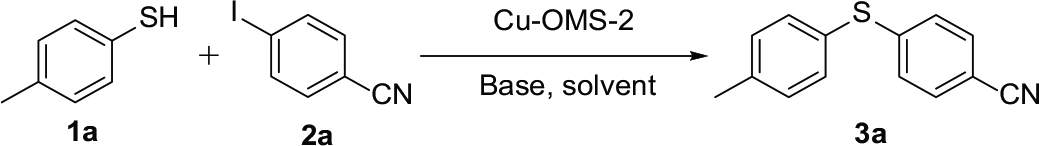
Abstract
We describe a practical protocol for efficiently preparing diaryl sulfide compounds using Cu–OMS-2 as the catalyst. Cu–OMS-2 originates from manganese oxide octahedral molecular sieves modified with copper ions and catalyzes the C–S coupling reaction of substituted thiophenols and aryl halides. This protocol has the advantages of environmental friendliness, simple operation, high yields, good tolerance of functional groups, and the Cu–OMS-2 catalytic material can be recycled several times.

Introduction
Sulfides, appearing frequently in natural products and medicines,1,2 are critical sulfur-containing organic compounds, especially in the field of new synthetic drugs. However, the traditional method of synthesizing sulfides by transition metal–catalyzed C–S coupling reaction of aryl halides with thiols has the shortcomings of high cost, harsh reaction conditions, and long response times.3–5 Therefore, it is necessary to develop new and efficient synthetic methods by taking advantage of new catalytic materials.
In 1978, a C–S bond coupling reaction catalyzed by palladium was reported by Kosugi et al. 6 , who used Pd(PPh3)4 as the catalyst to produce a series of thioether compounds by coupling iodobenzene or bromobenzene compounds with thiophenols or mercaptans. In 2009, Xu et al. 7 reported a method to construct C–S bonds using halogenated hydrocarbons and mercaptans with Cu2O. The main advantages of this method were that cheap and readily available Cu2O replaced the expensive and specialized copper salts, and that the reaction could be carried out in air without N2 protection. Compared with traditional methods, the transition metal–catalyzed coupling reaction of halogenated hydrocarbons and sulfur compounds has the advantages of simple operation, high efficiency, low cost, and mild reaction conditions, which have firmly attracted the attention of researchers. In 2012, Das and Chakraborty 8 revealed a method to synthesize thioethers using thiophenol and organic boric acid as substrates with AgOTf, 4Å molecular sieves, and KOH (Scheme 1(a)). In 2015, Anilkumar who has devoted significant efforts to study transition metal–catalyzed coupling reactions to prepare sulfides9–11 reported a method to produce sulfides using an Fe salt as the catalyst (Scheme 1(b)). We have also reported some methods for synthesizing aryl sulfides by heterogeneous catalysis.12–14

Manganese oxide octahedral molecular sieves (OMS-2), the structure of which is similar to zeolite molecular sieves, are a new type of material that can supply a high specific surface area. Furthermore, the specialized framework structure of OMS-2 with a large number of mixed valence Mn ions (Mn2+, Mn3+, Mn4+) allows it to perform well in redox reactions. Later, researchers found that OMS-2 also demonstrated an excellent performance in the field of organic synthetic catalysis when different metal ions were used to modify OMS. Reflux,15–18 ball milling, 19 impregnation, 20 and multi-step hydrothermal methods21–23 can produce OMS doped with Cu.
Finally, a one-step hydrothermal method was developed to synthesize the copper-doped manganese oxide octahedral molecular sieves (Cu–OMS-2). Figure 1 illustrates that the synthesized Cu–OMS-2 catalyst has a typical cryptomelane structure (KMn8O16).24,25 Obvious characteristic peaks observed at the angles of 12.6°, 17.9°, 28.7°, 37.5°, 41.9°, 49.9°, and 60.1° are basically consistent with the data for standard OMS-2 PDF card (JCPDS-29-1020). Furthermore, the image indicates that Cu did not affect the crystal bulk structure of OMS-2 in the prepared Cu–OMS-2 catalyst. In addition, no obvious characteristic diffraction peaks corresponding to copper crystal phase oxides were detected in the X-ray diffraction (XRD) pattern, implying that the doped copper was highly dispersed in Cu–OMS-2. It is further speculated that some copper ions are located into the framework or pores. The morphology of the catalyst was observed by scanning electron microscope (SEM). Figure 2 illustrates the rod-like morphology of Cu–OMS-2.

