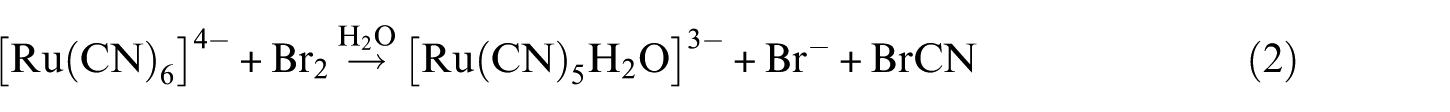Abstract
The formation kinetics of the complex, [Ru(CN)5INH]3−, formed through the ligand substitution reaction between isoniazid (INH) and aquapentacyanoruthenate(II) ([Ru(CN)5H2O]3−), have been investigated, under pseudo first-order conditions, as a function of concentrations of [INH] and [Ru(CN)5H2O]3−, ionic strength and temperature at pH = 4.0 ± 0.02 in 0.2 M NaClO4 spectrophotometrically at 502 nm (λmax of intense yellow colour product [Ru(CN)5INH]3−) corresponding to metal-to-ligand charge-transfer transitions, in aqueous medium. The pseudo first-order condition was maintained by taking at least 10% excess of [INH] over [Ru(CN)5H2O]3−. The stoichiometry of the reaction product was found to be 1:1 which was further supported and characterized using elemental analysis, infrared, nuclear magnetic resonance and mass spectrometric techniques. Thermodynamic and kinetic parameters have also been computed, using the Eyring equation, and the values of ΔH≠, Ea, ΔG≠ and ΔS≠ were found to be 47.3 kJ mol−1, 49.8 kJ mol−1, −8.62 kJ mol−1 and 187.6 J K−1mol−1, respectively. The reaction was found to obey first-order kinetics with respect to [INH]. It exhibited a negative salt effect on the rate upon variation of ionic strength of the medium. A tentative mechanistic scheme was proposed on the basis of experimental findings.
Keywords
Introduction
There has been a considerable advancement in the past few years concerning the kinetics and mechanism of ligand substitution reactions of catalysed and uncatalysed transition metal complexes in aqueous medium.1–15 Previous studies revealed that the transition metals, exhibiting complexes like [M(CN)5L]n− (where M(II/III) = Fe, Ru and L = H2O, CN−, imides) show wide applications towards physiological as well as industrial approaches.16–23 Numerous transition metal ions, alone or as binary mixtures, such as osmium(III) oxide, palladium(II) chloride, ruthenium(III) chloride, platinum(IV) chloride and iridium(III) chloride, have been used extensively in redox reactions as a homogeneous catalysts, and some systems among these have been proved quite suitable for the purpose of kinetic analysis.24–26 The present developing field of transition metal–mediated organic methodologies has incorporated several new and desirable properties, in combination with ruthenium metal, to attain the present requirement of organic synthesis. 25 Various oxidation processes of organic and inorganic substrates, involving ruthenium metal as a catalyst,26,27 have been reported. Several ruthenium compounds possess the ability to damage genetic materials,28–31 so they act as bacterial mutagens. Research concerning the activity of ruthenium complexes shows that many have antitumor properties; moreover, they are found to be less toxic in comparison with cisplatin.32,33 The bioactivity of ruthenium(II/III) complexes still requires much attention. 34
At present, isoniazid (isonicotinoylhydrazide; INH) is a first-choice medical drug, known for the prevention and treatment of tuberculosis (TB). 35 INH, a pro-drug, activated by Mycobacterium tuberculosis catalase-peroxidase KatG, generates an isonicotinoyl acyl radical, which in subsequent steps checks the synthesis of mycolic acid which is a necessary component in cell wall synthesis of M. tuberculosis. 36 Treatment of TB currently involves several issues such as its latency, co-infection with HIV and resistance developed against drugs due to the emergence of multidrug-resistant tuberculosis (MDR-TB) and extensively drug-resistant tuberculosis (XDR-TB). 37 MDR-TB emerges due to resistance against most potent first-line antitubercular drugs such as INH and rifampicin (RIF), while XDR-TB is resistant not only to INH and RIF but also to fluoroquinolones and at least one injectable second-line drug.
A wide variety of complexes containing nitrogen, oxygen, sulphur and phosphorous donor atoms have been synthesized from the labile [Fe(CN)5H2O]3− ion, forming the pentacyanoferrate(II) complex, [Fe(CN)5L]3−.38,39 The ligand substitution reactions of low-spin pentacyano(ligand)ferrate(II) complexes with various ligands, containing heterocyclic atoms, have attracted much attention.40,41 The ligand substitution behaviour of the low-spin [Ru(CN)5H2O]3− ion with some nitrogen-containing aromatic heterocyclic ligands, with reference to its kinetics and mechanism, was first studied by Hoddenbagh and Macartney, 42 Baran and Ulger 39 and recently by Naik et al. 9 The low-spin aquapentacyanoruthenate(II) ion, [Ru(CN5)H2O]3−, has the capability of binding easily with one incoming or substituting ligand, it easily loses easily a coordinated water molecule and subsequently forms a highly stable nitrogen heterocyclic substituted complex.
