Abstract
Photoelectrochemical biofuel cells can convert light and chemical energy into electrical energy using a dye-sensitized titania (TiO2) fluorine-doped tin oxide photoanode and a platinum-coated fluorine-doped tin oxide cathode. TiO2 of the photoanode serves both as a support for dyes and as an electron-transporting medium, the structure of which can limit electron trapping and charge transporting and then affect the performance of the photoelectrochemical biofuel cells. TiO2 nanotube array films have been shown to enhance the efficiencies of both charge collection and electron injection, and hence a vertically aligned TiO2 nanotube array is investigated as a conductor for the tetrakis(4-carboxyphenyl)porphyrin dye to construct a new two-compartment photoelectrochemical biofuel cell. The photoelectrochemical biofuel cell containing the TiO2 nanotube array photoanode yields a short-circuit (Isc) current of 110 μA and an open-circuit (Voc) potential of 1010 mV. In contrast, the photovoltaic parameters, Isc and Voc of the photoelectrochemical biofuel cell with the mesoporous TiO2 nanocrystal fluorine-doped tin oxide photoanode, are 96.96 μA and 740 mV, respectively. Photovoltaic measurements show that the maximum incident photon-to-collected electron conversion efficiency was 58% at 430 nm through the spectral range (400–800 nm) for the photoelectrochemical biofuel cell with the TiO2 nanotube array fluorine-doped tin oxide photoanode. These results revealed that the TiO2 nanotube array had great potential for the photoelectrochemical biofuel cells.
Keywords

Photoelectrochemical biofuel cell with the TiO2 nanotube array photoanode was fabricated and characterized by photocurrent action spectra and current–voltage measurements, and it showed excellent photoelectric conversion performance.
Introduction
Photoelectrochemical biofuel cells (PEBFCs) have received considerable attention as they are able to convert light and chemical energy into electrical energy.1–8 PEBFCs use the same dye-sensitized photoanode and cathode, but the electrolyte is different from that of dye-sensitized solar cells (DSSCs),1–8 as shown in Figure 1, which has been published in other journals. 9 PEBFCs represent an exciting photovoltaic cell, since they combine the attractive features of an enzyme-catalyzed biofuel cell (BFC), while overcoming the organic solvent limitation of DSSCs, and also provide an ingenious idea for designing light-driven biosensing based on the cell. Biosensors based on BFC or enzymatic fuel cell (EFC) –— substantial attentions because of their unique merits such as simplicity, rapidness, low cost, anti-interference, and ease of use.10–15 However, various efforts toward efficiency improvement in PEBFCs are directed toward the development of dye-sensitized photoanodes which influences the absorption of photons and transfer of electrons. For PEBFCs, the photoanode features dyes adsorbed onto the surface of mesoporous titania (TiO2) nanocrystals. Typical dyes used with PEBFCs are porphyrin compounds because they possess an intense Soret band at 400–450 nm and moderate Q-bands at 500–650 nm. We have previously reported PEBFCs based on porphyrin compounds that exhibited excellent photovoltaic performance.16–19 In addition, photoanodes basing on the photosensitization of a copper chlorophyll trisodium salt 20 or [Ru(H2dcbpy)3]Cl2 (H2dcbpy = 2,2ʹ-bipyridine-4,4ʹ-dicarboxylic acid) 21 have good photochemical and photoelectrochemical properties. Despite achieving high conversion efficiencies, their performance is limited by electron trapping in the nanoparticle film. For the photoanode, mesoporous TiO2 nanocrystals offer longer electron diffusion lengths and shorter electron transport times, which degrade the efficiency of the PEBFC. To achieve higher efficiency for PEBFCs, research efforts have focused on improving the transport and recombination of the electrons which are major determinants of the overall efficiency. Electron diffusion lengths can be increased by transporting the charge through highly ordered nanostructure such as a TiO2 nanotube array.16–18 One of the promising approaches is to replace the mesoporous TiO2 nanocrystal by a TiO2 nanotube array with high optical absorption efficiency and longer stability.22–24 Recently, TiO2 nanotube arrays have been used to increase electron collection efficiency by reducing the undesirable loss of electrons at the boundaries via recombination with redox couples.25–29

Schematic diagram of the PEBFC.
