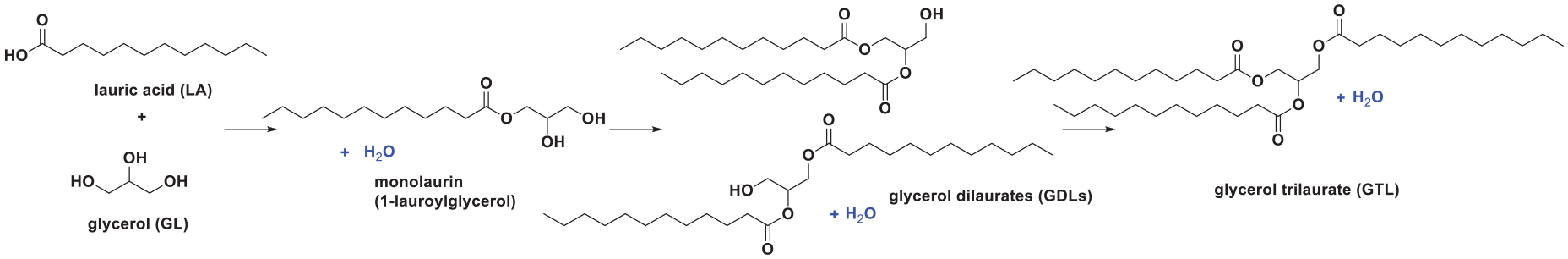
Esterification between GL and LA.
Methods to obtain monolaurin include hydrolysis/alcoholysis/glycerolysis and esterification.3–15 The reactions can be catalyzed either by enzymes or chemical catalysts. The reported chemical catalysts for esterification are
One of the most important features of microreactor technology is its highly effective mass-transfer rate. With microreactor technology, the time to reach the chemical equilibrium of an enzymatic transformation can be reduced significantly. The selectivity toward monolaurin in the reaction is affected by multiple factors, such as solvent selection, the molar ratio of GL to LA (lauric acid), and the temperature. Combining the high mass-transfer rate of microreactor technology and the high specificity of biocatalysis, the enzymatic synthesis of monolaurin was conducted in a microreactor for both high conversion of LA and high selectivity for monolaurin in a short time.
In order to synthesize glycerol monolaurin using an immobilized enzyme in a microreactor, the experimental conditions needed to be optimized. Some of the parameters such as enzyme screening, the solvent system, the temperature, and the GL/LA molar ratio can be optimized under batch methods and then transferred to a microreactor method. Other factors, which are unique parameters, such as retention time, need to be optimized with microreactors. The optimized batch and flow reactions will be compared based on conversion, selectivity, and space–time yield to demonstrate the advantages of the microreactor method for the synthesis of monolaurin.
Results and discussion
The optimization of the key parameters by esterification in batch is presented.
Enzyme screening
The catalytic activity of four enzymes, including Novozym® 435, Lipozyme®RM-IM, CalB, and Lipozyme®TL-IM, on the esterification between GL and LA was investigated to identify the best immobilized enzyme for the microreactor technique. The enzyme screening was conducted in batch without a solvent as described in the Supplemental material. The results are summarized in Figure 2. The 90.16% conversion of LA and 63.55% selectivity for monolaurin were obtained in the esterification catalyzed by Novozym® 435. The reaction catalyzed by Lipozyme®RM-IM afforded similar results. Considering the price of these enzymes, Novozym® 435 was selected as the optimum catalyst for further research.

Catalytic performance in the esterification between GL and LA using various enzymes.
Solvent screening
The appropriate solvent choice needs to be determined in order to dissolve LA and GL, and to avoid pump/microchannel blockage and inaccurate flow rates in the microreactor. At the same time, the choice of solvent is also an important factor for high selectivity toward monolaurin over the dilaurate and trilaurate products.18,19 The commonly used solvents in esterification, including

Effect of solvents on the esterification between GL and LA.
The esterification in
Temperature optimization
The optimal temperature for the Novozym® 435 was investigated for the esterification between LA and GL using the batch esterification process. The suitable temperature range for Novozym® 435 is from 30 °C to 60 °C. The temperatures investigated were 46 °C, 50 °C, 54 °C, 58 °C, and 62 °C, and the results are depicted in Figure 4. The conversion of LA and the selectivity for monolaurin reached the highest values (81.04% conversion, 79% selectivity) when the esterification was performed at 58 °C.