In the recent past, we have been interested in investigating the ligand substitution kinetics of aquapentacyanoruthenate(II) with some nitrogen heterocyclic ligands 6 and some naphthalene-substituted ligands. 9 In order to strengthen our previous proposed mechanistic scheme, we considered it worthwhile to investigate further the kinetics and mechanism of the aquapentacyanoruthenate(II) anion with the antitubercular drug isoniazid [INH] as a ligand, leading to the formation of [Ru(CN)5INH]3−, as a function of pH, concentration of [Ru(CN)5H2O]3−, [INH], ionic strength and temperature at 502 nm (λmax of [Ru(CN)5INH]3− complex). The product composition has been found to be 1:1 by the mole ratio and slope ratio methods. The structure of the complex is shown in Figure 1.
Structure of the product, [Ru(CN)5(INH)]3−.
Experimental
Materials and methods
All chemicals used were of analytical reagent grade and used as received. All solutions were prepared by weighing them accurately in doubly distilled deionized water. K4Ru(CN)6·3H2O (Alfa Aesar), INH (S D Fine Chem Ltd), NaClO4 (Aldrich) and KBr (S D Fine Chem Ltd) were used. Using bromine from an ampoule, a 10−2 M stock solution of bromine was prepared and standardized regularly against a standard solution of sodium thiosulphate (Sarabhai M Chemicals), using starch and KI solution as an indicator (as reported in the literature). 43 A 10−3 M stock solution of INH and a 0.2 M stock solution of NaClO4 were prepared by directly weighing the compound. All the prepared stock solutions were wrapped in aluminium foil to avoid any photodecomposition.
A solution of [Ru(CN)5H2O]3− was prepared by rapid aquation of the [Ru(CN)6]4− ion by mixing equimolar concentrations of K4[Ru(CN)6] and bromine, in the presence of a 10-fold excess of potassium bromide by the method described in Johnson and Shepherd. 44 Standard BDH buffers were used to standardize the pH meter before use. Adjustment of the pH up to the desired value was attained, using KCl/HCl or potassium hydrogen phthalate and HCl/NaOH buffers. NaClO4 was used to maintain the ionic strength of the reaction mixture.
Kinetic measurements
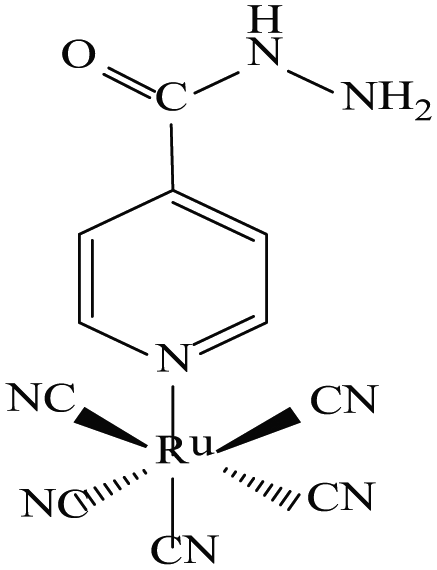
All kinetic runs were performed, under pseudo first-order conditions, by taking [INH] in at least a 10-fold excess over [Ru(CN)5H2O]3− in aqueous medium at 25°C. Solutions of the desired concentrations were obtained by accurate dilution of their stock solutions whenever required. A double-beam UV-Vis spectrophotometer (Systronics-2203) was used to record the spectra. A single-beam visible spectrophotometer model DIGI-110 (SISCO) equipped with a circulatory arrangement of water for thermostating the cell compartment was used to observe the kinetics of the reaction between [Ru(CN)5H2O]3− and INH spectrophotometrically. All kinetic measurements were performed at 502 nm (λmax of light yellow [Ru(CN)5INH]3− formed). All pH measurements were made on a Systronics µ pH System (Model-361). No absorbance correction was made since at this wavelength the only strongly absorbing species is [Ru(CN)5INH]3− and absorbances due to other reactants, namely, [Ru(CN)5H2O]3−, NaClO4 and INH, have negligible values at 502 nm (Figure 2).
Absorption spectrum of the product [Ru(CN)5INH]3− under the conditions: [Ru(CN)5H2O]3− = 1 × 10−4 M, [INH] = 4 × 10−3 M, ionic strength = 0.2 M (NaClO4), temperature = 25.0 ± 0.01°C and pH = 4.0 ± 0.02.
In order to maintain thermal equilibrium, all reactant solutions were thermally equilibrated at 25.0 ± 0.01°C, placing them in a self-designed thermostat prior to the start of reaction for about 30 min. All the thermally pre-equilibrated reagent solutions were mixed (2 cm3) in the order of [Ru(CN)5H2O]3−, NaClO4 and the ligand INH into a 50 mL Borosil flask. The mixture was shaken thoroughly and quickly transferred into a 10 mm quartz cuvette cell in a temperature-controlled cell compartment of the spectrophotometer.
The absorbance at time t (At) was measured at 502 nm and that at infinite time (A∞) was measured after completion of the reaction by heating the reaction mixture at 60°C or by keeping it overnight at room temperature. The first-order rate constants (kobs) were obtained by least-squares fits of the data by plotting ln(At – A∞) versus time t as given in equation (1)
where At is the absorbance at time t and A∞ is the final absorbance.
The standard error was found to be within ±2% during the determination of the kobs values.