In this paper, electron-transporting TiO2 nanotube arrays have been used in the photoanode of PEBFCs. Photoanodes were fabricated by coating tetrakis(4-carboxy-phenyl)porphyrin (TCPP) onto the TiO2 nanotube array fluorine-doped tin oxide (FTO) electrode. The PEBFC based on the TiO2 nanotube array photoanode demonstrated a short-circuit current (Isc) and open-circuit potential (Voc) of 110 μA and 1010 mV, respectively. Peaks in the photocurrent action spectrum were observed at 430, 520, and 660 nm and the incident photon-to-collected electron conversion efficiency (IPCE) values were estimated to be ca. 58%, 26.2%, and 16.6%, respectively. The experimental data revealed that TiO2 nanotube arrays are attractive electrode materials for PEBFCs.
Results and discussion
X-ray diffraction and field-emission scanning electron microscope
Typical X-ray diffraction (XRD) patterns of TiO2 are given in Figure 2. Both the TiO2 nanotube array and the mesoporous TiO2 nanocrystal showed six well-resolved peaks, which could be readily assigned to (101), (004), (200), (105), (211), and (204), respectively. These peaks could be assigned to the tetragonal anatase TiO2 (I41/and (No. 141), Joint Committee on Powder Diffraction Standards file No. 71-1168).30,31

XRD patterns of (a) the TiO2 nanotube array and (b) the mesoporous TiO2 nanocrystal.
Typical scanning electron microscope (SEM) images of the TiO2 nanotube array and the mesoporous TiO2 nanocrystal are shown in Figure 3. From Figure 3(a), the TiO2 nanotubes had the general shape of a common laboratory test tube, with the top of the tube open and the bottom closed. 24 For the mesoporous TiO2 nanocrystal, the mean particle diameter is 20 nm with a standard deviation of 6 nm for 177 particles.

SEM graphs of (a) the TiO2 nanotube array and (b) the mesoporous TiO2 nanocrystal.
UV-Vis absorption spectra of the TCPP-sensitized TiO2 electrodes
The UV-Vis absorption spectra of the dye TCPP in solution and of the TiO2 electrodes were measured so as to have a preliminary evaluation on their light-harvesting capacity (Figure 4). In Figure 4(a), the UV-Vis absorption spectrum of the dye TCPP in the ethanol and co-adsorbate solution includes a Soret band at 414 nm and Q-bands at 522, 556, 595, and 650 nm, which are in good agreement with those previously reported for similar porphyrins.32–39 The dye TCPP absorption peak observed in the ethanol solution broadens with sensitization of the TiO2 FTO electrodes, as shown in Figure 4(b) and (c). The UV-Vis absorption spectrum of the TCPP coated on the TiO2 nanotube array FTO electrode demonstrates the Soret band at 420 nm, which is red-shifted by 6 nm compared with that in solution; the absorption peaks in the Q-band region are at 524, 560, 600, and 654 nm (Figure 4(b)), which are red-shifted by 2, 4, 5, and 4 nm, compared with those in the solution, respectively. The absorption spectrum of the TCPP coated on the mesoporous TiO2 nanocrystal FTO electrode (Figure 4(c)) showed a strong peak at about 421 nm (Soret band) and four peaks at 526, 563, 601, and 657 nm (Q-bands), which showed red shifts of 7, 4, 7, 6, and 7 nm, respectively, compared with the absorption peak of the TCPP in the solution. For TCPP coated on the mesoporous TiO2 nanocrystal FTO electrode, it is obvious that the value of the shift is higher than that for TCPP coated on the TiO2 nanotube array FTO electrode.
