Abstract
Ir and Rh “PNP” complexes with different ligands are utilized for the oxidation of n-octane. Based on the obtained conversion, selectivity, and the characterized recovered catalysts, it is found that the combination of Ir and the studied ligands does not promote the redox mechanism that is known to result in selective formation of oxo and peroxo compounds [desired species for C(1) activation]. Instead, they support a deeper oxidation mechanism, and thus higher selectivity for ketones and acids is obtained. In contrast, these ligands seem to tune the electron density around the Rh (in the Rh-PNP complexes), and thus result in a higher n-octane conversion and improved selectivity for the C(1) activated products, with minimized deeper oxidation, in comparison to Ir-PNP catalysts.

Introduction
The activation of alkanes is a desirable but challenging reaction due to the saturated carbon–hydrogen bonds having relatively inert character. In addition, the activated products are often more reactive than the alkane substrate, which may lead to over-oxidation of the products. There is a current need and economic urgency to replace current petrochemical feedstocks (olefins) with easily accessible alkanes, which can result in more proficient use of energy and efficient strategies for fine chemical synthesis.1–10 Terminally oxidized hydrocarbons are valuable starting materials in the pharmaceutical and chemical industries. 11 However, one of the main challenges in the activation of alkane C–H bonds is the low selectivity in forming the desired products, that is, the preferential activation of sp2 over sp3 hybridized C–H bonds.12–14 Furthermore, the chemical inertness of alkanes, together with their high ionization energy, pKa values, and low electron affinity, makes activation of C–H bonds difficult.11,13,15–17
To overcome such problems, there has been a rapid development of catalytic systems inspired by the biological systems of cytochrome P450 and methane monooxygenase.18–27 These enzymes have the ability to efficiently catalyze a number of exogenous and endogenous organic compounds, such as medium chain alkanes, to large molecules including triterpenes, as well as steroidal, polyaromatic compounds, and, more importantly, oxygenates.28–36 Site selective C–H activation by iron complexes has been reported by White and co-workers in trying to mimic enzymes.37–41 These catalytic processes are carried out by a variety of oxidants, namely, PhIO, NaOCl, H2O2, alkyl hydroperoxides, percarboxylic acids, and molecular oxygen.29,34,42–49
The design of a suitable ligand system is one way of achieving selective activation of paraffins. These systems could include the aminodiphosphine or PNP ligand system.50–58 These hybrid ligands, which consist of both soft (N) and hard (P) donors, are of particular interest in that they are part of a system that can display high activity, stability, and variability.57–62 By modifying the ligand backbone through substitution of the donor atoms, the activity of the metal can be tailored.63,64 These ligands may bind to the metal centers as either monodentate, bidentate, or as bridged ligands, which allow the reactions of the metal ions to be selective due to the high demand the ligands place on the stereochemistry of the complex. 65
In general, phosphine-based ligands are limited in their application in oxidation reactions due to ligand loss and degradation. 66 Ruthenium-based phosphine complexes have been studied for the oxidation of n-octane; however, low conversions and over-oxidation are prevalent.67,68 The activation of alkane C–H bonds by rhodium and iridium complexes has not been thoroughly explored. However, Nomura and Uemura 69 have used rhodium compounds such as Rh3O, [Rh(acac)3], [RhCl(CO)2]2, [RhCl(PPh3)3], and [Rh2(OAc)4] for the oxidation of cyclohexane using peracetic acid, H2O2, TBHP (t-butyl hydroperoxide), and m-CPBA (meta-chloroperoxybenzoic acid). 69
Considering the worldwide increase in gas to liquid plants, there is an increase in the production of medium- to long-chain paraffins, which increases the need to functionalize these hydrocarbons.70–72 In continuation of our previous work on metal complexes containing aminodiphosphine ligands for n-octane oxidation,
73
this study focuses on the application of iridium (

General reaction scheme and the structures of the complexes used as catalysts for the oxidation of n-octane. 74
Catalyst preparation
The complexes
Results and discussion
Oxidation of n-octane
The optimization of the substrate to oxidant ratio
Our recent study on the application of these catalysts showed that these two families of complexes follow similar chemistry for styrene oxidation.