Results and discussion
For the preparation of the above-mentioned complex, [Ru(CN)5INH]3−, to a solution of 0.1 mmol K4[Ru(CN)6]·3H2O in 5 cm3 of water, 1 mmol INH was added and further, 1.7 cm3 of aqueous bromine solution (0.0117 M in Br2 and 0.0117 M in KBr) was added slowly dropwise. Then, at room temperature, the mixture was left for 30 min and more INH was added (1 mmol) to this solution. After 24 h, the solution was made to evaporate in a rotary evaporator at 25°C, until the volume reduced to 1 cm3. Upon the addition of 5 cm3 of acetone to the reaction mixture at low temperature (0°C), a precipitate was obtained. The synthesized solid exhibited hygroscopic properties and so it was stored under CaCl2. 45 The product was further characterized using elemental analysis along with other techniques such as infrared (IR) spectroscopy, mass spectrometry and nuclear magnetic resonance (NMR) spectroscopy. Finally, the formed product was identified as the [Ru(CN)5INH]3− complex (as given in Scheme S1 in the supplemental information).
Elemental composition of the complex [Ru(CN)5INH] 3− . Yield: 68%–72% (0.268 g). Anal. calcd for C11H7N8ORu: C, 35.06; H, 1.88; N, 30.16; found: C, 35.87; H, 1.92; N, 30.42%.
[Ru(CN)6]4− reacts with Br2 very rapidly and produces a pale yellow coloured solution of the aquapentacyanoruthenate(II) ion [Ru(CN)5H2O]3−, as given in equation (2) 44
This pale yellow solution of the aquapentacyanoruthenate(II) ion, formed during the reaction, showed λmax at 310 nm. The [Ru(CN)5H2O]3− generated above reacts with INH and forms the [Ru(CN)5INH]3− complex which follows the Beer–Lambert law at 502 nm (molar extinction coefficient (ε) at 1640 ± 50 M−1 cm−1) over a wide range of concentrations. The [Ru(CN)5H2O]3− ion undergoes a slow dimerization reaction at higher concentrations (>10−4 M) (Kd ≈ 10−2 M−1 s−1 at pH = 7, 25.0°C) to yield most probably a cyanide-bridged Ru2(CN)106− ion, 46 as reported in the case of the pentacyanoiron(II) system. 47
The dependence of reaction rate on various reaction variables is discussed below.
The IR spectrum
The IR spectral study can be achieved in vapour, liquid, solution and in the solid phase. Since water shows absorption near 3710 and 1630 cm−1, the sample used should be completely dried. To record the spectrum of a solid sample, 1–3 mg of compound and 100–200 mg of alkali halide (KBr or KCl) were mixed together in powder form, dried further to remove moisture and pressed under pressure to produce a small transparent disc (pellet), which produces a clean spectrum. The IR spectrum of the product (KBr disc) showed strong bands at 3302.13, 3111.18, 2054.19 and 1668.43 cm−1 together with other prominent bands at 1635.64, 1556.55, 844.82 cm−1, etc., (see supplemental material in Figure S1 and Table 1). The presence of strong absorption bands at 3049.46 and 3014.74 cm−1 for asymmetric and symmetric stretching vibrations, respectively, shows the presence of a free amino (NH2) group. The very sharp band present at 1668.43 cm−1 indicates the presence of a carbonyl group in the complex. The small band observed at 530 cm−1 corresponds to the formation of a Ru–N bond in the product. 48 Therefore, the spectrum suggests that ruthenium(II) is likely to be coordinated through the nitrogen atom of the pyridine ring due to the strong coordinating ability of the nitrogen atom.
IR absorption bands (cm−1) and assignments for the [Ru(CN)5INH]3− complex.

IR: infrared.
NMR spectrum
The NMR spectrum of the complex, (see Figure S2 in the supplemental material), gave chemical shifts at 8.62 and 7.64 ppm, respectively. The first value is attributed to the protons, attached directly to the electronegative nitrogen atoms. This high value is due to the deshielding effect of the electronegative nitrogen atom of the pyridine ring as compared to the second value, that is, 7.64 ppm, for subsequent protons attached at vicinal carbon atoms. Since D2O was the solvent used, which leads to the rapid exchange of NH and NH2 protons with the deuterium atoms, this suppresses the peaks of NH and NH2 protons in the spectrum. All values are tabulated in Table S1 in the supplemental material.
Mass spectrum
The electrospray ionization-mass spectrum of the formed complex is shown in Figure S3 in the supplemental material. The major peak, [M + H]+, recorded at m/z = 365.00, confirms the stoichiometry of the complex formed. Some deviations, observed in the first-order behaviour of reactions of the [Ru(CN)5L]n− complex, were attributed to possible side reactions such as cyanide substitution and dimer formation. 49 The peak at m/z = 684 is due to the substitution of cyanide ion by excess INH and water, and the other peaks at m/z = 536 and 610 are due to the formation of dimers as well as substitution of cyanide.