Effect of temperature on the esterification between GL and LA.
Reaction in flow
Flow experimental setup
Continuous flow reactions were performed using a Vapourtec fixed-bed reactor and an R-Series machine (Scheme 1).
Optimization of the flow esterification
Influence of the molar ratio of GL to LA on the flow esterification
After the key parameters of reaction had been optimized in batch, the reactions in the microreactor were conducted with a solvent system of

Effect of the GL/LA molar ratio on the flow esterification between GL and LA.
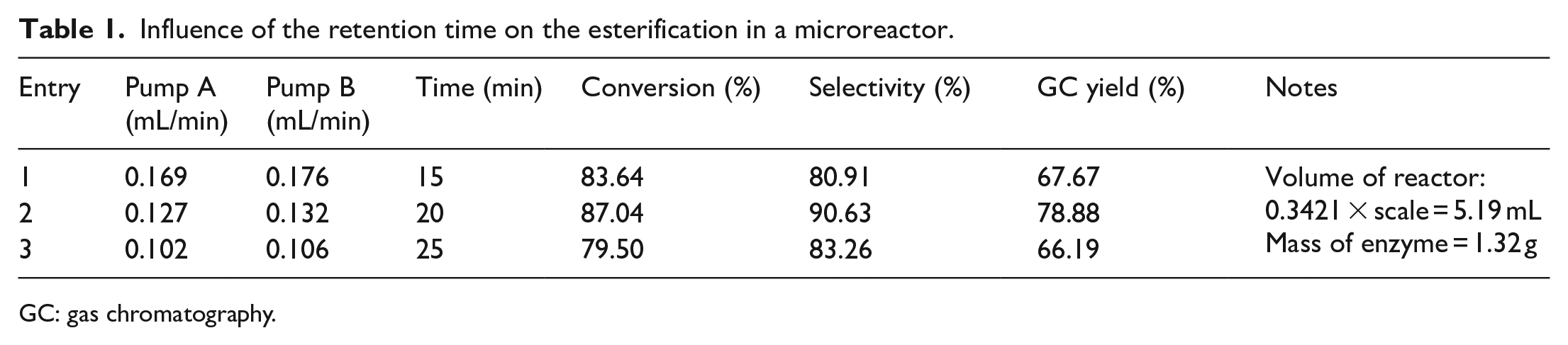
Influence of retention time on flow esterification
Furthermore, the influence of the retention time on the flow esterification was investigated. The reactions with retention times of 15, 20, and 25 min were conducted in a microreactor and the results are given in Table 1. The esterification after 20 min of residence time achieved 87.04% conversion of LA and 90.63% selectivity toward monolaurin. Hence, the optimum residence time in flow was 20 min. Also, purification of the reaction was conducted affording 52.80 g monolaurin as a white waxy solid starting from 47.52 g of LA. The yield was 81.12%.
Influence of the retention time on the esterification in a microreactor.
GC: gas chromatography.
A comparison between the reaction in batch and in a microreactor
In addition, the batch method was optimized in this study in order to compare the results with those using microreactor method. The factors to be optimized with the batch method included treatment of the water produced in the reaction, the enzyme amount, and the reaction time.
The esterification reaction between GL and LA is a reversible reaction and continuously produces water (Figure 1). Therefore, the method of handling the generated water can affect the reaction equilibrium significantly, especially in the batch method. To investigate the effect of water on the reaction in batch, three systems, including a closed system, an open system, and a 4 Å molecular sieves system, were designed. The highest conversion of LA and selectivity toward monolaurin was achieved using the open system. For the closed system, accumulation of water will have an adverse effect on the conversion of LA. For the 4 Å molecular sieves system, removing the produced water would be beneficial for the esterification process but also resulted in low selectivity for monolaurin. The conversion of LA was 87.59% and the selectivity for monolaurin was 72.39% in the open system for the batch method. Open system was thus selected for the batch method.
The influence of the enzyme amount on the esterification in the batch method was examined by varying the enzyme amount from 1% to 9% (relative to LA + GL) and the optimum enzyme loading was set as 3 wt% (relative to LA + GL) where the conversion of LA was 73.81% and the selectivity for monolaurin was 88.1%.
The reaction time for the batch method was also optimized. The results indicated that the reaction achieved equilibrium after 5 h, which afforded 81.06% of LA conversion and 81.13% of selectivity for monolaurin from 2 g of LA under the optimum conditions in batch.
Finally, the esterification between GL and LA was conducted in batch, with the optimum conditions as follows: GL/LA, 5:1 (mol/mol); reaction temperature, 58 °C; reaction time, 5 h;
The comparison between the reaction in batch and in a microreactor is shown in Table 2. The space–time yield of the reaction in the microreactor was 380.91 g/h/L. With 30 g of starting material, the space–time yield of the reaction in batch was 10.44 and 47.09 g/h/L with and without solvent, respectively.
Comparison between the reaction in batch and in a microreactor.