74
Therefore,
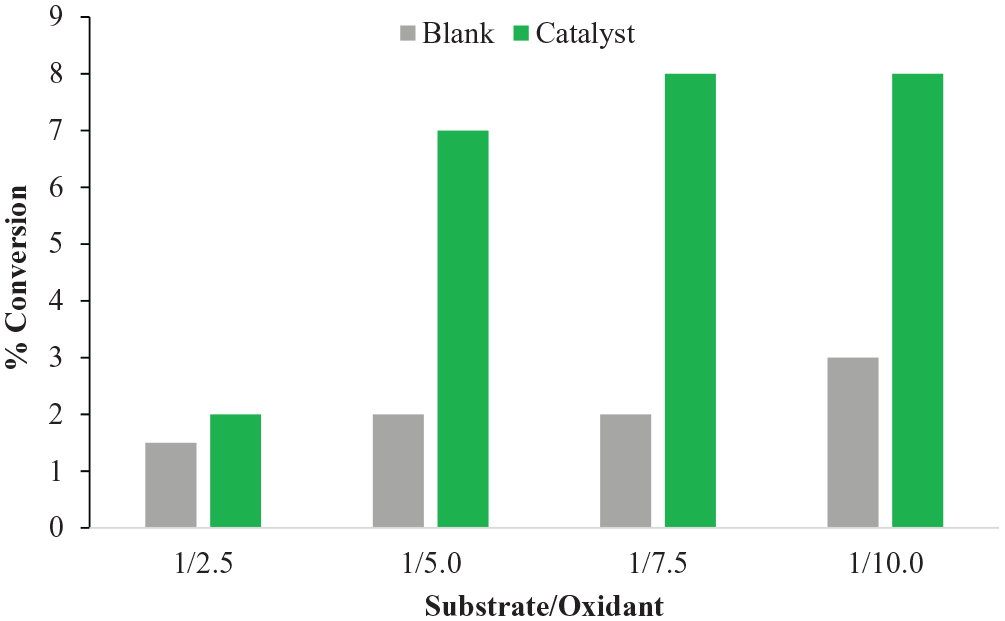
Optimization of the substrate/oxidant ratio in the oxidation of n-octane.
Temperature optimization
Before optimizing the temperature for this reaction, another popular solvent [dichloroethane (DCE)] was tested for this reaction at the same temperature as for the optimized conditions in MeCN. Interestingly, a better catalytic performance was observed that might be linked to the better solubility of the substrates in this solvent in comparison to MeCN. Thereafter, the temperature optimization was performed using DCE at 25, 50, and 80 °C (Figure 3) with a catalyst/substrate molar ratio of 1:100 and a substrate/oxidant molar ratio of 1:5. At 25 °C, octanoic acid was the only product that formed with a 1% conversion of n-octane. At 50 °C, a 4% conversion of n-octane was noted. The highest conversion (9%) was obtained at 80 °C with good selectivity for the primary activation products (C1 activation). This also could be attributed to the solubility of the oxidant in the solvent. Similar studies carried out in alkene oxidation showed better conversion in chlorinated solvents.75,76 It has been shown that some catalytic reactions proceed more efficiently in a mixture of solvents; 77 however, when a 1:1 ratio of DCE/MeCN was used, the reactions with and without the catalyst were comparable.

Optimization of the temperature in the oxidation of n-octane.
