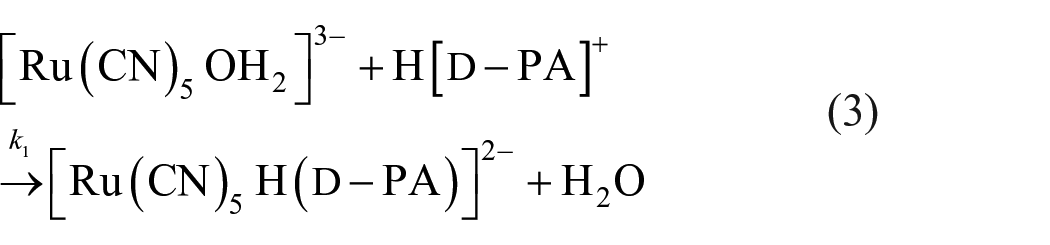Abstract
The kinetics of the formation of the light orange–coloured complex [Ru(CN)5D-PA]3− are studied spectrophotometrically through substitution of a coordinated H2O molecule in aquapentacyanoruthenate(II) [Ru(CN)5OH2]3− by interaction with D-penicillamine [D-PA] in aqueous medium at 490 nm (λmax of [Ru(CN)5D-PA]3−). The reaction is monitored under pseudo-first-order conditions, taking [D-PA] in excess over [Ru(CN)5OH23−]. The effects of various reaction parameters on the rate of the reaction are investigated. Experimental observations reveal that the variation in [D-PA] obeyed the first-order rate law while it is found to be invariant with [Ru(CN)5OH23−] in the whole experimental range. With ionic strength variation, as the reaction advances a decrease in the reaction rate is noticed. The product stoichiometry is assigned as 1:1. The ease of substitution at an H2O molecule in [Ru(CN)5OH23−] is considered on the basis of the electronic effect generated through interactions of the M–OH2 bond. A provisional mechanism based on the calculated results is proposed based on the slowest step of the reaction.
Overlay scan for the product, [Ru(CN)5D-PA]3− formation at different time intervals between 200 and 900 nm:

