Abstract
In this study, the mechanism for both the Huisgen 1,3-dipolar cycloaddition and the base-catalyzed cycloaddition reactions between phenylacetylene and azidobenzene has been investigated with density functional theory, namely at the M06-2X/6-31G(d) computational level. Later, the reaction has been modeled with a representative simple alkyne and a simple azide to concentrate solely on how the difference bases affect the mechanism of the reaction between phenylacetylene and azidobenzene as charged components. In this study, another mechanism of this reaction with uncharged components has been proposed to compare the calculated thermodynamic and kinetic properties for charged and uncharged systems. The calculated activation barrier differences between the catalyzed and the uncatalyzed reactions are consistent with the faster and the regioselective synthesis of the triazole product in the presence of solvents. The calculated barrier of the rate-determining step in the base-catalyzed mechanism with the uncharged system is lower than that with charged systems. Finally, the reaction leading to final product formation in uncharged system is more spontaneous than that in the charged system, and the same applies to the total reaction in the presence of solvents.
Keywords

Introduction
1,2,3-Triazoles have been regarded as one of the most important nitrogen heterocycles because of their broad application in organic synthesis, biology, medicinal chemistry, and materials science.1–8 Therefore, tremendous attention has been focused on the development of synthetic methods for the assembly of these valuable heterocyclic compounds. As early as 1963, Huisgen had developed the thermally induced 1,3-dipolar cycloaddition of alkynes with organic azides to construct 1,2,3-triazoles, although this suffered from poor regioselectivity and low efficiency.9–11 Rostovtsev and Tornøe proposed a significant improvement to the Huisgen reaction and established the copper-catalyzed azide-alkyne cycloaddition (CuAAC) for the synthesis of 1,4-disubstituted 1,2,3-triazoles, greatly increasing the reaction rate and regioselectivity.12,13 Recently, some alternative methods have been established for the synthesis of triazole derivatives including the reactions of azides with alkenes or ketones.14–23 It was found that most of these methods often utilized significant amounts of toxic transition metal catalysts, which are not ideal for the biological applications in view of their toxicity. Therefore, a simple and green protocol is still highly desirable for the construction of triazole and its derivatives.
To treat this problem, transition-metal-free strategies have been developed for the preparation of specific 1,2,3-triazoles, such as an organocatalytic enamine-mediated amino acid or amine catalyzed [3+2] cycloaddition of different carbonyl compounds with organic azides,24–32 base-promoted oxidative catalyzed cycloaddition of chalcones with azides in aqueous solution, 33 and base-catalyzed cycloaddition with azides for the preparation of 1,5-diarylsubstituted 1,2,3-triazoles. 34 Kwok et al. showed that 1,5-diarylsubstituted 1,2,3-triazoles are formed in high yields from aryl azides and terminal alkynes in dimethyl sulfoxide (DMSO) in the presence of catalytic tetraalkylammonium hydroxide. The reaction is experimentally simple, does not require a transition-metal catalyst, and is not sensitive to atmospheric oxygen and moisture. Scheme 1 shows the proposed mechanism for this reaction. No established high-level theoretical characterizations of previously reported reaction mechanisms 34 are available in the literature. Thus, in this paper, the mechanism of the base-catalyzed reaction will be investigated in detail using density functional theory (DFT) employing the M06-3X method with the basic set 6-31g(d) in the gas phase and in the presence of some solvents.