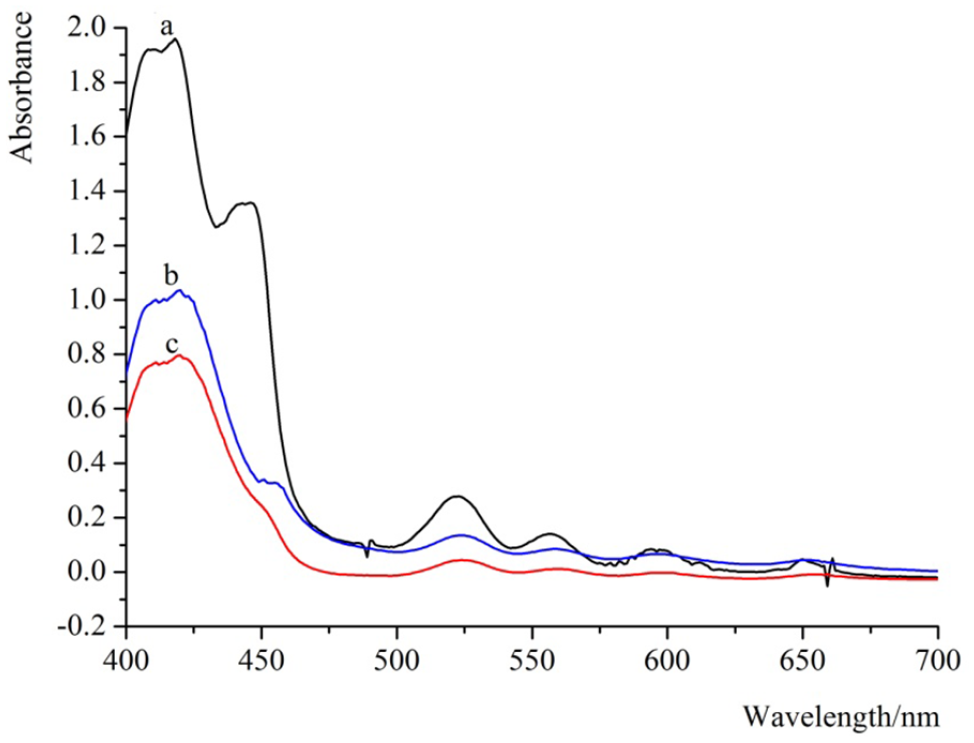
Absorption spectra of (a) 1.5 × 10−4 M TCPP in ethanol solution containing 1 × 10−3 M 3α,7α-dihyroxy-5β-cholic acid, (b) TCPP coated on the TiO2 nanotube array FTO electrode, and (c) the mesoporous TiO2 nanocrystal FTO electrode.
Features of the FTIR spectra
The above results obtained for the UV-Vis absorption spectra have proved that there is a substantial interaction between the dye TCPP and TiO2, but the modes of the interaction are not yet clear. Thus, to investigate the coordination states of the dyes on the TiO2 surfaces, the FTIR spectra of the dyes in the solid state and the adsorption state on TiO2 were measured. Figure 5 shows the FTIR absorption spectra of the dyes over the range of 500–4000 cm−1 at room temperature, and also, for comparison, displays the FTIR spectra of TCPP dye powder and the dye coated on the TiO2 electrodes. The strong C=O band due to carboxyl group was observed at 1691 cm−1 for TCPP in the solid state (Figure 5(a)). When TCPP was adsorbed on the mesoporous TiO2 nanocrystals, the intensity of the C=O band decreased and two intense bands appeared at 1639 and 1399 cm−1, which were assigned to the antisymmetric (υasym–CO2−) and symmetric (υsym–CO2−) carboxylate vibrations (Figure 5(b)). When TCPP was adsorbed on the TiO2 nanotube array, the antisymmetric (υasym–CO2−) and symmetric vibrations of (υsym–CO2−) were at 1644 and 1409 cm−1. The splitting of the carboxylate stretching bands [Δ = (υasym–CO2−) − (υsym–CO2−)] in the FTIR spectra of metal complexes with carboxyl groups can be used to analyze the classification of possible modes during the coordination of the carboxyl group to the metal.40–42 While the dye with the carboxyl group is coordinated via bridging or bidentate chelate with TiO2, the splitting of the carboxylate stretching bands is Δ = 248 cm−1. 43 In our study, the splitting of TiO2/TCPP was located at Δ = 240 and 235 cm−1, which are very close to the above value; so the carboxyl group of the TCPP is also coordinated by bridging or bidentate chelating modes with TiO2.