X-ray powder diffractogram of Cu–OMS-2.

SEM image of Cu–OMS-2.
Based on our previous research,26–32 herein, we report a method to efficiently synthesize diaryl sulfide compounds by utilizing copper-doped manganese oxide octahedral molecular sieves (Cu–OMS-2) to catalyze the heterogeneous C–S coupling reaction of thiophenols and aryl halides (Scheme 1(c)).
Results and discussion
We began our initial trials with 4-methylthiophenol (
Reaction optimization. a

Reaction conditions: 4-methylthiophenol (1 mmol), 4-cyanoiodobenzene (1 mmol), base (2 mmol), Cu–OMS-2 (10 mg), solvent (2.5 mL), 95 °C, 6 h.
Isolated yield.
Cu–OMS-2 (5 mg).
Cu–OMS-2 (16 mg).
No Cu–OMS-2, 95 °C.
The optimal reaction condition.
With the optimal reaction conditions having been established, we next focused on investigating the scope of substrates. So as to synthesize diaryl sulfide products, different aryl iodides and substituted thiophenols were reacted under the optimal conditions (Table 2). We succeeded in preparing various C–S coupling products with high yields ranging 74%–90%. Moreover, fluorine- or chlorine-substituted thiophenols can react with aryl iodides to afford the target products (
Scope of thiophenols and aryl iodobenzenes. a

Reaction conditions:
Furthermore, recycling studies using Cu–OMS-2 were investigated by operating the reaction under the optimized conditions (Scheme 2). We can clearly see that recycled Cu–OMS-2 worked well and gave an excellent yield of 86% of the diaryl sulfide compound after six recycles. In addition, the results indicate that Cu–OMS-2 is stable over several cycles.