Effect of pH on initial rate of reaction
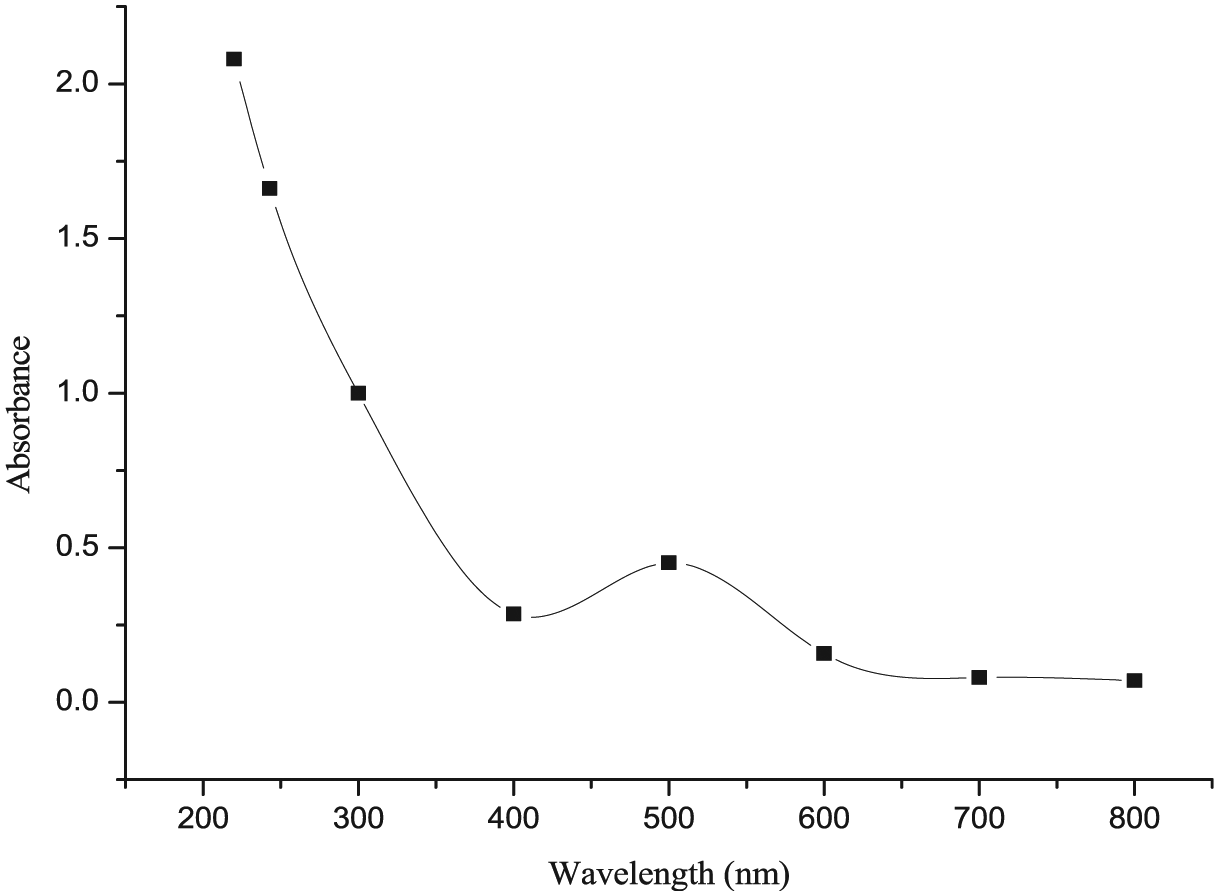
In order to select a suitable pH value, corresponding to the optimum rate of reaction, the pH of the reaction mixture was varied from 2.0 to 8.0, keeping all the other reaction variables constant. The pH up to 6 was varied using potassium hydrogen phthalate/NaOH or HCl buffer. A higher pH of all the working solutions was maintained using 5 M NaOH. The effect of pH on the rate of reaction was studied, using the fixed time procedure method as a measure of the initial rate. For each pH, the values of kobs and kf were determined and kf (kf refers to formation rate constant) was plotted against pH as shown in Figure 3 which indicates that the rate of reaction was initially low at low pH and increased with increasing pH value up to 4.0 ± 0.02, became maximum and then decreased slowly.
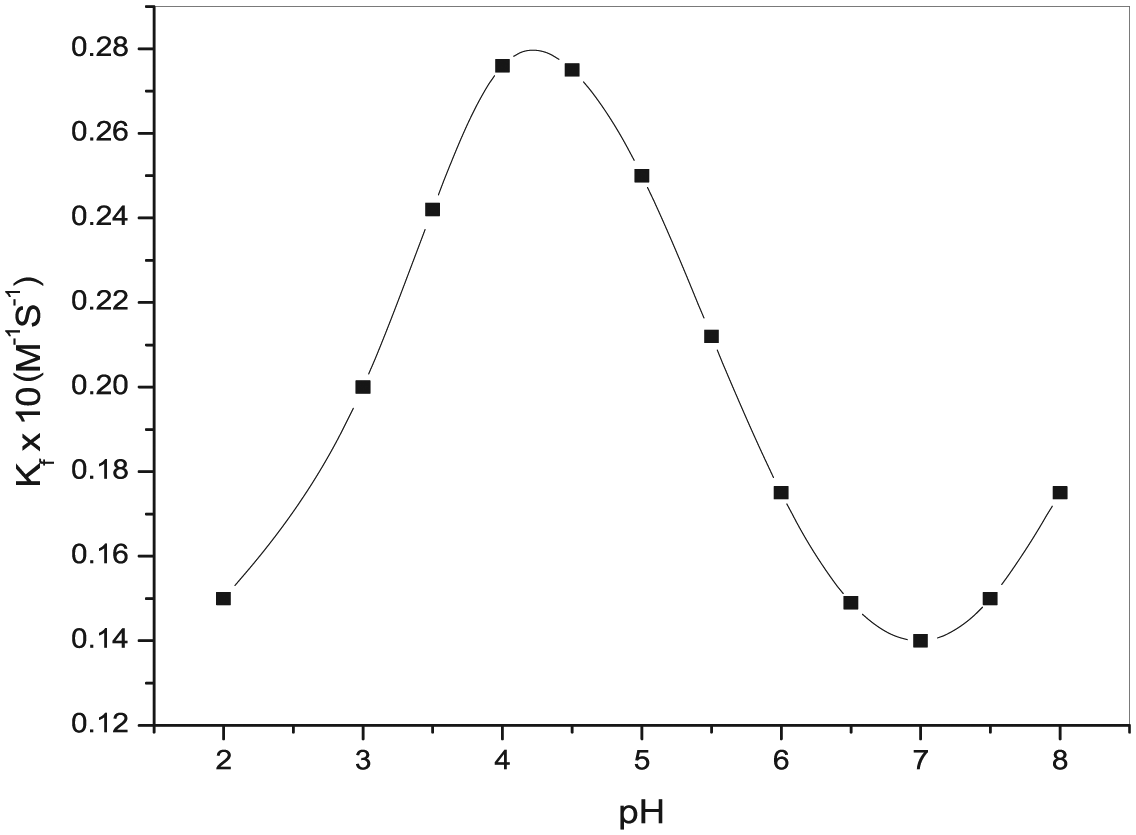
Effect of pH for the reaction of [Ru(CN)5H2O]3− with isoniazid ligand in aqueous medium under the optimum reaction experimental conditions: [INH] = 4 × 10−3 M, [Ru(CN)5H2O]3− = 1 × 10−4 M, ionic strength = 0.2 M and temperature = 25.0 ± 0.01°C.