Oxidants
Hydrogen peroxide and m-CPBA (meta-chloroperoxybenzoic acid) were also investigated for this reaction. However, the use of these two oxidants decomposed the catalyst, and hence poor catalytic performances were achieved. This finding is in agreement with reports that some catalysts are not able to activate certain peroxides, resulting in a low selectivity of the desired products.78–80
Metal
Using the optimum conditions (catalyst/substrate molar ratio of 1:00, substrate/oxidant molar ratio of 1:5 at 80 °C in DCE), screening of the iridium (
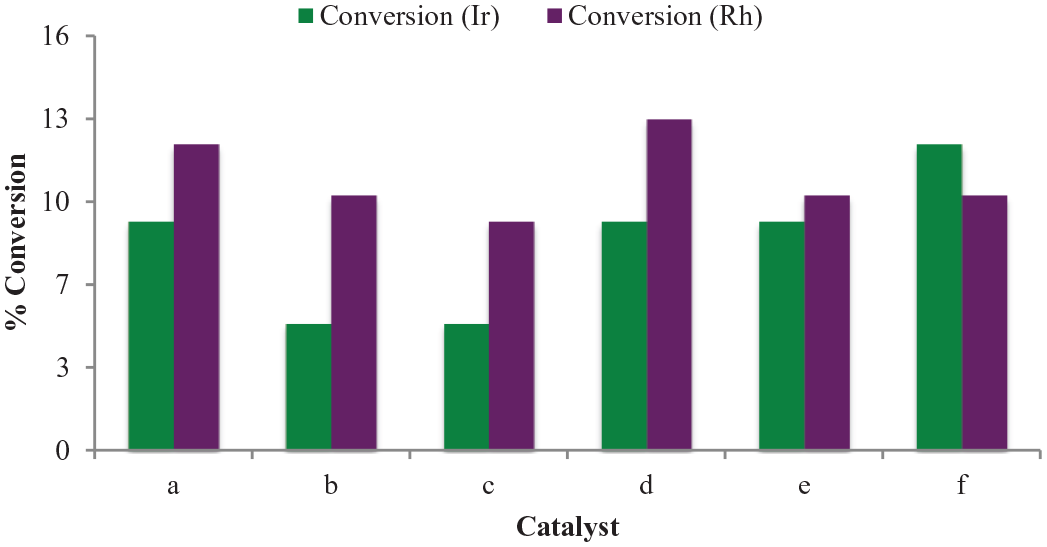
Screening of the iridium (Ir) and rhodium (Rh) catalysts in n-octane oxidation with the different substituents on the nitrogen atom (
Comparing the catalysts with the alkyl substituents on the nitrogen atom (
The selectivity for the products of oxidation by the Ir catalysts is shown in Figure 5. Deeper oxidation to the ketonic product is prevalent, as seen by the high selectivity for the octanones (59%–77%) for all the Ir-based catalysts. The extent of the oxidation is referred to as depth here. So, deep oxidation refers to the catalyzed reactions that yield highly oxidized products like the acid. Secondary or moderate oxidation (deeper oxidation) is a relative term, meaning if it is used in the case of alcohols, it refers to ketones (deeper oxidation products). When applied to ketones, it refers to acids (deeper oxidation).