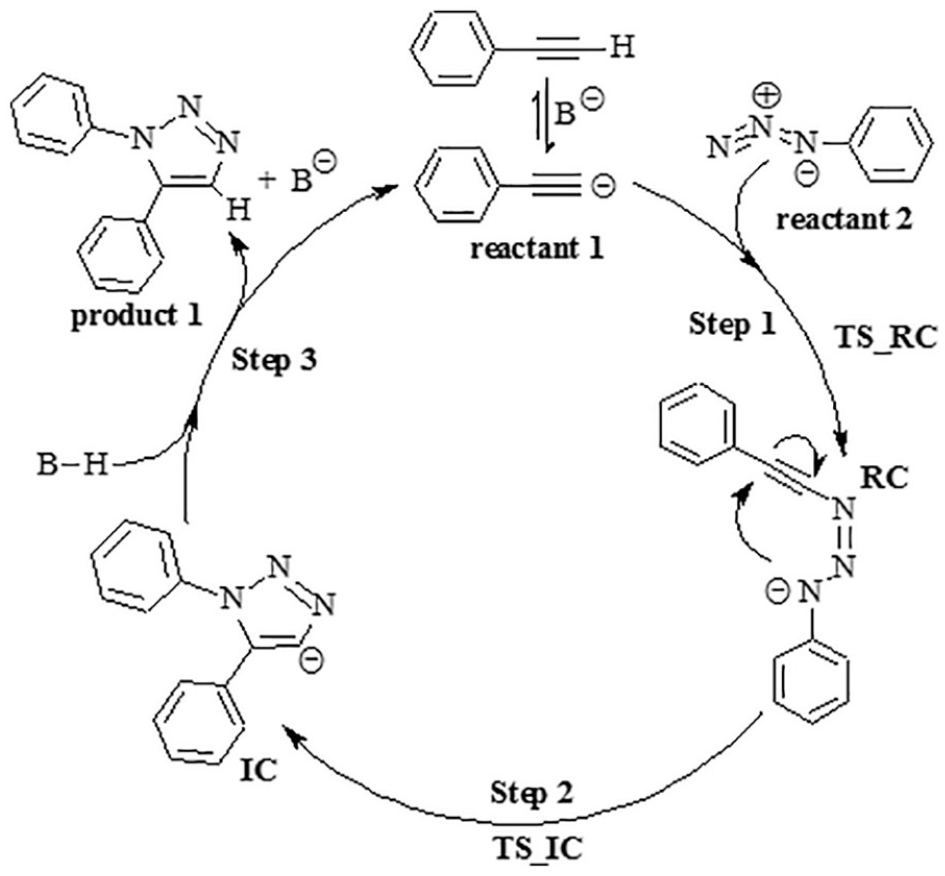
Proposed mechanism for base-catalyzed synthesis of 1,5-disubstituted-1,2,3-triazoles.
Computational methods
DFT is a valuable tools used to explore various issues in computational organic chemistry. 35 Therefore, the M06-2X method 36 was chosen with the basic set 6-31g(d) to clarify the mechanism of the base-catalyzed cycloaddition reaction between phenylacetylene and azidobenzene. All the geometric parameters of reactants, products, and potential stable points characterized by imaginary frequency were determined by level of theory M06-2X/6-31g(d) according to the proposed mechanism as in Scheme 1. The effect of solvent on the reaction was considered by single-point SCRF (self-consistent reaction field) calculations with the polarizable continuum model (PCM) method37–39 at the M06-2X/6-31g(d) level. The free energies in acetonitrile, DMSO, and water as solvents were obtained by using the frequencies from the gas-phase results. All calculations included in this paper were performed by the program Gaussian09. 40 The Cartesian coordinates for all compounds after geometry optimization using M06-2X/6-31g(d) level can be found in the Supporting Information.
Results and discussion
Huisgen 1,3-dipolar cycloaddition
To compare the reaction of the Huisgen 1,3-dipolar cycloaddition with the base-catalyzed cycloaddition, we studied first the mechanism of the Huisgen reaction in the gas phase and in the presence of solvents based on the mechanism shown in Scheme 2. Figure 1 shows the optimized structures of reactants, products (P1 and P2), and transition states (TS1 and TS2) using M06-2X/6-31g(d)-level calculations.

Mechanism for Huisgen 1,3-dipolar cycloaddition reaction phenylacetylene and azidobenzene.

M06-2X/6-31g(d) optimized structures of reactants, products, and transition states with the values of imaginary frequencies.
The electronic M06-2X/6-31g(d) energy, Hcorr, Gcorr, and the zero-point vibrational energies (ZPE) for reactants, products, and transition states are reported in Table 1, while Table 2 shows thermodynamic and kinetic properties of Huisgen 1,3-dipolar cycloaddition reaction between phenylacetylene and azidobenzene in the gas phase and in the presence of solvents at 298.15 K. Changes of Gibbs free energies along reaction pathway are shown in Figure 2.
Calculated total electronic energy (Eo), Hcorr, Gcorr, and the ZPE at the M06-2X/6-31g(d) level (in Hartree).