The equilibrium constant values for protonation of the hexacyanoruthenate(II) ion have still not been reported in the literature, but they are assumed to follow the same pattern as in the case of H+[Fe(CN)6]4− equilibria. 50 At low pH, the reduction observed in rate and ultimately in the kf values is attributed to the formation of various much less reactive protonated species of [Ru(CN)6]4−,51 in comparison to [Fe(CN)6]4−. Two protonated species of [Ru(CN)6]4−, namely, monoprotonated H[Ru(CN)6]3− and diprotonated H2[Ru(CN)6]2−, have their protonation constants known, 51 while two other protonated species of [Ru(CN)6]4−, namely, H3[Ru(CN)6]1− and H4[Ru(CN)6], have their protonation constants still unknown in the literature.
This is the reason that distributions of all the species of the hexacyanoruthenate(II) ion (for protonated and deprotonated forms) as a function of pH have not been obtained. However, as provided in the literature, on the basis of various species of [Fe(CN)6]4− and dissociation constants of INH, it is quite reasonable to say that deprotonated forms of [Ru(CN)6]4− and INH are found to be the active species at a pH value of 4.00. Hence, it supports the justified criteria for the constancy of the initial rate in the pH range between 2.0 and 8.0.
Effect of ligand [INH]
The effect of variation of INH on the initial rate was determined by varying its concentration from 4 × 10−3 to 15 × 10−4 M by keeping the fixed optimum conditions for other reaction variables. The plot of kobs versus [INH] was a straight line (R2 = 0.9891), as shown in Figure 4.
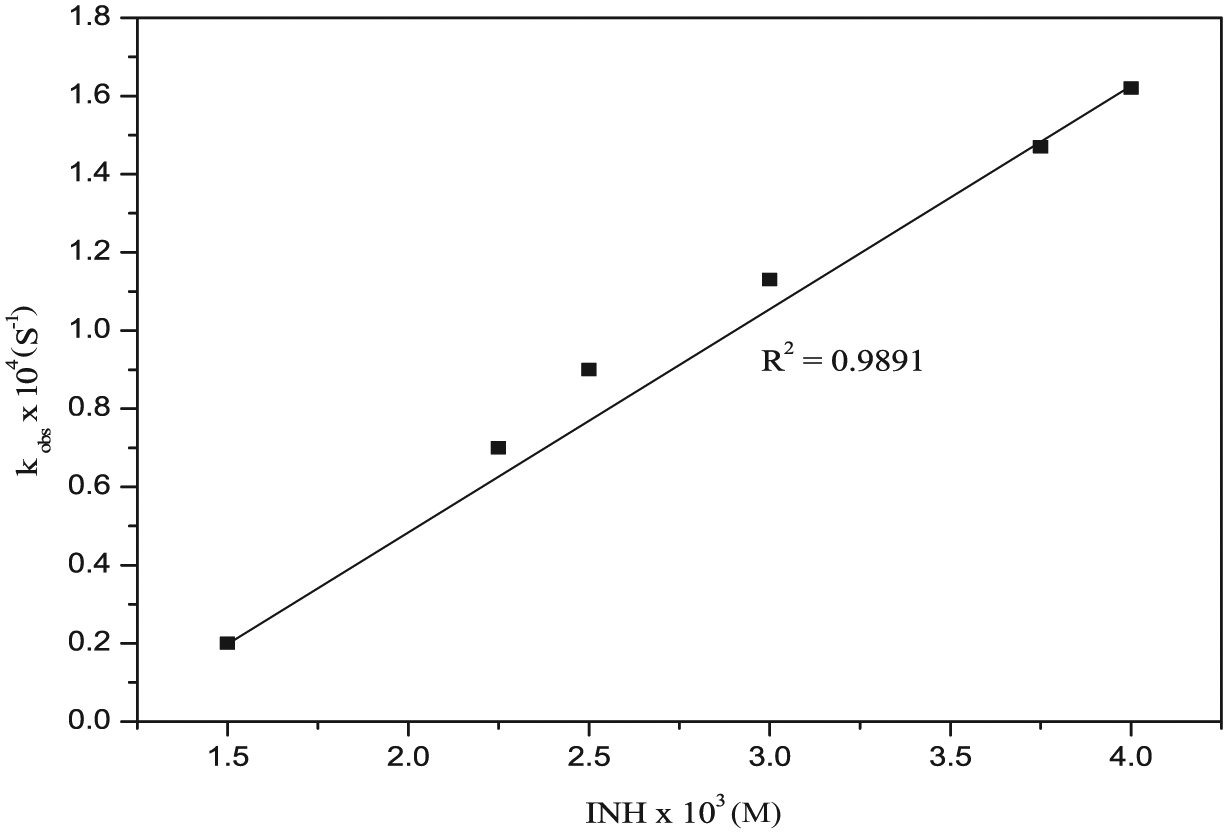
Effect of ligand [INH] for the reaction of [Ru(CN)5H2O]3− with isoniazid in aqueous medium under the optimum reaction experimental conditions: [Ru(CN)5H2O]3− = 4 × 10−4 M, pH = 4.0 ± 0.02, ionic strength = 0.2 M and temperature = 25.0 ± 0.01°C.