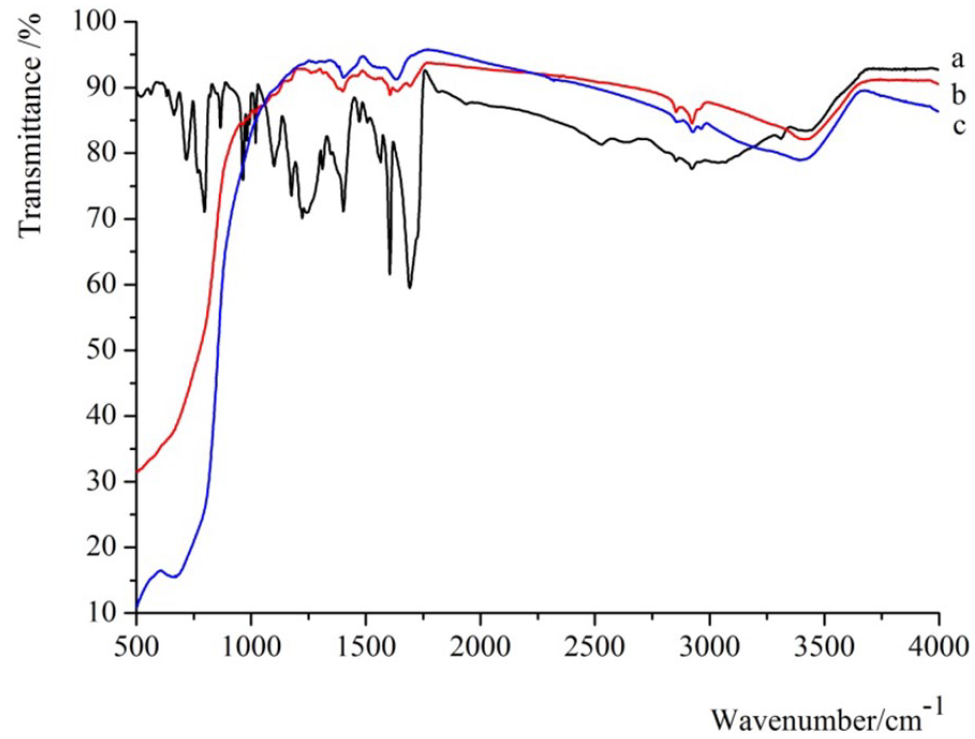
FTIR spectra of (a) TCPP dye, (b) the dye coated on the mesoporous TiO2 nanocrystal FTO electrode, and (c) the dye coated on the TiO2 nanotube array FTO electrode.
Photocurrent action spectrum
To evaluate how the TiO2 structure affects the performance of the PEBFC, the photocurrent action spectra for PEBFCs with the TiO2 nanotube array FTO electrode and the mesoporous TiO2 nanocrystal FTO electrode sensitized by TCPP were obtained and are shown in Figure 6. The shapes of the action spectra obtained were similar but slightly broader than those of the absorption spectrum. The onset wavelengths of the IPCE spectra for the PEBFCs were less than 800 nm and the IPCE had a significant magnitude up to 700 nm. The IPCEs of the PEBFC with the TiO2 nanotube array at wavelengths are much higher than those of the PEBFC with the mesoporous TiO2 nanocrystal. Absorption peaks in the photocurrent action spectra for the PEBFC with the mesoporous TiO2 nanocrystal were observed at 436, 520, and 656 nm, and the corresponding IPCEs were ca. 36.0%, 24.0%, and 8.0%, respectively. For the PEBFC with the TiO2 nanotube array, the corresponding IPCE values at 430, 520, and 660 nm were 58%, 26.2%, and 16.6%, respectively. These results indicate that the photovoltaic performance of the TiO2 nanotube array is better than that of the mesoporous TiO2 nanocrystal. One of the reasons is that the TiO2 nanotube arrays have a larger surface area (as shown in Figure 7), significantly higher charge-collection efficiencies, and better light-harvesting efficiency than the mesoporous TiO2 nanocrystals. Another reason could be the difference in the electron injection, charge separation, and transfer efficiency at the interface, namely, the vertically oriented TiO2 nanotube arrays of the regular surface decouple exciton diffusion from light absorption and facilitate the injection and transfer of the electron, while the mesoporous TiO2 nanocrystals of the random network lead to recombination of photo-generated electrons and holes at the boundaries.22–24

Photocurrent action spectra of the photoelectrochemical biofuel cells with the TiO2 nanotube array FTO electrode (solid line) and the mesoporous TiO2 nanocrystal FTO electrode (dashed line).

Schematic diagram of the photoanode.