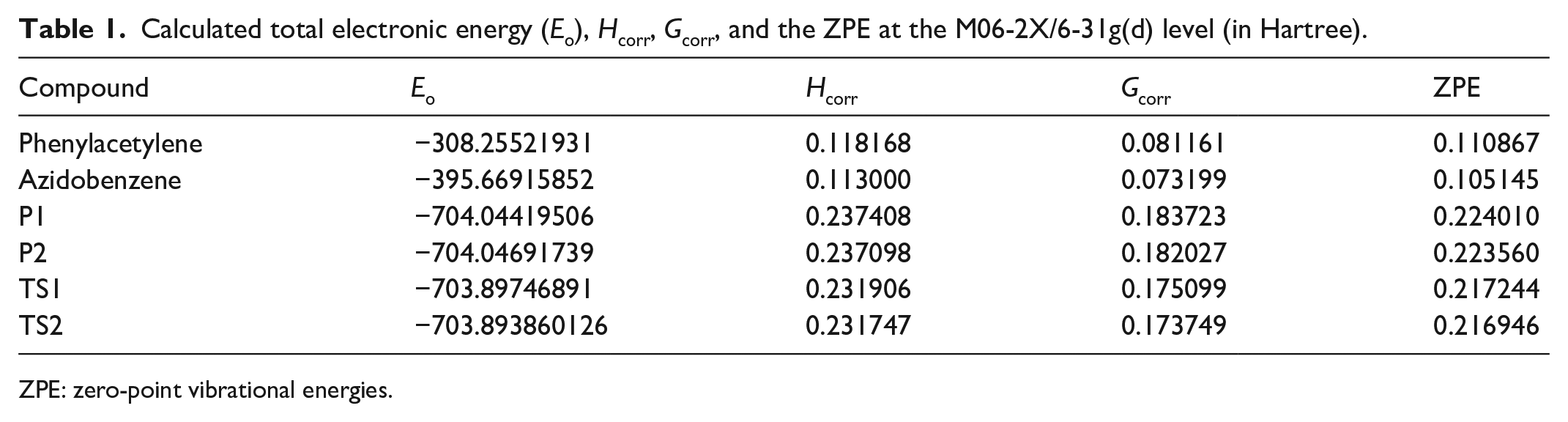
ZPE: zero-point vibrational energies.
Calculated Gibbs free energies, activity energy (Ea) (in kJ mol−1), and the rate constants (in L mol−1 s−1) at the M06-2X/6-31g(d) level in the gas phase and in the presence of solvents.
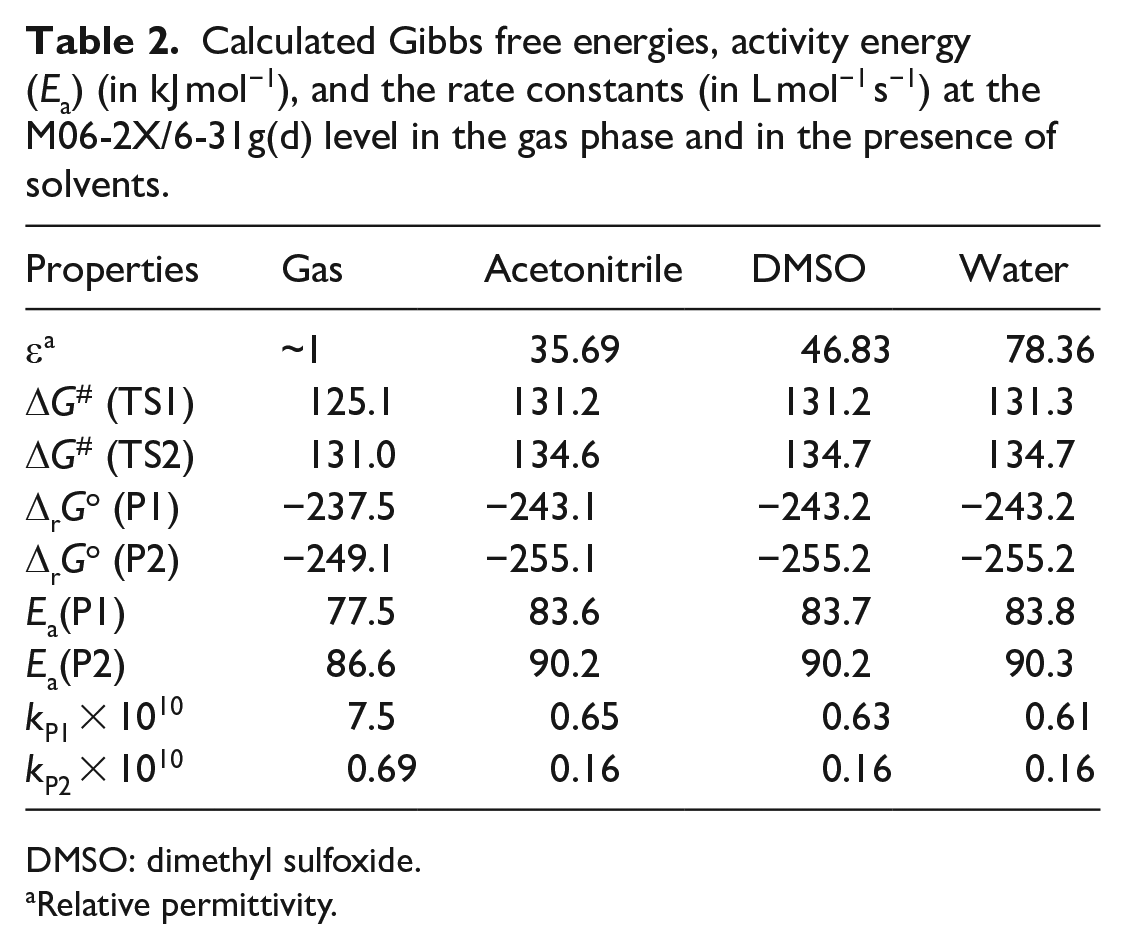
DMSO: dimethyl sulfoxide.
Relative permittivity.
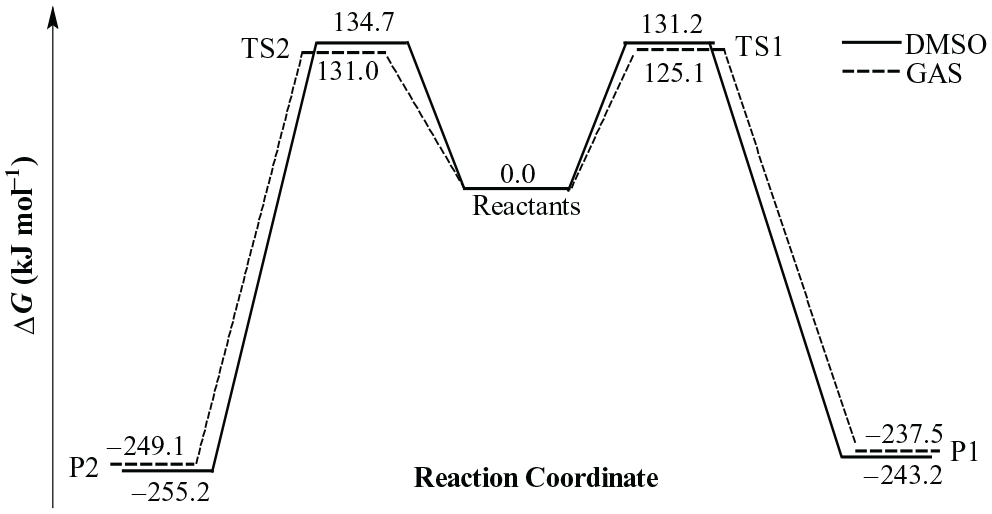
Changes of Gibbs free energies along reaction pathway in the gas phase and in the presence of DMSO as solvent.
The computational results show that the cycloaddition reaction is carried out spontaneously for the products P1 and P2, where the latter is more spontaneous than former, but the former is faster by about 10 times in gas phase and by about four times in the presence of solvents. Moreover, we note from Table 2 that the solvents do not show a significant effect on the kinetic properties of the cycloaddition reaction compared to those in the gas phase. This can be explained by the fact that the charges are not significantly redistributed in the transition states.