The reaction is considered to proceed through equations (3) and (4) as reported in our earlier investigation 9
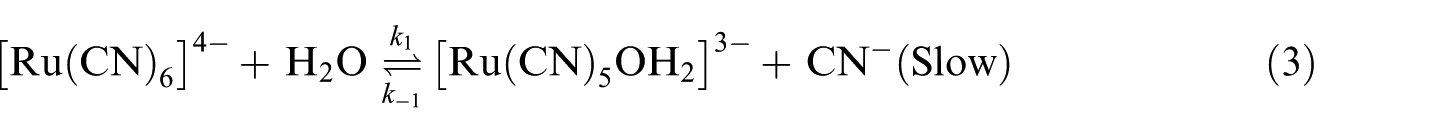
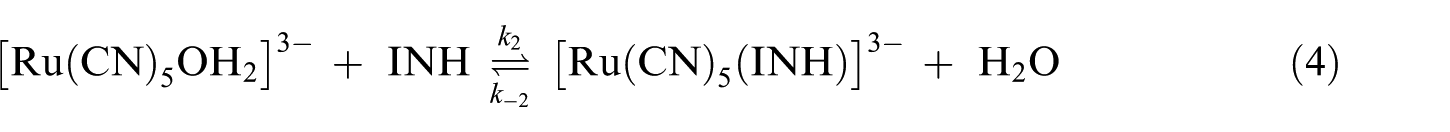
The lack of intercept in Figure 4 suggests the minimum role of the reverse reaction in equation (4). A similar route for substitution reactions of [Ru(CN)5L]3− has also been observed with some nitrogen-containing heterocyclic ligands. 52
Effect of complex concentration
To study the effect of variation of concentration of [Ru(CN)5H2O]3−, the rate of reaction was measured as a function of [Ru(CN)5H2O]3−, by changing its concentration in the range 4 × 10−4–2 × 10−4 M, keeping all other experimental variables fixed at optimum values. The rate of reaction was found to be invariant with the concentration of [Ru(CN)5H2O]3−. The variation of kobs with [Ru(CN)5H2O]3− is shown in Table S2 in the supplemental information.
Effect of ionic strength (µ)
The effect of ionic strength variation on the rate of reaction was studied by varying the concentration of NaClO4 within the experimental range 0.025–0.2 M, keeping all other experimental variables fixed at their optimum values. The plot of ln(kf) versus I1/2/(1 + I1/2) was found to be linear (Figure 5) (R2 = 0.9988). The graph was in accordance with the Brønsted–Bjerrium–Christiansen relationship 53 and found to exhibit a negative salt effect, that is, as the ionic strength of the medium increases, the rate of reaction decreases.