GL: glycerol; LA: lauric acid; GC: gas chromatography.
Temperature: 58 °C, Solvent:
Product quantity/time/enzyme, quantity of product produced per unit time per amount of enzyme.
2.97 g Novozym® 435.
With esterification starting from 30 g of LA in 500 mL batch in 5 h.
1.79 g Novozym® 435.
1.32 g Novozym® 435.
1.32 g Novozym® 435, running for 12 h.
1.32 g Novozym® 435.
1.32 g Novozym® 435, running for 10 h.
With the space–time yield of the reaction in the microreactor being one order of magnitude higher than that of the batch method, the high effective mass transfer in the microreactor is obvious. The larger specific surface area in the microreactor enhanced the efficiency of the mass transfer between the enzyme and the reactants. The ratio of the enzyme/starting material in the system was very high. So, the apparent reaction rate was improved significantly. However, the quantity of product produced per unit time per amount of enzyme was similar. This was caused by the small volume of the microreactor. Therefore, keeping the reaction running for a long time to overcome the disadvantage was a common solution. As shown in Figure 6, with the microreactor method, the reactant went through the column reactor and creates a concentration gradient of the product, namely, monolaurate, dilaurate, and trilaurate. The concentration of these products will be the lowest at the entrance and the highest at the exit of the column. In the meantime, the dilaurate and trilaurate concentrations will depend on the monolaurate concentration since these are the products of subsequent reactions. This concentration gradient will favor the reversible esterification reaction toward producing monolaurate at the entrance of the column. With an appropriate retention time selection, monolaurate will leave the column with little conversion to the dilaurate/trilaurate. Thus, optimization of the retention time will improve both the conversion of LA and selectivity for monolaurin. This helps to explain the results of the retention time experiment for the microreactor method. A 15-min retention time did not provide sufficient time for the esterification reaction before the reactants exited the column, thus resulting in lower conversion and selectivity compared to that of the 20 min retention time experiment. The 25 min retention time experiment also resulted in a lower conversion and selectivity compared to that of the 20 min retention time experiment, as a longer retention time leads to more tendency to reverse the esterification reaction, or toward dilaurate and trilaurate. Hence, the retention time of 20 min was the optimum time for monolaurin.
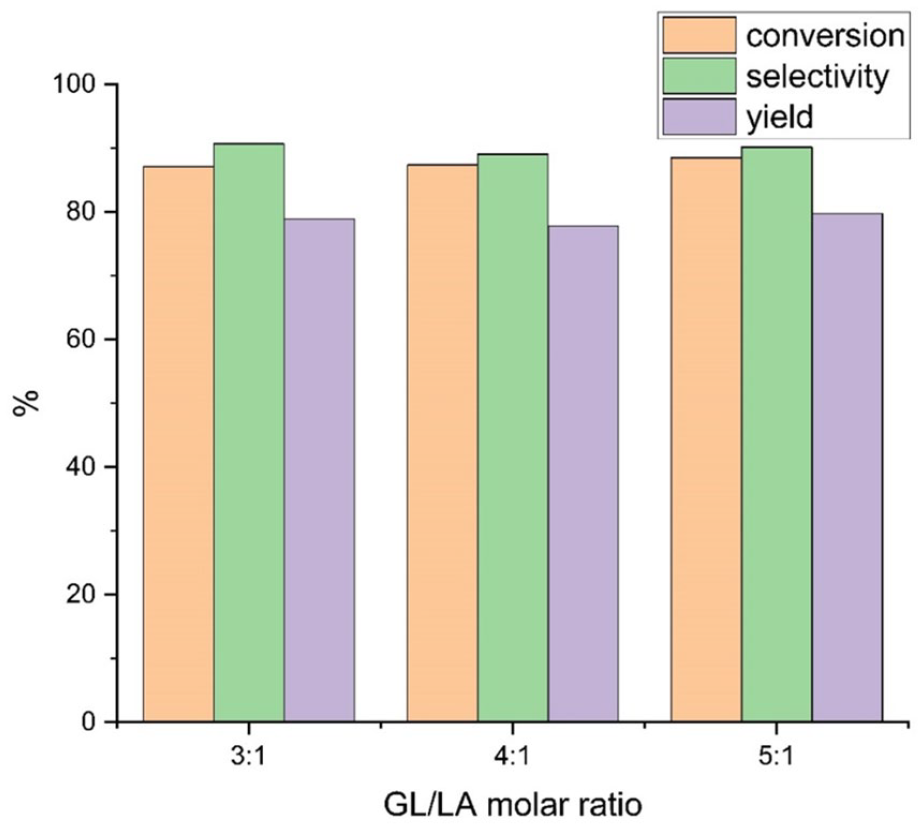
Illustration of the production of glyceryl laurate esters in the microreactor.
Conclusion