Current–voltage curve
The photoanodes were illuminated with 2.33 mW/cm2; the current–voltage curves obtained for the PEBFCs are shown in Figure 8. The photoelectrochemical characteristics for the two PEBFCs were quite different. The short-circuit current (Isc), the open-circuit potential (Voc), the maximum power density (Pmax), the fill factor (FF), and the power output (P) of the PEBFC fabricated from the mesoporous TiO2 nanocrystal FTO electrode were 96.96 μA, 740 mV, 0.67 μW/cm2, 0.43, and 0.19 μW, respectively. In contrast, the photovoltaic parameters, Isc, Voc, Pmax, FF, and P of the PEBFC with the TiO2 nanotube array FTO electrode, were 110 μA, 1010 mV, 59 μW/cm2, 0.52, and 16.70 μW. The photovoltaic parameters of the two PEBFCs were listed in Table 1. Obviously, the electrochemical parameter values for the PEBFC with the TiO2 nanotube array FTO electrode were greater than those of the PEBFC with the mesoporous TiO2 nanocrystal FTO electrode. The reason for this difference in the characteristics of the two PEBFCs is considered to be the difference in the electron transport and the electronic injection efficiency, as mentioned above. The increase in the PEBFC performance relies largely on the highly ordered material architectures offering longer electron diffusion lengths and shorter electron transport times than those in conventional randomly oriented nanoparticle films. 44 These results indicate that the electronic injection efficiency at the interface is an important factor for the performance of the PEBFC.

Current–voltage characteristics for the PEBFCs with the TiO2 nanotube array FTO electrode (solid line) and the mesoporous TiO2 nanocrystal FTO electrode (dashed line) under light intensity of 2.33 mW/cm2.
Performances of the PEBFCs with the TiO2 nanotube array electrode and the mesoporous TiO2 nanocrystal electrode.
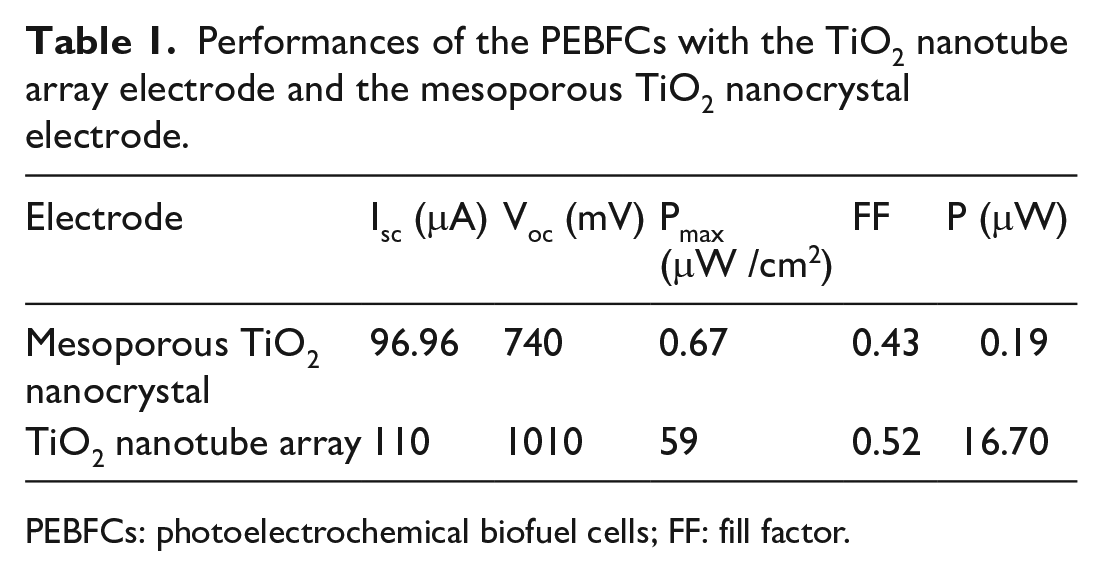
PEBFCs: photoelectrochemical biofuel cells; FF: fill factor.
Conclusion
A photoelectrochemical biofuel cell has been constructed with the TCPP photosensitization of the TiO2 nanotube arrays vertically oriented from the FTO photoanode. The Isc, Voc, Pmax, and FF values of the PEBFC based on the TiO2 nanotube array FTO photoanode were higher than those of the PEBFC based on the common mesoporous TiO2 nanocrystal FTO photoanode because of the superior charge transport. Therefore, it is believed that the breakthroughs in achieving high efficiency of PEBFCs probably derive from electrode morphology control of the TiO2 nanotube array through improvements in electron transport. Research in this aspect is under way.