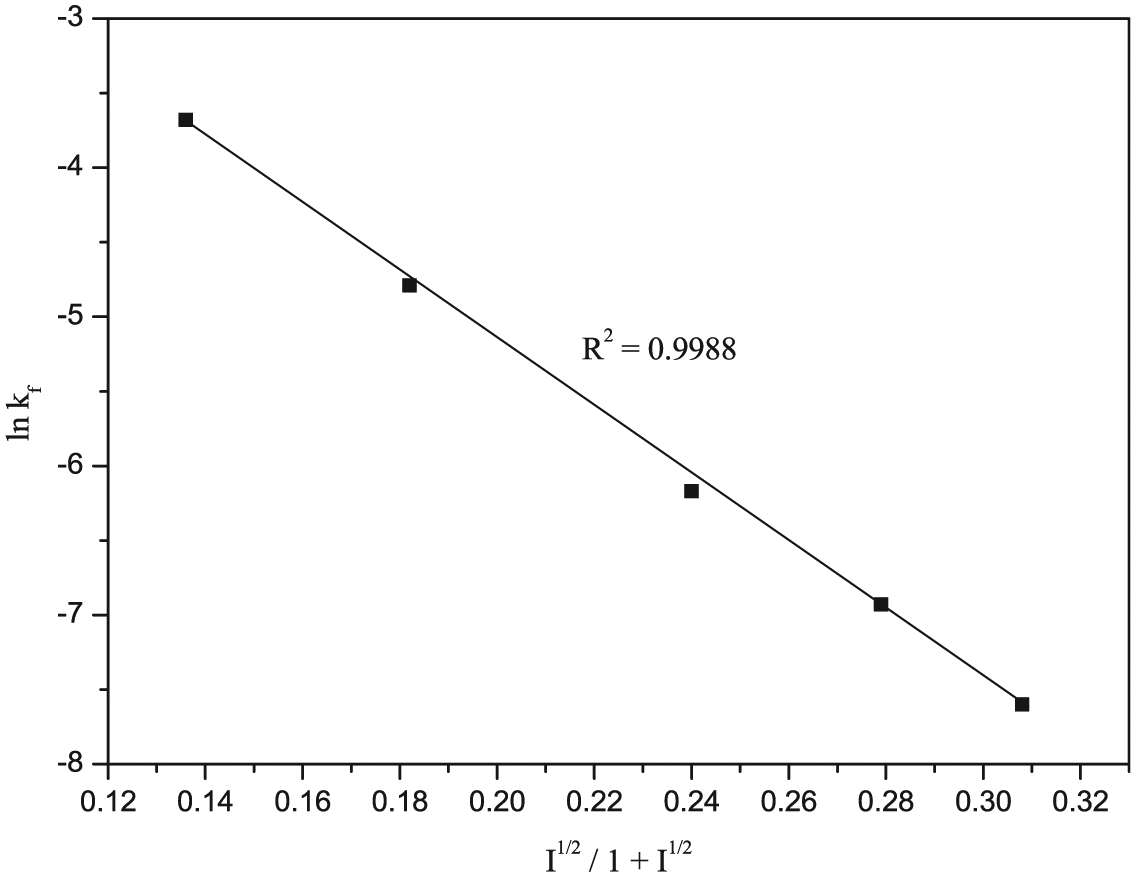
Effect of ionic strength for the reaction of [Ru(CN)5H2O]3− with isoniazid in aqueous medium under the optimum reaction experimental conditions: [INH] = 4 × 10−3 M, [Ru(CN)5H2O]3− = 4 × 10−4 M, pH = 4.0 ± 0.02 and temperature = 25.0 ± 0.01°C.
Effect of temperature
The effect of variation of temperature on the reaction rate was analysed in the range 303–318 K, keeping other reaction variables fixed. A higher temperature was avoided to reduce the possibility of decomposition of the product. The values of the forward rate constant (kf) were determined at different temperatures. The reaction followed Eyring's equation. The forward rate constant (kf) is related to the entropy of activation (ΔS≠) and enthalpy of activation (ΔH≠) through Eyring's equation, given in equation (5)

The plot of ln (kf/T) versus 1/T exhibited a straight line as shown in Figure 6 (R2 = 0.9985). The values of activation parameters (ΔH≠ and ΔS≠) were estimated from the slope and intercept of the above plot and are tabulated in Table 2. The high positive ΔS≠ value represents a greater degree of bond breaking (
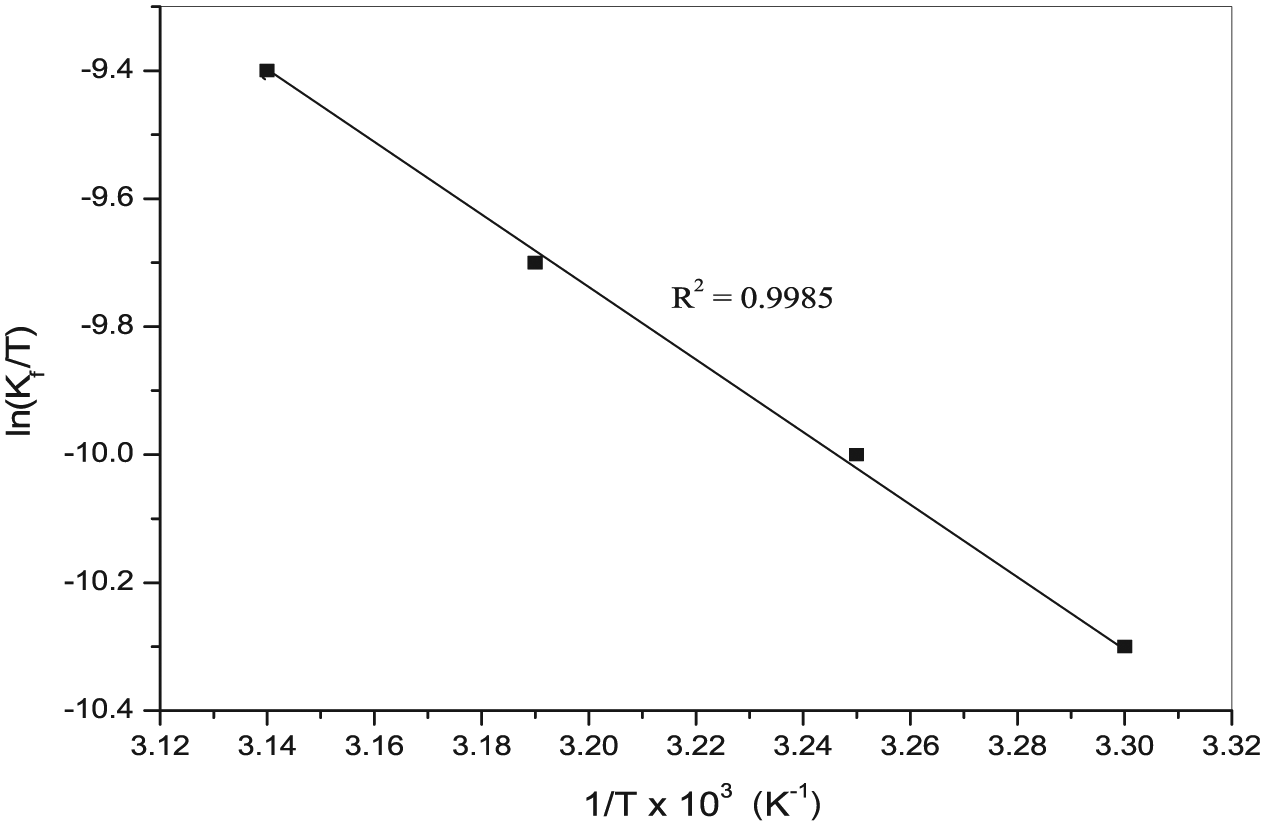
Effect of temperature for the reaction of [Ru(CN)5H2O]3− with isoniazid in aqueous medium under the optimum reaction experimental conditions: [INH] = 4 × 10−3 M, [Ru(CN)5H2O]3− = 4 × 10−4 M, pH = 4.0 ± 0.02 and ionic strength = 0.2 M (NaClO4).