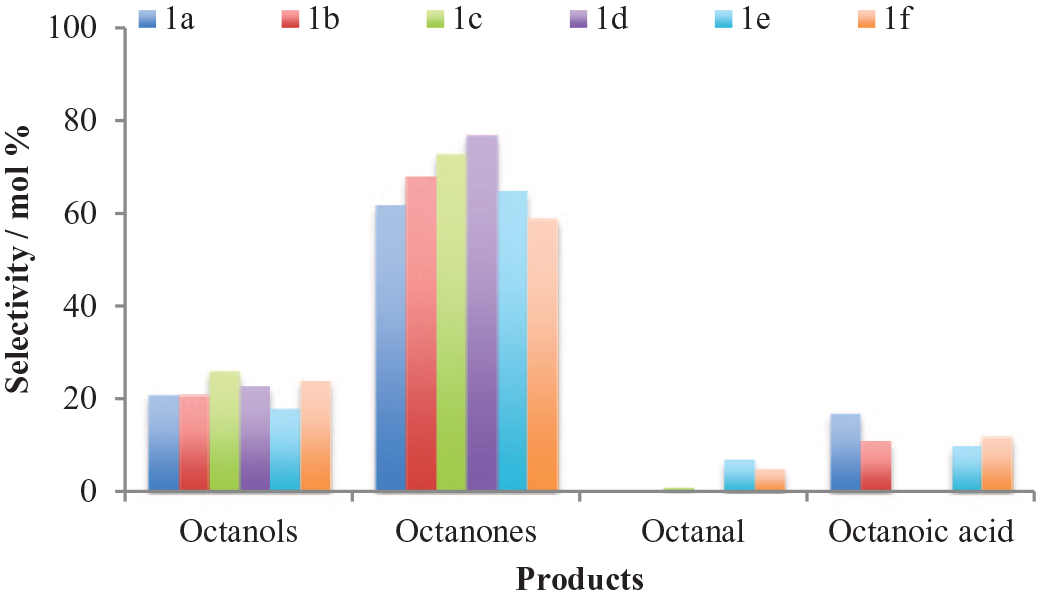
Selectivity for the products of oxidation of n-octane by the Ir (
However, the octanols are the second major products with selectivities of 18%–26%. The C(1) position of the n-octane chain is the least activated, with the C(2) and (C3) positions being the most activated carbons. For the catalysts with ligand backbones containing substituted phenyl groups (
The deep oxidation process (ketone formation) is much slower with the rhodium catalysts in comparison to the Ir catalysts (Figure 6), which is noted by the higher selectivity for the octanols (11%–41%), with catalyst

Selectivity for the products of oxidation of n-octane by the Rh (
Selectivity parameters for the Ir and Rh catalysts in the oxidation of n-octane. a

Conditions: catalyst/substrate (1:100); substrate/oxidant (1:5); solvent: DCE; temperature: 80 °C.
Parameters C(1):C(2):C(3):C(4) are the relative reactivities of the hydrogen atoms at carbons 1, 2, 3, and 4 of the n-octane chain.
The calculated reactivities from the % selectivity are normalized, that is, calculated taking into account the number of hydrogen atoms at each carbon.