Introduction
Transition metals have received significant attention because of their applicability in the area of inorganic medicinal chemistry.1–4 One of the important properties associated with transition metal complexes is their profound action as chemotherapeutic agents. 5 For example, complexes of ruthenium(II),6,7 rhodium(III) 8 and iridium(III)9–11 (possessing d 6 configuration) exhibit antitumor properties by interacting selectively with biomolecules in nano- to micro-molar doses.
D-Penicillamine, that is, C5H11NO2S or D-PA, is a decomposition product of β-lactam antibiotics, produced naturally by β-lactam ring degradation in penicillin (Figure 1).
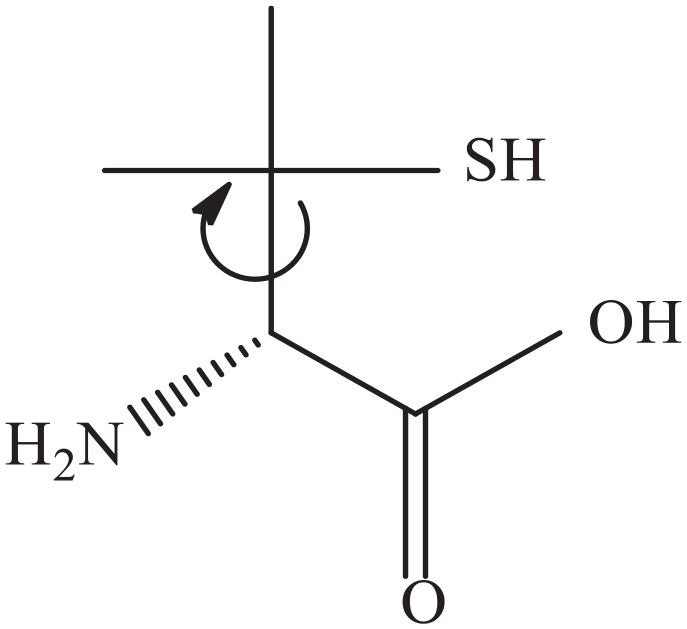
Structure of D-penicillamine.
D-PA is an amino acid, and its structure is quite similar to that of cysteine and valine and is therefore regarded as β,β-dimethylcysteine and β-mercaptovaline, respectively. The presence of –SH, –NH2 and –COOH groups in penicillamine imparts different modes of binding with various organic12–14 and inorganic15,16 substrates, or both,17,18 during complexation. Out of the D- and L-enantiomers of penicillamine, the former is employed clinically because of the reported high toxic behaviour of the latter. 19 D-PA has strong chelation ability, especially with metal ions that possess a strong affinity for chelation with a sulfur atom. 20 The exceptional pharmaceutical importance of D-PA lies in its excellent potential to bind with metal ions. 21 On account of its excellent properties, D-PA has been used for the treatment of Wilson’s disease by decreasing excess copper levels through reduction of Cu(II) to Cu(I), in heavy metal poisoning, in cystinuria, in rheumatoid arthritis, and also has several significant applications in biological systems.22–29
In the past, ruthenium compounds have been widely employed as potential cytostatic agents as well as a possible alternative to drugs containing platinum metal.30–32 Amino and cyano complexes of ruthenium metal, that is, [(NH3)5RuL]2+ and [(CN)5RuL]3+ with pyrazine or pyridine aromatic heterocycles, 33 have been explored previously along with other pentacyano(ligand) complexes of Ru(II)34–37 and Fe(II).38,39 The reason behind the use of low-spin [Ru(CN)5OH2]3− in the case of amino acids 40 lies in its high coordination potential to bind effectively with additional ligands, which further easily substitutes a coordinated molecule of water and ultimately forms a complex of high stability. Such types of substituted pentacyanoruthenate(II) complexes show an excellent potential to explore their kinetic properties.41,42 In this regard, research concerning the kinetics of [Ru(CN)5OH2]3− with heterocyclic ligands43,44 in aqueous medium was performed earlier by Naik and co-workers. The kinetic properties of [Fe(CN)5OH2]3− with penicillamine in aqueous 45 as well as in acidic 46 media have also been investigated, but in contrast to Fe(II) complexes, very minimal information is available on the substitutional kinetic behaviour of Ru(II) complexes with ligands containing sulfur. Therefore, we considered it important to strengthen our understanding by detailed kinetic studies of ligand substitution of sulfur containing D-PA with [Ru(CN)5OH2]3− in aqueous medium, forming the complex [Ru(CN)5D-PA]3−. A stoichiometry of 1:1 at 490 nm was determined on the basis of various reaction parameters (namely, pH, ionic strength, [Ru(CN)5OH23−], D-PA and temperature).
Results and discussion
The impact of different reaction parameters (namely, pH, [Ru(CN)5OH23−], D-PA, ionic strength and temperature) was analysed on the rate of the reaction by modifying the concentration of each variable at a time and maintaining the others as constant to their optimum values.
Dependency of the reaction rate on pH
To select a suitable value of pH for the optimum reaction rate, the complexation efficiency was first tested as a function of pH in the range of 2.0–9.0, with the other experimental parameters fixed to their optimum values using the fixed time procedure method. Phthalate/sodium hydroxide or HCl buffer solution was used to maintain the pH of the reaction mixture. At each pH, the corresponding kobs and kf values were determined. The kf versus pH plot (Figure 2) indicates clearly that the reaction rate was markedly slow at low pH, and increased on increasing the value of pH before being retarded at higher pH values. The pH value (3.8 ± 0.02) was recommended as an optimum value for kinetic analysis, since at this pH, appreciable changes in absorbance were noticed. At low pH, a noticeable decrease in kf values represented its close proximity with that of observed with further Fe(II) complex 47 earlier, suggesting the coordinated cyanide ligands protonation is liable for the decreased lability of H2O molecule in the protonated species [(HNC)Ru(CN)4OH2]2−, 41 as mentioned in equations (1)–(4)