Catalyst recycling studies.
Conclusion
In conclusion, we have described a practical protocol for efficiently preparing diaryl sulfides using Cu–OMS-2 to catalyze the C–S coupling reactions of substituted thiophenols and aryl halides. This protocol has the advantages of environmental friendliness, simple operation, high yields, good tolerance of functional groups, and the Cu–OMS-2 catalyst material can be recycled several times.
General experiments
All the obtained products were characterized by 1H NMR and 13C NMR spectroscopy (400 or 100 MHz). Chemical shifts are reported in parts per million (ppm, δ) downfield from tetramethylsilane. Proton coupling patterns are described as singlet (s), doublet (d), triplet (t), and multiplet (m); TLC was performed using commercially prepared 100–400 mesh silica gel plates (GF 254), and visualization was effected at 254 nm; all the reagents were purchased from commercial sources and were used without further purification.
Preparation of Cu–OMS-2
Manganese sulfate (8.8 g) and Cu(NO3)2·3H2O (0.6375 g) in purified water (100 mL) were added to a solution of purified water (30 mL) containing concentrated nitric acid (3 mL) and potassium permanganate (5.89 g). The mixed solution was placed in an oven at 100 °C for the hydrothermal reaction for 24 h. The product was then filtered, washed, and dried at 120 °C for 8 h. Finally, the dry Cu–OMS-2 was calcined in a muffle furnace at 350 °C for 2 h to afford a black powder.
Typical procedure for the synthesis of 3
Benzenethiol
4-(p-Tolylthio)benzonitrile (3a )
Yield: 89%, white solid, m.p. 95-96 °C (lit. 33 ); 1H NMR (400 MHz, CDCl3): δ: 7.40-7.32 (m, 4H, Ar-H), 7.18 (d, J = 8.4 Hz, 2H, Ar-H), 7.04 (d, J = 8.4 Hz, 2H, Ar-H), 2.3 (s, 3H, CH3); 13C NMR (101 MHz, CDCl3): δ: 146.65, 140.01, 135.01, 132.32, 130.80, 126.78, 126.69, 118.97, 108.28, 21.37.
4-[(2-Chlorophenyl)thio]benzonitrile (3b )
Yield: 79%, colorless oily liquid; 1H NMR (400 MHz, CDCl3): δ: 7.49-7.42 (m, 4H, Ar-H), 7.34-7.28 (m, 1H, Ar-H), 7.26-7.20 (m,1H, Ar-H), 7.12 (d, J = 8.8 Hz, 2H, Ar-H); 13C NMR (101 MHz, CDCl3): δ: 143.19, 138.14, 135.87, 132.61, 130.75, 130.73, 130.56, 128.17, 127.94, 118.71, 109.49.
(2-Chlorophenyl)(4-nitrophenyl) sulfane (3c )
Yield: 81%, yellow solid, m.p. 111-113 °C; 1H NMR (400 MHz, CDCl3): δ: 8.10 (d, J = 7.2 Hz, 2H, Ar-H), 7.57 (t, J = 6.4 Hz, 2H, Ar-H), 7.41 (t, J = 6.0 Hz, 1H, Ar-H), 7.33 (t, J = 6.0 Hz, 1H, Ar-H), 7.20 (d, J = 6.8 Hz, 2H, Ar-H); 13C NMR (101 MHz, CDCl3): δ: 145.92, 145.85, 138.54, 136.32, 131.06, 130.84, 130.11, 128.01, 127.45, 124.21.
4-[(3-Chlorophenyl)thio]benzonitrile (3d )
Yield: 83%, colorless oily liquid; 1H NMR (400 MHz, CDCl3): δ: 7.45 (d, J = 8.8 Hz, 2H, Ar-H), 7.40 (s, 1H, Ar-H), 7.34-7.25 (m, 3H, Ar-H), 7.16 (d, J = 8.8 Hz, 2H, Ar-H); 13C NMR (101 MHz, CDCl3): δ: 144.11, 135.49, 133.45, 132.64, 131.88, 130.91, 130.64, 129.38, 128.29, 118.63, 109.58.
4-[(4-Chlorophenyl)thio]benzonitrile (3e )