It includes the % selectivity of octanoic acid, octanal, alcohols, and ketones and the values are normalized.
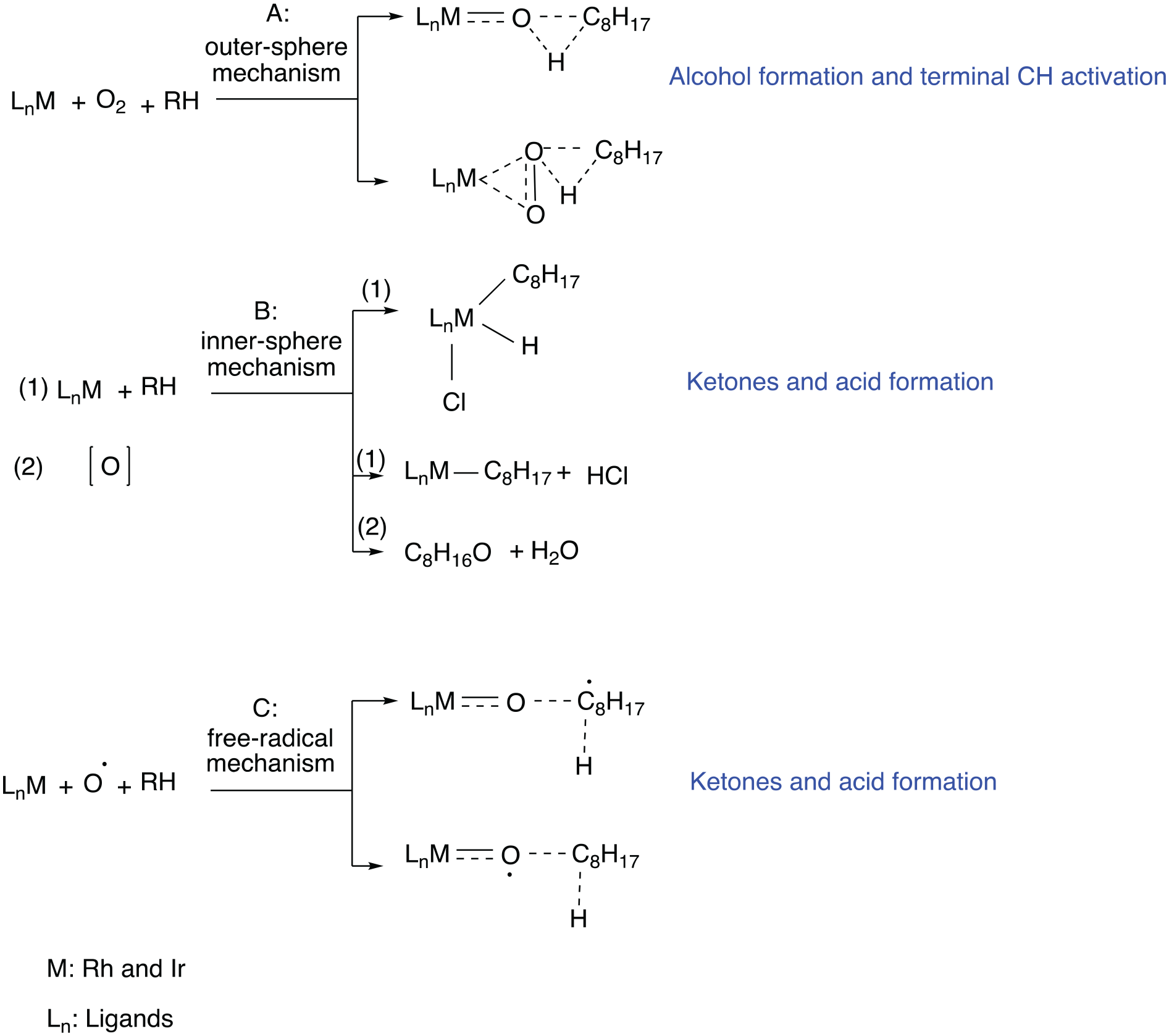
According to the recent work by Chepaikin et al.,
85
oxidative homogeneous catalysis of alkanes can follow two mechanisms. Mechanism A is referred to as an outer-sphere mechanism (A), where the reaction takes place using the in situ generated oxo and peroxo metal complexes. This reaction mechanism is known to yield alcohols (Scheme 1). In contrast, the inner-sphere mechanism (B) happens mainly with catalysts that contain Cl with M-alkyl radical intermediate formation, and results in the deep oxidation of alkanes to ketone and acids. Also, the free-radical mechanism (C) is known to result in deep oxidation and forms products such as ketones and acids as discussed for mechanism (B).
86
It is important to note that in the majority of cases, the latter two mechanisms dominate. However, generally, and considering the selectivity for different products observed over the Rh-based catalysts (except for
Catalyst recovery
It is known that catalysts following mechanism A are highly sensitive toward the metals and ligands that are used to synthesize the catalysts.