Effect of pH on the reaction of [Ru(CN)5OH2]3− with D-PA as the ligand in aqueous medium under the optimum reaction conditions: D-PA = 4.5 × 10−3 M, [Ru(CN)5OH23−] = 4.5 × 10−4 M, μ = 0.25 M and temperature = 30.0 ± 0.01 °C.
Dependency of the reaction rate on d -PA
At the optimum pH value (3.8 ± 0.02), the influence of D-PA on the rate of reaction was analysed with the variation of D-PA in the range of 1.5 × 10−3 to 4.5 × 10−3 M, following pseudo-first-order conditions by taking D-PA in excess over [Ru(CN)5OH2]3− and maintaining the other experimental variables constant to their optimum values. The obtained plot of kobs versus D-PA is linear in nature (Figure 3) and passes through the origin (R2 ⩾ 0.9952, standard deviation (SD) ⩽ 0.0698).

Effect of D-PA on the reaction of [Ru(CN)5OH2]3− with D-PA in aqueous medium under the optimum reaction conditions: pH = 3.8 ± 0.02, [Ru(CN)5OH23−] = 4.5 × 10−4 M, μ = 0.25 M and temperature = 30.0 ± 0.01 °C.
On the basis of studies performed previously, 44 the reaction most probably occurs through equations (5) and (6)
Equation (6) can be represented in terms of the rate expression in equation (7)
where
Dependency of the reaction rate on the reactant [Ru(CN)5OH23−]
The values of kobs were evaluated as a function of [Ru(CN)5OH23−] by varying its concentration in the range of 1 × 10−4 to 4.5 × 10−4 M while maintaining the values of the other reaction variables fixed. Low concentration values of [Ru(CN)5OH23−] were strictly avoided to minimize the possibility of dimerization. 47 The obtained values of kobs and kf with [Ru(CN)5OH23−] are compiled in Table 1 and depict no significant variation with increasing concentration of [Ru(CN)5OH23−] over the whole concentration range. Thus, the reaction rate was found to be invariant with respect to the Ru(II) concentration.
Effect of [Ru(CN)5OH23−] under the optimum conditions: pH = 3.8 ± 0.02, D-PA = 4.5 × 10−3 M, µ = 0.25 M (NaClO4) and temperature = 30.0 ± 0.01 °C.

kobs (av) = 0.41 ± 0.042; kf (av) = 0.088 ± 0.02.
Dependency of the reaction rate on the ionic strength (µ)
The electrolyte effect on the ligand substitution kinetics was studied by changing the neutral electrolyte concentration, that is, NaClO4 in the range of 0.025–0.25 M, while maintaining the other parameters constant to their optimum values. The graph plotted between ln kf versus √I/1 + √I was observed to be a straight line (Figure 4) (R2 ⩾ 0.9915, SD ⩽ 0.8721) and confirms with the relationship proposed by Bronsted–Bjerrum–Christiansen. 48 The graph is indicative of a negative salt effect, that is, as the NaClO4 concentration increases, the reaction rate decreases, probably due to interactions of the cation (i.e. Na+ from NaClO4) with the CN− ion, which leads to a decrease in the electron density on the ruthenium metal centre, resulting in the requirement of more energy to release H2O from [Ru(CN)5OH2]3−.

Effect of ionic strength in the reaction of [Ru(CN)5OH2]3− with D-PA in aqueous medium under the optimum reaction conditions: pH = 3.8 ± 0.02, D-PA = 4.5 × 10−3 M, [Ru(CN)5OH23−] = 4.5 × 10−4 M and temperature = 30.0 ± 0.01 °C.
Dependency of the reaction rate on the temperature
The impact of the temperature on the rate of the reaction was studied in the range of 308–323 K within the accuracy of ±0.02 °C, under optimized conditions. Kinetic studies at high temperature are circumvented to minimize the possibility of decomposition of the product. The value of kf was evaluated and is related to different thermodynamic parameters (namely, ∆S# (entropy of activation) and ∆H# (enthalpy of activation)) through Eyringʼs equation as shown in equation (8)

The linear least square plot between ln(kf/T) versus 1/T is obtained as a straight line (Figure 5) with R2 ⩾ 0.9962 and SD ⩽ 0.2753.