Yield: 85%, white solid, m.p. 88-89 °C (lit. 34 ); 1H NMR (400 MHz, CDCl3): δ: 7.51 (d, J = 8.8 Hz, 2H, Ar-H), 7.46-7.39 (m, 4H, Ar-H), 7.18 (d, J = 8.8 Hz, 2H, Ar-H); 13C NMR (101 MHz, CDCl3): δ: 144.88, 135.71, 135.53, 132.47, 130.12, 129.49, 127.51, 118.56, 109.12.
(4-Chlorophenyl)(4-nitrophenyl)sulfane (3f )
Yield: 85%, yellow solid, m.p. 89-90 °C (lit. 35 ); 1H NMR (400 MHz, CDCl3): δ: 8.10-8.06 (m, 2H, Ar-H), 7.50-7.41 (m, 4H, Ar-H), 7.22-7.16 (m, 2H, Ar-H); 13C NMR (101 MHz, CDCl3): δ: 147.66, 145.64,136.10, 135.90, 130.32, 129.16, 126.97, 124.20.
1-[4-((4-Chlorophenyl)thio)phenyl]ethan-1-one (3g )
Yield: 74%, white solid, m.p. 83-84 °C (lit. 34 ); 1H NMR (400 MHz, CDCl3): δ: 7.48 (d, J = 8 Hz, 2H, Ar-H), 7.42-7.35 (m, 4H, Ar-H), 7.22 (d, J = 8 Hz, 2H, Ar-H), 2.56 (s, 3H, CH3); 13C NMR (101 MHz, CDCl3): δ: 197.11, 144.06, 135.03, 134.90, 134.84, 130.95, 129.92, 129.04, 127.83, 26.53.
4-[(4-Fluorophenyl)thio]benzonitrile (3h )
Yield: 80%, white solid, m.p. 85-86 °C (lit. 33 ); 1H NMR (400 MHz, CDCl3): δ: 7.54-7.50 (m, 2H, Ar-H), 7.48 (d, J = 6.8 Hz, 2H, Ar-H), 7.17-7.10 (m, 4H, Ar-H); 13C NMR (101 MHz, CDCl3): δ: 164.58, 162.58, 145.83, 137.07, 132.43, 126.88, 125.84, 118.71, 117.34, 108.81.
(4-Fluorophenyl)( 4-nitrophenyl)sulfane (3i )
Yield: 83%, yellow solid, m.p. 82-84 °C (lit. 36 ); 1H NMR (400 MHz, CDCl3): δ: 7.99 (d, J = 7.2 Hz, 2H, Ar-H), 7.51-7.44 (m, 2H, Ar-H), 7.12-7.05 (m, 4H, Ar-H); 13C NMR (101 MHz, CDCl3): δ: 164.72, 162.72, 148.48, 145.45, 137.26, 126.33, 125.56, 124.11, 117.46.
(4-Nitrophenyl)(p-tolyl)sulfane (3j )
Yield: 88%, yellow solid, m.p. 78-80 °C (lit. 36 ); 1H NMR (400 MHz, CDCl3): δ: 8.10 (d, J = 7.2 Hz, 2H, Ar-H), 7.49 (d, J = 6.4 Hz, 2H, Ar-H), 7.32 (d, J = 7.2 Hz, 2H, Ar-H), 7.19 (d, J = 7.2 Hz, 2H, Ar-H), 2.47 (s, 3H, CH3); 13C NMR (101 MHz, CDCl3): δ: 149.35, 145.17, 140.25, 135.10, 130.87, 126.52, 126.16, 123.99, 21.35.
4-[(4-Methoxyphenyl)thio]benzonitrile (3k )
Yield: 90%, white solid, m.p. 92-93 °C (lit. 33 ); 1H NMR (400 MHz, CDCl3): δ: 7.48-7.43 (m, 4H, Ar-H), 7.07 (d, J = 8 Hz, 2H, Ar-H), 6.97 (d, J = 8 Hz, 2H, Ar-H), 3.86 (s, 3H, OCH3); 13C NMR (101 MHz, CDCl3): δ: 160.99, 147.37, 137.10, 132.26, 126.08, 120.42, 118.96, 115.59, 108.05, 55.47.
(2-Methoxyphenyl)(phenyl)sulfane (3l )
Yield: 36%, colorless liquid; 1H NMR (400 MHz, CDCl3): δ: 7.36-7.27 (m, 5H, Ar-H), 7.23-7.21 (m, 1H, Ar-H), 7.09-7.07 (m, 1H, Ar-H), 6.91-6.85 (m, 2H, Ar-H), 3.87 (s, 3H, OCH3); 13C NMR (101 MHz, CDCl3): δ: 157.34, 140.50, 134.51, 131.64, 131.53, 129.19, 128.33, 127.12, 124.12, 121.29, 110.89, 55.93, 29.75.
(4-Methoxyphenyl)(phenyl)sulfane (3m )
Yield: 43%, colorless liquid; 1H NMR (400 MHz, CDCl3): δ: 7.43 (d, J = 8.4 Hz, 1H, Ar-H), 7.24-7.20 (m, 2H, Ar-H), 7.14-7.10 (m, 1H, Ar-H), 6.56-6.53 (m, 2H, Ar-H), 6.63 (d, J = 2.4 Hz, 1H, Ar-H), 6.56-6.53 (m, 2H, Ar-H), 3.82 (s, 3H, OCH3); 13C NMR (101 MHz, CDCl3): δ: 162.15, 157.59, 136.81, 135.72, 133.41, 128.07, 125.12, 124.77, 113.52, 107.26, 105.81, 99.46, 54.42.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge financial support of this work by the National Natural Science Foundation of China (21602123) and the 111 Project (no. D20015).