Thermodynamic activation parameters: enthalpy (ΔH≠, kJ mol−1), energy of activation(Ea, kJ mol−1), Gibbs free energy (ΔG≠, kJ mol−1) and entropy (ΔS≠, J K−1 mol−1), for the cyanideion–substituted reaction between [Ru(CN)5H2O]3− and isoniazid (INH) ligand in aqueous mediumunder the optimum experimental conditions and their change for reaction at 25.0 ± 0.01°C.

Mechanism
On the basis of kinetic data and activation parameters, the reaction under study preferentially takes place through an ion-pair dissociation mechanism. The most plausible mechanism is considered to take place through equations (6) and (7)
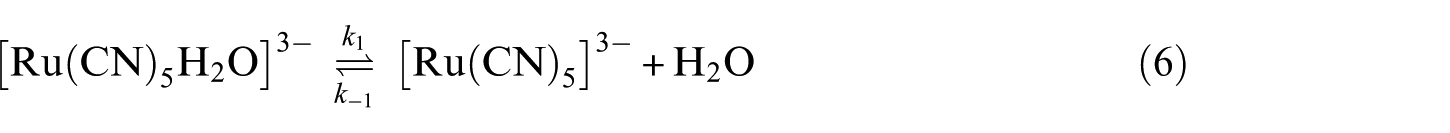

where n is the charge on the incoming ligand.
After applying the steady-state approximation for [Ru(CN)5]3−, one can deduce equation (8)
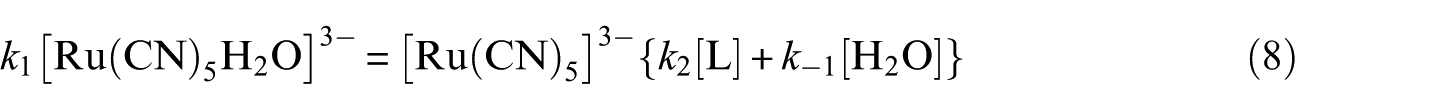
After simplifying equation (8), we obtain the concentration of

Since, according to equation (7), the rate of reaction depends on the concentration of intermediate and ligand, it can be expressed through equation (10)

After solving equations (9) and (10), equation (11) is deduced

Since the rate of reaction is directly proportional to the concentration of complex and ligand, we can easily represent the rate through equations (12) or (13)


After substituting equation (13) into equation (11), the value of kobs can be represented through equation (14)

Under the present experimental conditions

Therefore, equation (14) reduces to equation (15)

By considering

equation (15) can be transformed into equation (16)

This equation proves that the observed rate constant follows the first-order rate law. The plot of variation of the concentration of [INH] with kobs (kobs vs [INH]; Figure 4) shows a straight line having no intercept, which is in good agreement with equation (16). The values of kf = kap are obtained from the slope of the plot.
Conclusion
This work describes a successful method for the preparation of the [Ru(CN)5INH]3− complex which could be a better medication than INH itself for the treatment of M. tuberculosis like its [Fe(CN)5INH]3− counterpart. Further studies will be undertaken in our laboratory in this regard. A tentative mechanistic scheme for the substitution of one of the six coordinated cyanides in the hexacyanoruthenate(II) ion by the INH ligand has been proposed. The structural integrity of the synthesized complex has been made using IR, NMR, mass spectrometry and elemental analysis.
Supplemental Material
Supplementary_Information – Supplemental material for Kinetics and mechanism of formation of the complex [Ru(CN)5INH]3− through the ligand substitution reaction between the aquapentacyanoruthenate(II) anion and isoniazid
Supplemental material, Supplementary_Information for Kinetics and mechanism of formation of the complex [Ru(CN)5INH]3− through the ligand substitution reaction between the aquapentacyanoruthenate(II) anion and isoniazid by Rupal Yadav and Radhey Mohan Naik in Progress in Reaction Kinetics and Mechanism
Footnotes
Acknowledgements
The authors thank the Head, Department of Chemistry, Lucknow University, Lucknow, for providing required departmental facilities to carry out the research work. The authors are grateful to the Director of CDRI, Lucknow, for spectral analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.