Abstract
Two coordination polymers with two-dimensional and three-dimensional structures are, {[Zn3(bdc)3(py)2]·2NMP}n (

Two coordination polymers were synthesized by hot-solution reactions. The dimensions of two coordination polymer structures were changed from two-dimensional to three-dimensional with transformation of ligand geometry.
Introduction
In recent years, reasonable design and controllable syntheses of metal-organic coordination polymers have gained a significant interest due to their important application prospects.1–5 During the controlled synthesis of coordination polymers, some important factors such as the reaction temperature,6,7 the solvent species,8,9 the ligand geometry, 10 the pH of the solution11,12 and the presence of ancillary ligands13,14 can have profound effects on the crystal structures and self-assembly properties. In addition, the geometry and substituents on the ligands15–17 can change the frameworks of coordination polymers, and the shape and size of the cavities. With respect to stable network framework, ligands with different configurations will display multiple spatial orientations when they are coordinated to metal ions and accordingly will generate diverse structures. For ligands with elastic structures,18,19 their spatial configuration is prone to distortion when they occupy different coordination environments. However, rigid ligand configurations20,21 cannot usually be deformed in different reaction media or different coordination modes. The angle between the functional groups on the ligand will influence the spatial orientation of coordination polymerization, with different network structures generally being controlled by the steric aggregation structures of the ligands. Therefore, a change of spatial structure of the ligand will result in an obvious transformation of the coordination polymer structure in three-dimensional space. Furthermore, ancillary ligands may play an extremely important role on the process of structural assembly between metal ions and ligands, with examples including nitrogen donor ligand such as ammonium salts, pyridine, pyrazole, and pyrazine.22–24 Solvent molecules, such as H2O and C2H5OH demonstrate a stronger coordination ability compared to a carboxyally oxygen atom, or preferentially occupy the coordination vacancies due to metal ions.
We have systematically investigated the influence of the reaction temperature, mixed solvents, and ligand substituents on the crystal structures of various products.15,25,26 In this paper, we have synthesized two coordination polymers with two-dimensional (2D) and three-dimensional (3D) structures by hot-solution reactions of Zn(NO3)2·6H2O, pyridine (py), and two different ligands in NMP. The two products, {[Zn3(bdc)3(py)2]·2NMP}n (
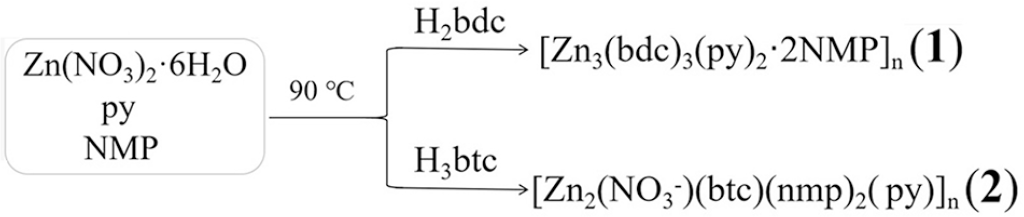
The formation of coordination polymers
Results and discussion
Descriptions of the crystal structures
[Zn3(bdc)3(py)2·2NMP]n (