85
Therefore, we focused on the recyclability of catalysts

Recyclability testing of catalysts

Recyclability testing of catalyst

Recyclability testing of catalyst
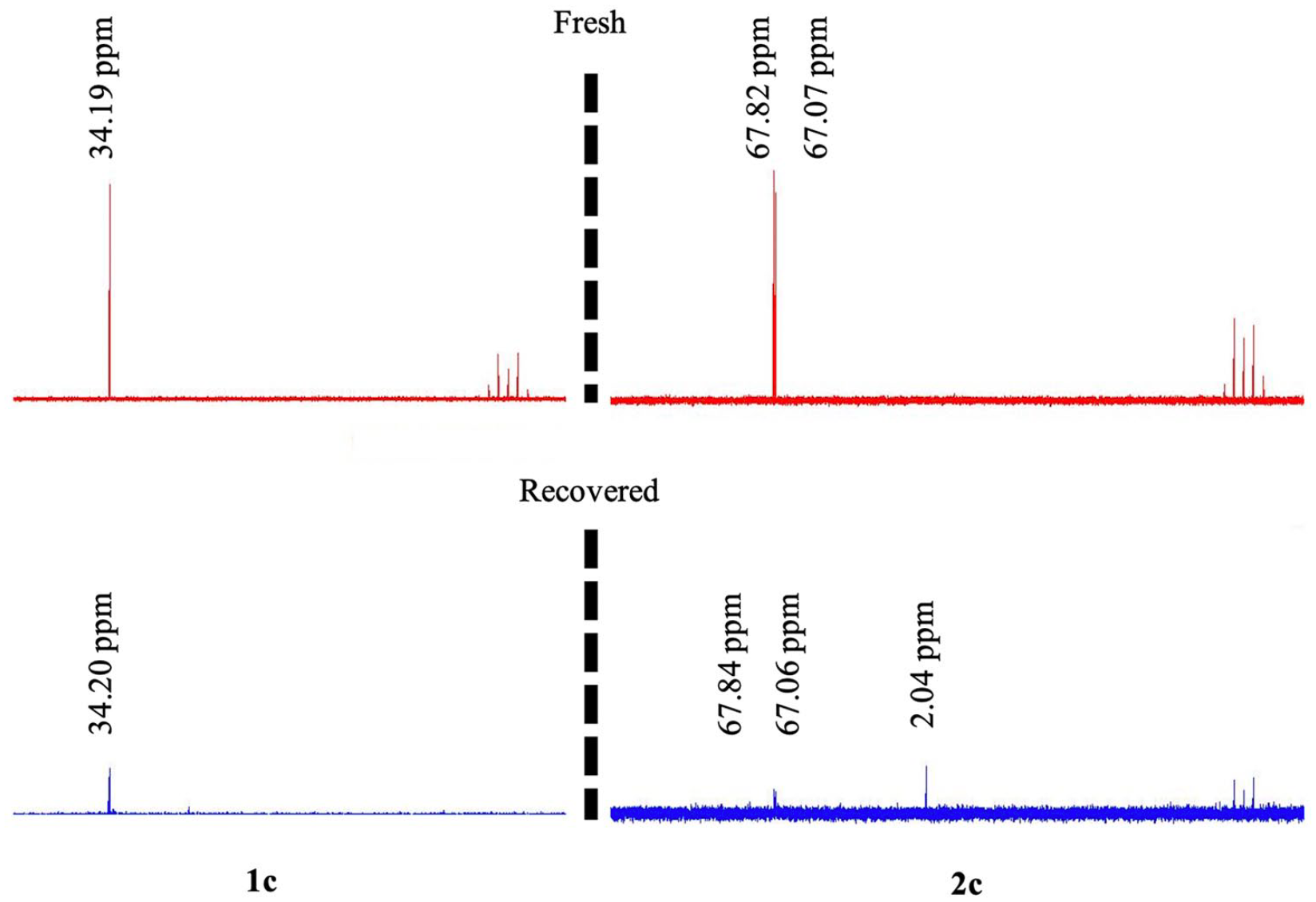
31P NMR spectra of the fresh and recovered catalysts
Ketone formation, due to deeper oxidation, is common when TBHP (t-BuOOH) is used as an oxidant.20,87 It can be postulated that the reaction mainly proceeds through the formation of hydroxyl or free radicals (mechanisms B and C), especially in the case of the Ir catalysts.84,88
Conclusion
Five different aminodiphosphine ligands were complexed with Ir and Rh as the metals, and were applied for the oxidation of n-octane using TBHP as the oxidant. The ligands were selected based on their chain length/steric hindrance, electron density, and their ability to yield different bite angles. Both catalysts gave relatively good n-octane conversion. In general, Ir-based complexes catalyze the deep oxidation pathway and they yielded ketones and acids as the major products, while following the inner-sphere mechanism, and involvement of M-alkyl radicals during the reaction. Interestingly, this mechanism seems to also promote the activation of internal carbons and ketone formation. Rh-based catalysts showed better selectivity for alcohols than the Ir complexes, suggesting a slightly greater contribution of the outer-sphere mechanism (A) (however, mechanism B is still dominant in this case as well) and forms more alcohols, while facilitating terminal carbon activation with high commercial values as starting materials for α-olefin formation. This study showed that mechanism A is sensitive to the structure of the complexes and the employed metals [high alcohol selectivity using sterically hindered ligands (hexyl and isopropyl)]. In contrast, the catalysts following mechanism B are not structure-sensitive, and can be recycled with the same performance, in spite of some structural changes to the catalyst during different cycles of the reaction.