Base-catalyzed cycloaddition
The mechanism involving base-catalyzed cycloaddition (Scheme 1) is proposed for a charged system, but the final step relates to the reaction of phenylacetylene with the base. If the base is KOH, this reaction can occur as follows

Based on this reaction, another mechanism could be proposed for an uncharged system as shown in Scheme 3 in order to determine the thermodynamic properties of step 3. Calculated total electronic energy (Eo), Hcorr, Gcorr, and ZPE for reactants, reactive complex (RC and RC−K), intermediate complex (IC and IC−K), and their transition state for charged and uncharged systems using M06-2X/6-31g(d) level are reported in Tables 3 and 4, respectively. Thermodynamic and kinetic properties of base-catalyzed cycloaddition in the gas phase and in the presence of DMSO at 298.15 K are shown in Table 5. The geometries for reactants (Figure 3), RC and IC (Figure 4), RC−K and IC−K (Figure 5) are optimized at the M06-2X/6-31g(d) level. The changes of Gibbs free energies along the reaction pathway are shown in Figure 6 for charged and uncharged systems.

Proposed mechanism of catalyzed process for uncharged system.
Calculated total electronic energy (Eo), Hcorr, Gcorr, and ZPE at the M06-2X/6-31g(d) level (in Hartree) for the charged system.

ZPE: zero-point vibrational energies; RC: reactive complex; IC: intermediate complex.
Calculated total electronic energy (Eo), Hcorr, Gcorr, and ZPE at the M06-2X/6-31g(d) level (in Hartree) for uncharged system.
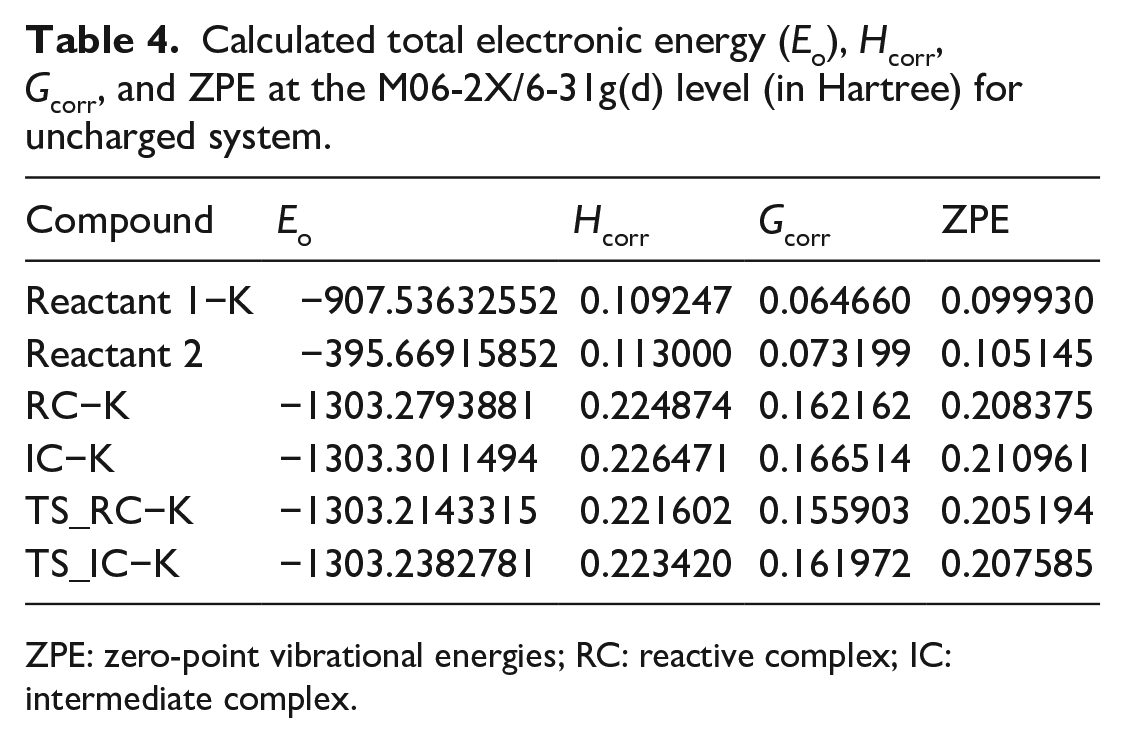
ZPE: zero-point vibrational energies; RC: reactive complex; IC: intermediate complex.