Effect of temperature for the reaction of [Ru(CN)5OH2]3− with D-PA in aqueous medium under the optimum reaction conditions: D-PA = 4.5 × 10−3 M, [Ru(CN)5OH23−] = 4.5 × 10−4 M, pH = 3.8 ± 0.02 and µ = 0.25 M.
The values of ∆H# and ∆S# were computed, respectively, from the slope and intercept of the plotted graph. The obtained values of Ea, ∆H#, ∆S# and ∆G# are 56.8 kJ mol−1, 48.2 kJ mol−1, 193.4 JK−1 mol−1 and −10.4 kJ mol−1, respectively. The ∆S# exhibits a high positive value, therefore taking into account the above-mentioned results, the reaction shows a high bond dissociative character. Analysis of the above-mentioned activation parameters and kinetic data suggests that the reaction under consideration preferentially follows a dissociation (D) mechanism. The most reasonable scheme for the whole mechanism is given through equations (9) and (10), corresponding to the substitution processes in pentacyanoferrate(II)49,50
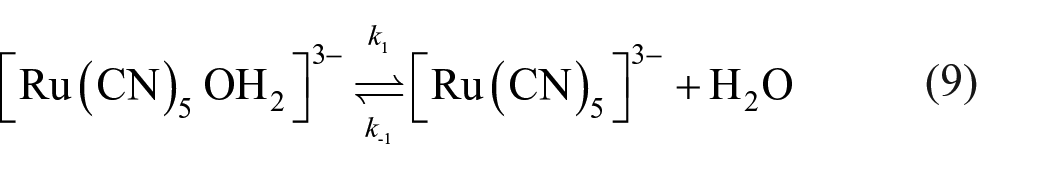

Applying steady-state approximation for a trigonal-bipyramidal intermediate, that is, [Ru(CN)5]3−, equation (11) can be derived

Upon simplification of equation (11), equation (12) can be obtained

Using equation (11), the reaction rate is expressed as follows in equation (13)

After solving equations (12) and (13), equation (14) is deduced

As the rate of the reaction is directly proportional to [Ru(CN)5OH23−] and D-PA, therefore it can be depicted as follows in equation (15) or in equation (16)


Substituting equation (16) in to equation (14), kobs can be expressed through equation (17)

As per the reaction conditions, we know

Therefore, equation (17) can be written as

By applying

Equation (18) converts into equation (19)