A new method for the synthesis of monolaurin in a microreactor has been reported for the first time. Commercially available Novozym® 435 was used as the catalyst in the esterification reaction of LA and GL in a medium of
With the microreactor method, the esterification was significantly accelerated, and under optimized conditions, 87% conversion of LA was reached and the retention time was reduced to 20 min compared with 5 to 6 h in batch. The reason for the improvement was due to the highly effective mass transfer in the microreactor, which can decrease the mass-transfer limitations of the enzymatic reaction. In the meantime, the selectivity toward monolaurin was also enhanced to 90% by using a solvent system of
The process in the microreactor could be maintained and kept stable for 12 h, thus allowing a repeatable synthesis of monolaurin. The space–time yield of the process in the microreactor was 380.91 g/h/L, compared with 10.44 ~ 47.09 g/h/L in batch. This showed that this process has good potential in industrial applications for preparing monolaurin. This new method described in this report may serve as an inspiration for applications to other multiphase and enzymatic reactions.
Experimental
Materials and methods
Novozym® 435, Lipozyme RM-IM, Lipozyme CalB, and Lipozyme TL-IM were purchased from Novozymes (Beijing, China). Monolaurin (Analytical Reagent (AR), 99%) was purchased from Sigma-Aldrich. LA (AR, 98%),
Using the standard curve methods, the quantitative determination of LA and monolaurin was performed via gas chromatography (GC, Agilent 7890A) equipped with a flame ionization detector. The calibration curves for LA and monolaurin were obtained by using LA (AR, 98%) and monolaurin (AR, 99%) as standards. The actual content was calculated by the equation “mass × purity” (mass: the number shown on the scale). The procedural details for generating the calibration curves are given in the Supporting Information. The reactant mixture (200 µL for LA determination and 50 µL for monolaurin determination) was added into a 20 mL volumetric flask and immediately made up to the mark with 1:1 hexane/ethyl acetate solvent mixture. The above solution (1 µL) was injected into an Agilent 7890A GC system (HP-5 Column, 30 m × 0.320 mm × 0.25 µm), with helium as the carrier gas at a flow rate of 2 mL/min. The detector and injector temperatures were set at 330 °C. First, the column temperature was set at 80 °C and kept for 1 min, then increased to 320 °C with a rate of 20 °C/min and kept for 2 min. The concentrations of LA and monolaurin in the samples could be calculated via the standard curve. From GC results, the yield of monolaurin could be calculated from equation (1). The “yield” is also called “GC yield” on assuming no product loss during the purification. The “yield” shown in all tables and schemes is the “GC yield”