Experimental
Material and methods
All experiments were performed using standard Schlenk techniques under inert conditions in moisture-free glassware with anhydrous solvents. All solvents were analytical grade. To render the reaction glassware moisture-free, it was heated with a heat gun followed by cycles of vacuum and nitrogen pressure. Diethyl ether and hexane were distilled from sodium benzophenone ketyl under nitrogen. Dichloromethane was distilled from P2O5, and ethanol from magnesium turnings. Deuterated solvents were used as received and stored in a desiccator. The NMR spectra were recorded at 162 MHz (31P) using a Bruker Ultrashield 400 MHz spectrometer. 31P NMR chemical shifts are reported in ppm from triphenylphosphine (17.6 ppm). All PNP ligands were synthesized using a procedure reported by our group. 74
Oxidation of n-octane
All products of the n-octane oxidation were analyzed using a PerkinElmer Auto System gas chromatograph connected to a flame ionization detector (FID) set at 260 °C. A PONA column (50 m × 0.20 mm × 0.5 μm) was utilized with the injector temperature set at 240 °C. Catalytic testing was carried out in a two-necked pear-shaped flask charged with 10 mg of the respective catalyst, pentanoic acid (as an internal standard), n-octane, the respective oxidant, and 10 mL of the solvent. The flask was equipped with a reflux condenser, and the contents were stirred, heated to the required temperature, and maintained at this temperature for 48 h in an oil bath. After that time period, an aliquot was removed using a Pasteur pipette and filtered through cotton wool and a silica gel plug, after which PPh3 was added (for reduction of the remaining TBHP and alkylperoxides which are formed as primary products in alkane oxidation). 43 An aliquot (0.5 μL) was injected into the gas chromatograph (GC) and quantified. A preliminary experiment showed that the standard, pentanoic acid, is not affected by the reaction medium.
Supplemental Material
SI-Supplemental_material_for_The_effects_of_metals_and_ligands_on_the_oxidation_of_n-octane_using_iridium_and_rhodium_PNP_aminodiphosphine_complexes – Supplemental material for The effects of metals and ligands on the oxidation of n-octane using iridium and rhodium “PNP” aminodiphosphine complexes
Supplemental material, SI-Supplemental_material_for_The_effects_of_metals_and_ligands_on_the_oxidation_of_n-octane_using_iridium_and_rhodium_PNP_aminodiphosphine_complexes for The effects of metals and ligands on the oxidation of n-octane using iridium and rhodium “PNP” aminodiphosphine complexes by Dunesha Naicker, Saba Alapour and Holger B Friedrich in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support from the National Research Foundation (NRF), Technology and Human Resources for Industry Programme (THRIP) (grant no. 1208035643) and the University of KwaZulu-Natal (University Research Fellowship (URF)).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