Calculated Gibbs free energies, activity energy (Ea) (in kJ mol−1), and the rate constants (k) at the M06-2X/6-31g(d) level in the gas phase and in the presence of DMSO as solvent. a
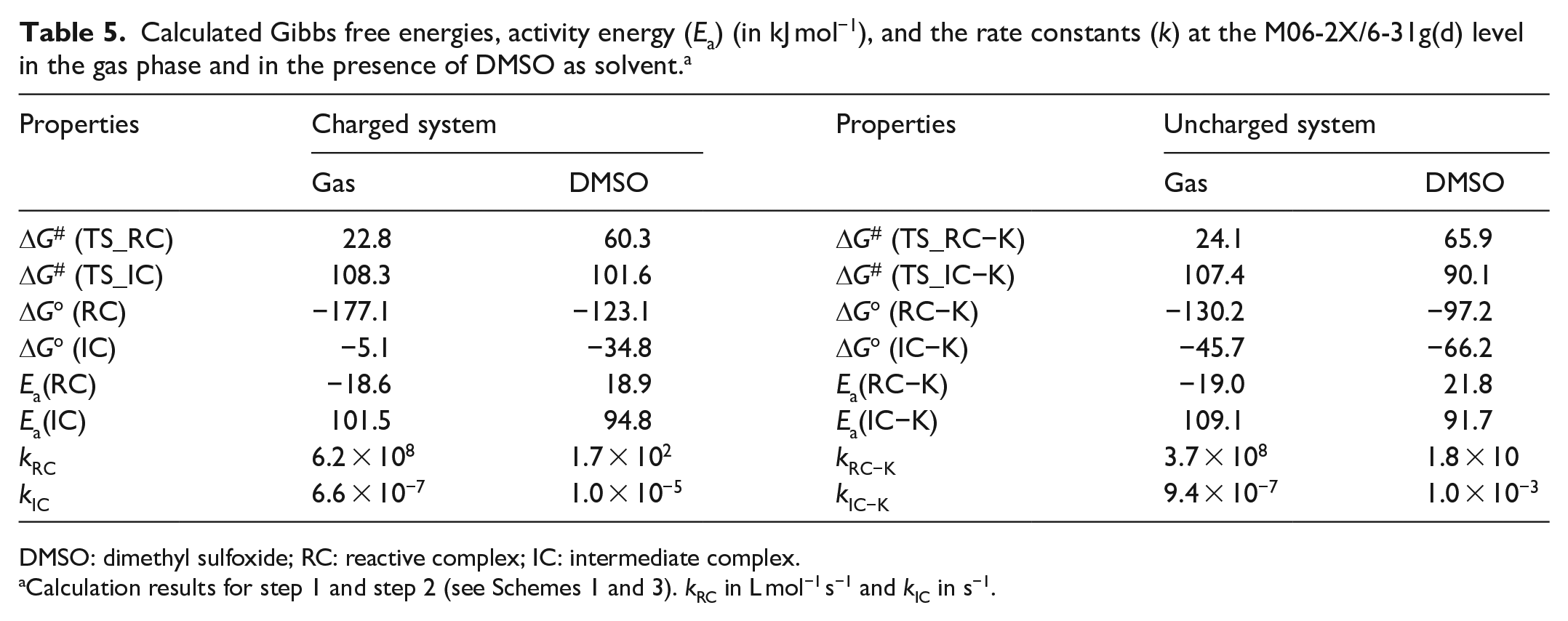
DMSO: dimethyl sulfoxide; RC: reactive complex; IC: intermediate complex.