Furthermore, the conversion rate of LA can be calculated from equation (2). The remaining LA was determined by GC using the calibration curve method

Finally, the selectivity was calculated from equation (3). The selectivity for monolaurin was defined as the ratio of the formed monolaurin (in moles) with respect to the converted starting material (in moles)

Synthetic procedures
Batch esterification process in solvent
GL (1.38 g) and LA (1.00 g) were added to the appropriate solvent (6 mL, 0.397 g/mL) in the 15 mL batch and were kept at the selected temperature using a thermostatic water bath, followed by the addition of lipase Novozym® 435. The catalyst amount (relative to LA + GL) was modified during the research. The reactions were carried out for the appropriate period of time (5–6 h) in an oil bath under continuous stirring conditions. The stirring rate was 350 r/min. For kinetic research, 200 µL aliquots were withdrawn every 1 h and analyzed by GC. For optimization, the reaction was quenched by adding 0.5 mL of acetone/ethanol (1:1) into the reaction mixture and 200 µL aliquots were withdrawn and analyzed by GC.
The purification of final product
The purification was conducted after the reaction under optimized reaction conditions (15 min, 58 °C, GL/LA 3:1) kept running for 24 h starting with 47.52 g LA. First, saturated sodium bicarbonate and ethylacetate (EtOAc) were added into the reaction mixture to remove the remained GL and extract the esters. Vacuum rotating distillation was done after workup, affording the final product as a white waxy solid (52.80 g). The 1H NMR and 13C NMR were determined and afforded in the Supporting Information.
Flow esterification process
All experiments under flow conditions were carried out using a Vapourtec fixed-bed reactor and an R-Series machine. As shown in Figure 7, the solution of GL was pumped through the orange pump (Pump A), and the solution of LA in solvent was pumped through the purple pump (Pump B). The reactant mixture passed through the column reactor filled with Novozym® 435. The inner diameter of the column reactor was 66 mm. The reactor volume was calculated from equation (4). The reaction details are listed in Table 3. All the reactions in the microreactor were run for 12 h. The sample collection was performed after 1 h, and the collection was continued for 2 to 12 h using 10 mL tubes. Six of the sample tubes were detected to obtain the mean values of the conversion and selectivity. In our work, the longest running time was 12 h. The formation of fine particles of the immobilized catalyst was noted after use. However, we believe the immobilized catalyst can be used for a longer time without blockage. The volume of the reactor was according to the manufacturer settings


Scheme of enzymatic synthesis of monolaurin by esterification between GL and LA in a microreactor.
Esterification in the microreactor.

GL: glycerol; LA: lauric acid.
GL in
LA in
The mass percentage of enzyme in the whole reactant mixture when the reaction was kept running for 12 h.
The total mass of enzyme filled in the fixed-bed reactor.
Calculation of the yield and space–time yield
The space–time yield was calculated as follows

unit: g/(h·L)

unit: g/(h·L)
The product quantity per time per enzyme, and the quantity of product produced per unit time per amount of enzyme is calculated as

The monolaurin produced in the study
The monolaurin produced in the study was confirmed to be 1-lauroyl-glycerol by 1H NMR and 13C NMR methods as shown in the Supporting Information. It was in good agreement with the reported data.
20
Also, a comparison between GC of 1-lauroyl-
Calculation of the residence time
The residence time was calculated from equations (8) and (9)


The porosity of Novozym® 435 we used was 98%. The residence time shown in the paper is the theoretical residence time

The number 0.3421 was the manufacturer setting, which is labeled on the tub. Scale is the bed length, which indicates where the catalyst was filled to, and is shown in the Supplemental material.
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820977164 – Supplemental material for Enzymatic esterification of lauric acid to give monolaurin in a microreactor
Supplemental material, sj-pdf-1-chl-10.1177_1747519820977164 for Enzymatic esterification of lauric acid to give monolaurin in a microreactor by Shanshan Miao and Xin Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by National Key Research and Development Project of China (2019YFD1101200) and the Jiangsu Synergetic Innovation Center for Advanced Bio-Manufacture (XTE1852).
Supplemental material
Procedures for determining the calibration curve for lauric acid (LA) and monolaurin, 1H NMR and 13C NMR of monolaurin are supplied as Supporting Information.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