Equation (19) shows that kobs follows first-order kinetics and the plotted graph between kobs versus D-PA (Figure 3) represents a straight line with a zero intercept, which is in good correlation.
Both metal complexes of Ru(II) and Fe(II) possess a low-spin d 6 transition state; therefore, a similar rate during their water exchange process is to be assumed. But the salient feature for the prevalence of the difference in rate constant during replacement of the coordinated H2O molecule in aqua complexes of Ru(II) and Fe(II) is the strength of the bond between the metal and the coordinated water molecule, which is slightly stronger in the case of Ru(II) metal complexes due to a much more favourable 4d orbital enhancement of the lowest unoccupied molecular orbital (LUMO) towards the lone pairs of the H2O molecule as compared to Fe(II) complexes. Removal of a coordinated water molecule from Ru(II) metal complexes is mainly attributed to effective charge reduction on the metal centre, which subsequently decreases the activation energy due to an interaction between the oxygen atom lone pairs and the ruthenium metal, and being responsible for M–OH2 dissociation. The large 4d orbital enhancement of the LUMO in Ru(II) accounts for the substantial π-back-bonding character of the CN ligands, which ultimately emphasizes the M(III) character of Ru(II) as compared to Fe(II), resulting in faster exchange of coordinated water in aqua complexes of Fe(II) than in Ru(II). The value of the rate constant in Ru(II) also decreases considerably in acidic environment for exchange of a coordinated water molecule due to coordinated cyanide ligand protonation. This cyanide ligand protonation changes the electronic environment, making the bond between ruthenium metal and coordinated water stronger. Even so, in the present kinetic study in aqueous medium, the high positive value of entropy is the main factor behind the occurrence of the dissociative (D) mechanism.
Finally, an overlay scan of the product formation was recorded at different time intervals between 200 and 900 nm for substitution of the water molecule in the complex [Ru(CN)5OH2]3− (λmax 310 nm, ε = 1.64 × 103 M−1cm−1) in aqueous medium by D-PA (Figure 6). The maximum absorbance was noticed at λmax 490 nm (ε = 1.97 × 103 M−1cm−1) due to the formation of an orange-coloured product [Ru(CN)5D-PA]3− as a function of time and attributed to a metal to ligand charge transfer transition where only the product shows a maximum absorbance without any intervention caused by the reactants. The metal-to-ligand transitions due to
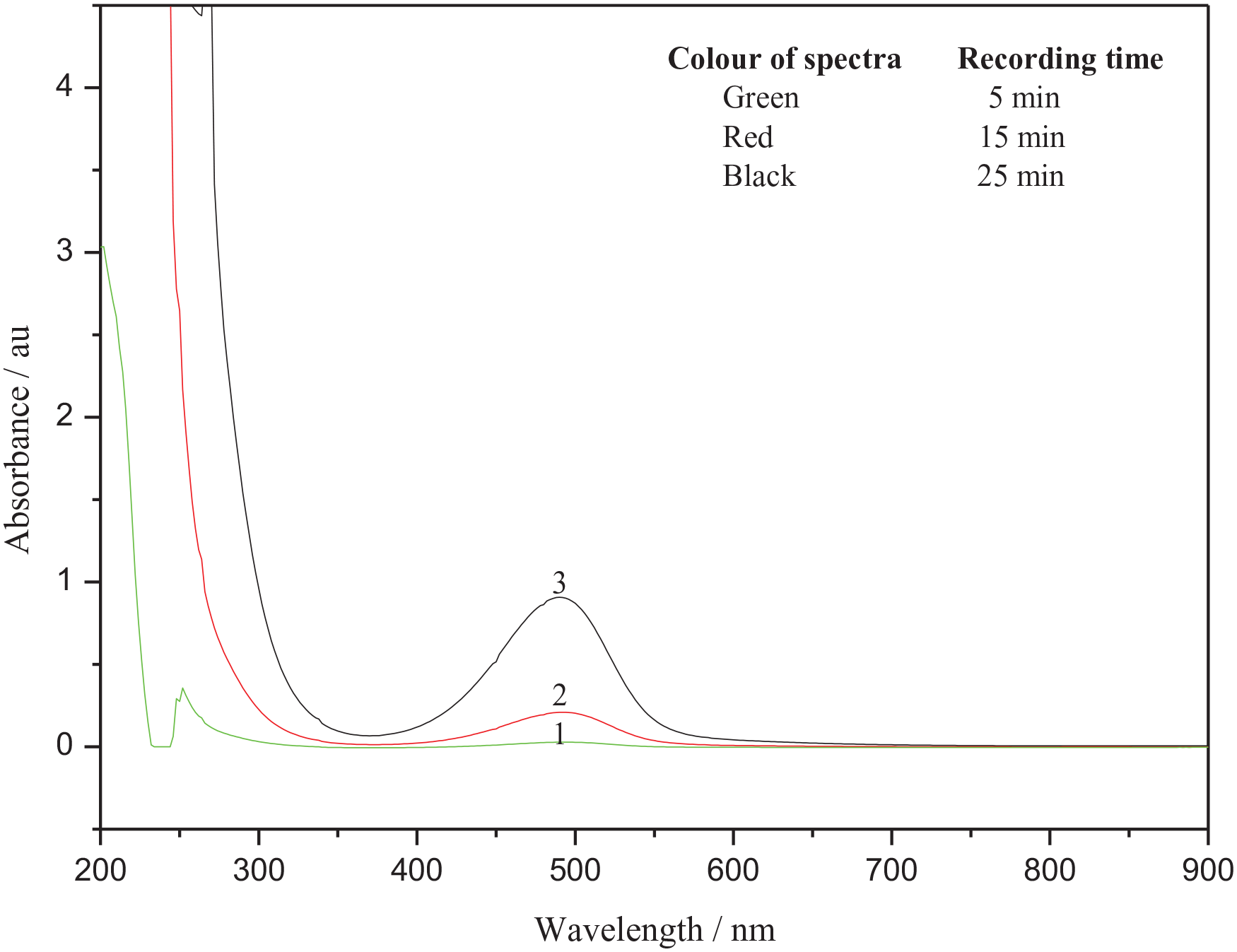
Absorption spectra of the product [Ru(CN)5D-PA]3− under the conditions: [Ru(CN)5OH23−] = 4.5 × 10−4 M, D-PA = 4.5 × 10−3 M, µ = 0.25 M, temperature = 30.0±0.01 °C and pH = 3.8 ± 0.02.
Conclusion
An orange-coloured substituted complex [Ru(CN)5D-PA]3− was formed as a final reaction product through substitution of a coordinated water molecule between [Ru(CN)5OH2]3− and D-PA through metal to ligand charge transfer transition