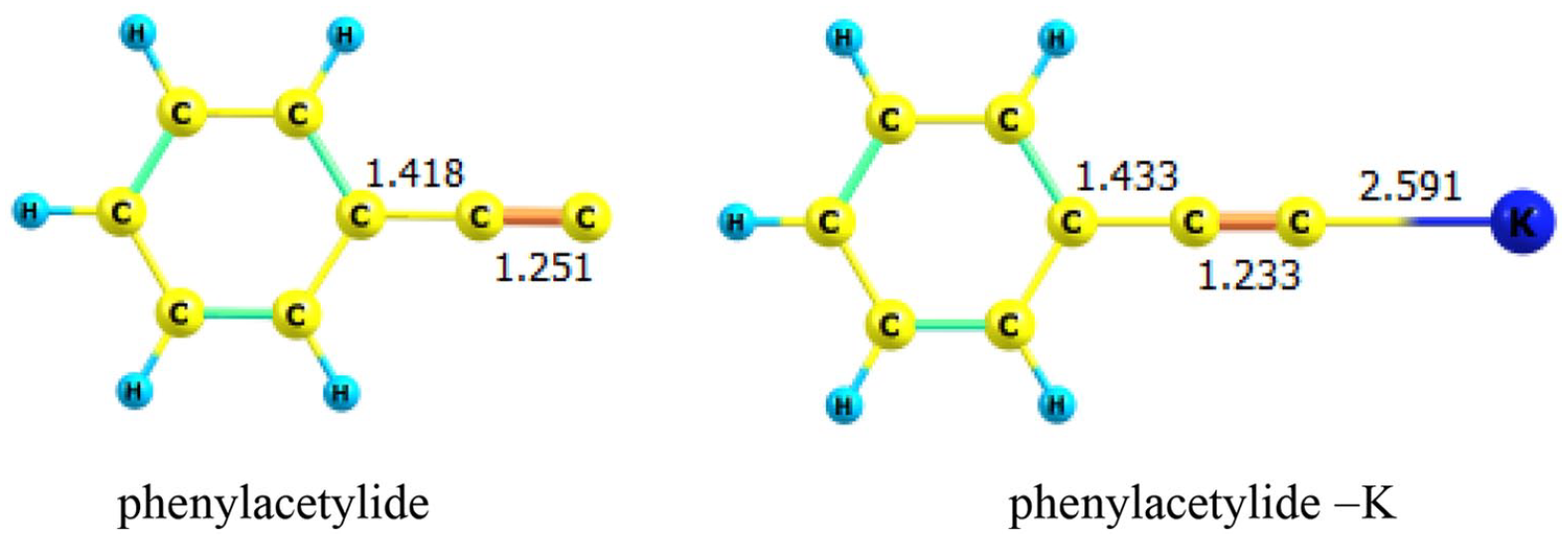
M06-2X/6-31g(d) optimized structures of phenylacetylide and phenylacetylide–K.

M06-2X/6-31g(d) optimized structures of RC, IC, and their transition states with the values of imaginary frequencies.

M06-2X/6-31g(d) optimized structures of RC−K, IC−K, and their transition states with the values of imaginary frequencies.
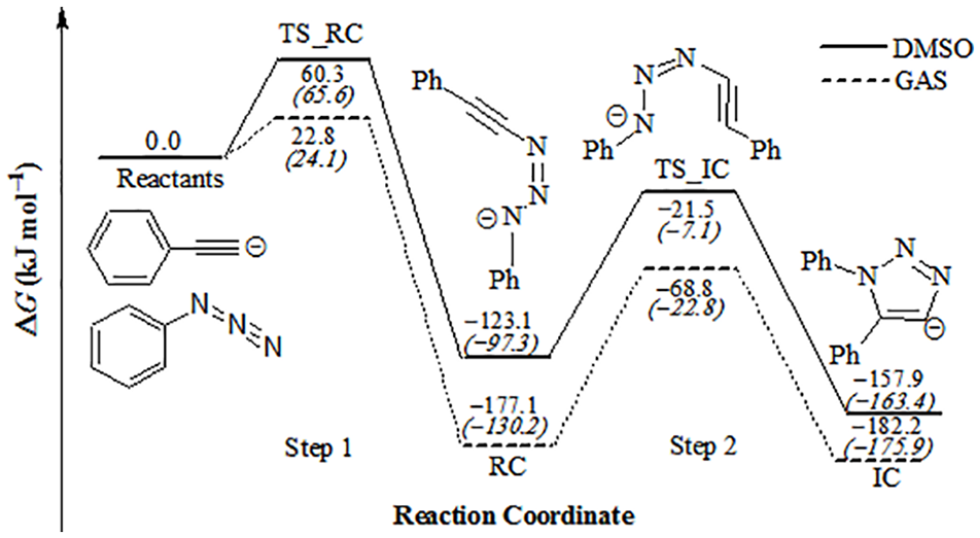
Changes of Gibbs free energies along reaction pathway in the gas phase and in the presence of DMSO as solvent. The numbers in parentheses refer to the uncharged system.
It is noted from the geometries of TS_RC and TS_RC−K that the length of the C−N forming bond for TS_RC−K (2.229 Å) is shorter than that for TS_RC (2.450 Å), but this bond for IC−K (1.375 Å) becomes shorter than that for RC−K (1.393 Å). This indicates that the formation of complex IC−K is faster than complex IC.