(a) The trinuclear zinc subunits, and (b) the 2D (3, 6) framework of complex
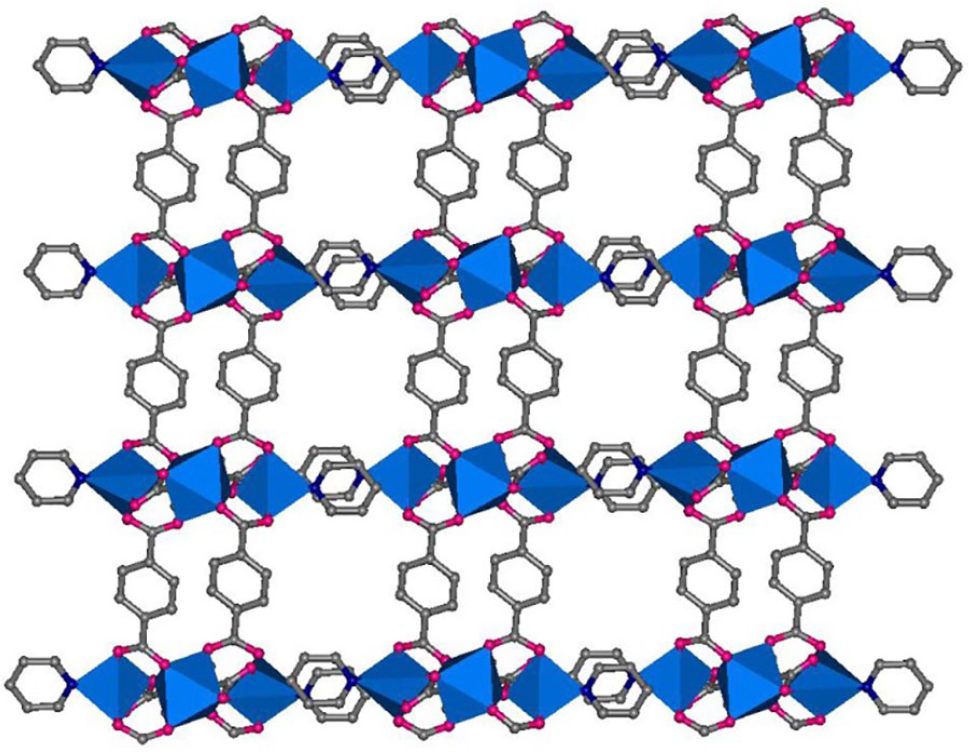
2D view of the (3, 6) framework of
[Zn2(NO3−)(btc)(nmp)2(py)]n (

(a) Connection between two zinc ions Zn1 and Zn2, and (b) linking of three carboxylic units from the btc ligands.

A perspective view of the 3D framework of
The different geometries of the ligands, that is, line-type and triangle-type, result in the different 2D and 3D structures of products
Comparison of the structural characteristic of the three coordination polymers.

Conclusion
In summary, we have successfully synthesized two coordination polymers
Experimental procedures
Materials and methods
All chemicals employed in the reaction process were analytical grade, and were directly used without further purification. Infrared spectra were obtained on a Nicolet AVATAR FTIR 360 spectrometer as KBr presser. Elemental analysis was performed on a CE instruments EA-1110 elemental analyzer.
Syntheses
[Zn3(bdc)3(py)2·2NMP]n (1 )
H2bdc (1 mmol, 166 mg,)and Zn(NO3)2·6H2O (1 mmol, 298 mg) were added to a 25-mL conical flask, then NMP (8 mL) was added and the mixture was stirred vigorously. Next, py (0.5 mL) was added dropwise to the reaction mixture. The mixture was heated from 30 °C to 90 °C over 5 h and then kept at 90 °C for 72 h. Finally, the temperature was slowly reduced to room temperature over 10 h through programmed temperature control. Colorless block crystals (0.165 g, 47%) were obtained. Crystal suitable for X-ray diffraction were separated by filtration, washed with distilled water, and dried in air. Anal. calcd for Zn3C44H36N4O14: C, 50.57; H, 3.48; N, 5.38; found: C, 49.89; H, 3.39; N, 5.17%. FTIR (KBr, cm−1): 3449 (w), 2978 (w), 2927 (w), 1591 (vs), 1404 (vs), 1346 (m), 1285 (w), 1192(w), 1037 (m), 919 (m),847 (m), 847 (s),797 (vs), 764 (s), 626 (w), 552 (m).
[Zn2(NO3−)(btc)(nmp)2(py)]n (2 )
Compound
X-ray crystallography
Crystal data collection was carried out on Bruker SMART Apex CCD diffractometer with graphite monochromated MoKα radiation (λ = 0.71073 Å). Absorption corrections were applied using the multi-scan program SADABS. 28 The structures were solved by direct methods, and non-hydrogen atoms were refined anisotropically by least-squares on F2 using the SHELXTL 29 and SHELXL-97 30 program packages. Crystallographic data, as well as details of data collection and refinement for the two compounds, are summarized in Table 2, and selected bond lengths and angles for the two crystal structures are listed in Table 3.
Crystal data and details of the data collection and refinement for complexes

Selected bond lengths and angles for the two coordination polymers (in Å and °).

CCDC number 969395 and 969396 contain the supplementary crystallographic data for
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge financial support from the National Natural Science Foundation of China (21471125), the Opening Project of PCOSS, Xiamen University (No. 201921), the Excellent Talent Foundation of Education Department of Anhui Province (No. gxyq2018055), the Natural Science Foundation of the Education Department of Anhui Province (Nos KJ2019A0731 and KJ2020A0096), and the fifth batch of “special branch plan” projects in Anhui Province.