Experimental
Materials
All solvents and reagents, unless otherwise stated, were of analytical grade and were used without further purification. β-Nicotinamide adenine dinucleotide, reduced form of disodium salt (β-NADH) was purchased from Sigma-Aldrich Company. The tris(hydroxymethyl)aminomethane (Tris) was purchased from J&K Chemical Ltd. Glucose dehydrogenase (GDH) was purchased from Aladdin (Changchun, China). The TiO2 nanoparticles are purchased from Fluka. The enzyme activity was assayed following a protocol provided by the manufacturer. TCPP was purchased from J&K Chemical Ltd. β-D-glucose, N,N-dimethyl formamide (DMF), and potassium chloride (KCl) were purchased from Jilin Jintai Chemical Glass Co., Ltd. (Changchun, China). 3α,7α-Dihyroxy-5β-cholic acid (cheno) was purchased from Fluka. Perfluorinated sulfonic acid proton-exchange membrane Nafion 117 (thickness: 80 µm, exchange capacity: 1.0 ± 0.02 mM/g) was obtained from Aladdin (Changchun, China). Deionized water was prepared using a Milli-Q water purification system. One unit of GDH activity is defined as the amount of enzyme per minute that reduces 1.0 mmol of NAD+ to NADH using glucose.
Preparation of the TiO2 nanotube array electrode
The common mesoporous TiO2 nanocrystal FTO electrode was prepared as follows: first, a 7-µm-thick transparent layer of TiO2 particles was deposited, via the screen-printing protocol, onto a precleaned FTO conductive glass (Nippon Sheet Glass, Solar, 4-mm-thick, resistivity, 20 Ω/sq, 10 × 10 mm2) and subsequently the second layer of scattering TiO2 particles (5-µm-thick) was coated on the first layer. 45
The TiO2 nanotube array conductive glass electrode was formed by anodizing the TiO2 films on FTO glass in a two-electrode electrochemical cell with TiO2 as the anode and platinum as the cathode, with a constant voltage across the electrodes. The TiO2 film was installed on the FTO glass electrode using direct-current (DC) magnetron sputtering of titanium targets under an argon atmosphere. For films with a thickness of 7 µm, the substrates were initially heated to 45–60°C.22–24 Furthermore, a 5-µm-thick scattering layer was used to coat the 7-µm-thick TiO2 nanotube array. Due to the scattering layer, incident light could be used many times in photoanode, which improved IPCE. 9
Characterization of TiO2
XRD measurement was performed on a Rigaku D/MAX-PC 2500 diffractometer with a Cu Kα radiation (λ = 0.15405 nm) from 20° to 80° at a scanning rate of 5°/min. The field-emission scanning electron microscope (FE-SEM) was carried out on the FE microscope (JEOL, 7500B) operated at an acceleration voltage of 10 kV.
Preparation of the platinum-coated FTO electrode
Details on the preparation of the platinum-coated FTO electrode are given elsewhere. In short, the platinum-coated FTO electrode was prepared by thermal decomposition of hydrogen hexachloroplatinate hexahydrate. 45
Fabrication of the PEBFC
The TiO2 nanotube array conductive glass electrode was heated for 30 min at 550°C and then cooled to room temperature. The above electrode was dipped for 24 h in ethanol solution containing 1.5 × 10−4 M of TCPP and 1 × 10−3 M of the co-adsorbate cheno. Fabrications of the PEBFC, the anodic compartment, and the cathodic compartment are reported elsewhere. 16
Photovoltaic characteristics
A Keithley 2400 source meter and a Zolix Omni-λ300 monochromator equipped with a 500-W xenon lamp were used for photocurrent action spectrum measurements with a wavelength sampling interval of 10 nm and a current sampling interval of 2 s under full computer control. A Hamamatsu S1337-1010BQ silicon diode was used for IPCE measurements and was calibrated at the National Institute of Metrology, China. A model LS1000-4S-AM1.5G-1000 W solar simulator (Solar Light Co., Glenside, PA, USA) was employed to supply irradiation of 100 mW/cm2. The light intensity was measured with a PMA2144 pyranometer and a calibrated PMA2100 dose control system. The current–voltage characteristics were measured with a Keithley 2602 source meter under full computer control. The measurements were fully automated using LabView 8.0 (USA).43,45,46
All photoelectrochemical measurements were carried out immediately at 25°C after the cells had been fabricated.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the Education Department of Jilin Province (No. JJKH20190450KJ).