Experimental
Chemicals and reagents
All the chemicals used were pure and were employed directly in each kinetic run. Potassium hexacyanoruthenate(II) (K4[Ru(CN)6].3H2O) was obtained from Alfa Aesar and D-PA from Fluka (Switzerland). NaClO4 (sodium perchlorate) and KBr (potassium bromide) were obtained, respectively, from Aldrich and S D Fine-Chem Limited. Stock solutions of each reagent were prepared by dissolving the required amount in double distilled de-ionized water and stored in the dark in amber-coloured bottles to rule out the possibility of decomposition and oxidation of the product. A stock solution of 10−3 M of D-PA was prepared and diluted as required prior to use. With the help of an iodometric method, 52 a 10−2-M stock solution of bromine was prepared and standardized daily. A 0.25 M solution of NaClO4 was used to maintain the ionic strength of the reaction mixture. Standardization of the pH meter was accomplished with BDH buffers at the commencement of each run. A pH of the desired value was maintained using phthalate–sodium hydroxide buffer solution. 53
Kinetic measurements
All kinetic measurements were carried out in aqueous medium, using a DIGI-110 single beam visible spectrophotometer (SISCO, India) accompanied with a self-made thermostating system having circulatory arrangement of water. 54 The spectral scan for the product formation was recorded using a double beam UV-Vis spectrophotometer (Systronic-2203). For pH measurements, a Systronics µ pH System (model-361) was employed. Kinetic measurements were performed ensuring pseudo-first-order conditions by considering D-PA in excess (at least 10 times) over the ruthenium ion solution, and all manipulations were executed at the desired temperature. A rapid aquation of [Ru(CN)6]4− produced [Ru(CN)5OH2]3− solution by reacting the same molar concentrations of hexacyanoruthenate(II) and bromine with KBr in 10-fold excess. 55 [Ru(CN)6]4− reacts with Br2 in an expeditious manner and produces [Ru(CN)5OH2]3− solution (pale yellow in colour, λmax 310 nm).
To start the reaction, the pre-equilibrated reagents were added in the given order: [Ru(CN)5OH2]3−, NaClO4 and D-PA (2 cm3). After appropriate mixing, the mixture was transferred into a 10-mm quartz cuvette cell inside the spectrophotometer to monitor the progress of the reaction at different intervals of time at 490 nm. The value of the pseudo-first-order rate constant (kobs) was assessed from equation (20) using the least-squares fitting method

where A0 is the absorbance at time zero, At is the absorbance at different intervals of time, and A∞ is the final absorbance, recorded when the reaction was finalized.
Footnotes
Acknowledgements
The authors are thankful to the Head of Department of Chemistry, Lucknow University, Lucknow, for providing us with departmental facilities to accomplish this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.