From Table 5 and Figure 6, we note that the reactions in step 1 and step 2 are carried out spontaneously in gas phase for the RC and IC products in both charged and uncharged systems, where the latter is more spontaneous than former, but the former is faster than latter. Moreover, the barrier of the rate-determining step (Step 2) is 101.5 and 109.1 kJ mol−1 in the gas phase for charged and uncharged systems, respectively.
The effect of solvent has been investigated for the charged and uncharged systems in DMSO that is most frequently used. It has been observed that the solvent decreases the barrier by 7.5 and 17.4 kJ mol−1 in the presence of DMSO for the charged and uncharged systems, respectively, as compared to that in the gas phase. It should be noted that the calculations in the presence of acetonitrile or water provide almost the same results. Moreover, the calculated barrier of the rate-determining step for uncharged systems is lower than that for charged systems by 3.1 kJ mol−1 in the presence of DMSO.
The rate constant for the uncatalyzed and catalyzed processes of 1,5-disubstituted triazole formation is 7.5 × 10−10 L mol−1 s−1 and 6.6 × 10−7 s−1 in the gas phase (6.3 × 10−11 L mol−1 s−1 and 1.0 × 10−5 s−1 in the presence of DMSO), respectively. This difference in rate constant of the catalyzed and uncatalyzed processes has exhibited significant acceleration of the reaction in the presence of the base catalyst.
Finally, the ΔG3 of step 3 can be calculated according to the following equation for charged and uncharged systems, respectively

Since the mechanism of formation of the final product (product 1) is carried out as sequential reactions, the Gibbs free energy of final product formation equals the sum of the free Gibbs energies ΔrG for the three sttif (in the presence of DMSO; see Table 5)
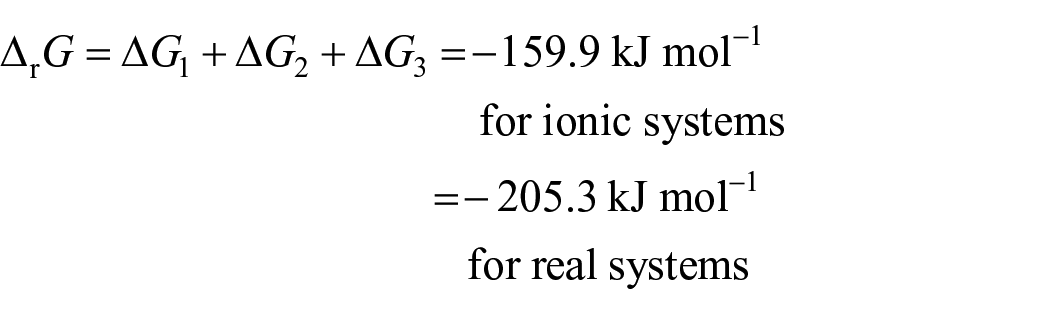
or according to the overall reaction:
It is found that the reaction of step 3 for the uncharged system is more spontaneous than that for the charged system, and the same applies to the total reaction.

Conclusion
In this study, the mechanism for both the Huisgen 1,3-dipolar cycloaddition and the base-catalyzed cycloaddition reactions between phenylacetylene and azidobenzene has been investigated with a quantum mechanical methods, namely at the M06-2X/6-31G(d) computational level. The computational results in the presence of solvents show that the barrier for the uncatalyzed reaction is slightly lower than that for the catalyzed reactions; however, the uncatalyzed reaction is slower as compared to that of uncatalyzed reaction in the presence of solvents. The calculated barrier of the rate-determining step in the base-catalyzed mechanism with the uncharged system is lower than that with the charged system. Finally, the reaction leading to final product formation in the uncharged system is more spontaneous than that in the charged system, and the same applies to the total reaction in the presence of solvents.
Supplemental Material
Supporting_Information – Supplemental material for Quantum mechanical investigations of base-catalyzed cycloaddition reaction between phenylacetylene and azidobenzene
Supplemental material, Supporting_Information for Quantum mechanical investigations of base-catalyzed cycloaddition reaction between phenylacetylene and azidobenzene by Mohammad Abd Al-Hakim Badawi and Sultan T Abu-Orabi in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
