Abstract
A series of novel 4-O-alkyltriazolylphenolic derivatives is first synthesized with good to excellent yields via the click reaction of 3-methoxy-4-O-propargylbenzaldehyde or 3-allyl-4-O-propargylacetophenone and aromatic azide derivatives. Next, the chalcones are prepared via the Claisen-Schmidt method from 4-O-alkylphenylketone derivatives in the presence of the corresponding (hetero)aromatic aldehydes as electrophiles. The structures of the newly synthesized compounds are confirmed from their infrared, nuclear magnetic resonance spectral data, and by elemental analysis. The main advantages of this procedure are the simplicity of the reaction conditions, easily available starting materials, and simple work-up. The antioxidant activity of several of the products is determined using the DPPH (2,2-diphenyl-1-picryl-hydrazyl-hydrate) radical scavenging assay. 4-O-propargylvanillin (IC50 = 14.54 µg/mL) had moderate antioxidant activity.
Keywords
The antioxidant activity of novel 4-O-alkyltriazolylphenylaldehydes/ketones, O-substituted triazolyl-chalcone derivatives, and vitamin C was determined using DPPH (2,2-diphenyl-1-picryl-hydrazyl-hydrate) radical scavenging assay. Some of these synthesized molecules had moderate antioxidant activities meanwhile other compounds displayed low antioxidant capacity.

Introduction
Chalcones (1,3-diphenyl-2-propen-1-ones) and their derivatives are an important class of natural products. They are found in a large number of plants and are considered to be precursors or intermediates in flavonoid and isoflavonoid synthetic pathways. 1 Chalcones are also useful compounds in pharmaceutical chemistry. Notably, chalcones and their derivatives have been found to be associated with various biological activities. 2 Our previous research on this class of molecules indicated that many natural and synthetic chalcones exhibit a wide spectrum of biological activities, such as antimalarial, 3 antimicrobial, 4 antifungal, 5 and antitumour activity.6,7
Recent work has explored the introduction of an alkyltriazolyl group in the chalcone skeleton at the C-4 position to afford lead compounds for the development of new therapeutic agents. Chalcones substituted with imidazoles, 8 triazoles, 9 amines, 10 and stilbene 11 have been reported to be potential antimalarial compounds. They have also been used as the synthetic intermediates of choice for functionalization of the α,β-position during the total synthesis of natural products such as the tropone-chalcones and heterocyclic derivatives which have attracted considerable interest due to their unique structure and properties. 12 The derivatives of 4-O-alkyltriazolylchalcone are known to possess antibacterial 13 and antifungal 14 activities. The interest of researchers in lead molecules containing the 1,2,3-triazole group as chemotherapeutic agents for various types of diseases is growing because they are known to exhibit a wide range of biological activity, such as antibiotic, 15 antifungal, 16 and anticancer properties. 17 Some 1,2,3-triazoles are used as deoxyribose nucleic acid (DNA) cleaving agents 18 and potassium channel activators. 19 Clinically used drugs containing a 1,2,3-triazole moiety include the β-lactam antibiotics tazobactam and cefatrizine, as well as the calcium channel blocker carboxyamidotriazole (Figure 1). Also, they have been widely used as synthetic intermediates and in industrial applications, for example, as dyes, anticorrosive agents, photostabilizers, photographic materials, and agrochemicals. 20 Therefore, the biological importance of the 1,2,3-triazole system as a link between two active molecules is useful to improve their biological properties.
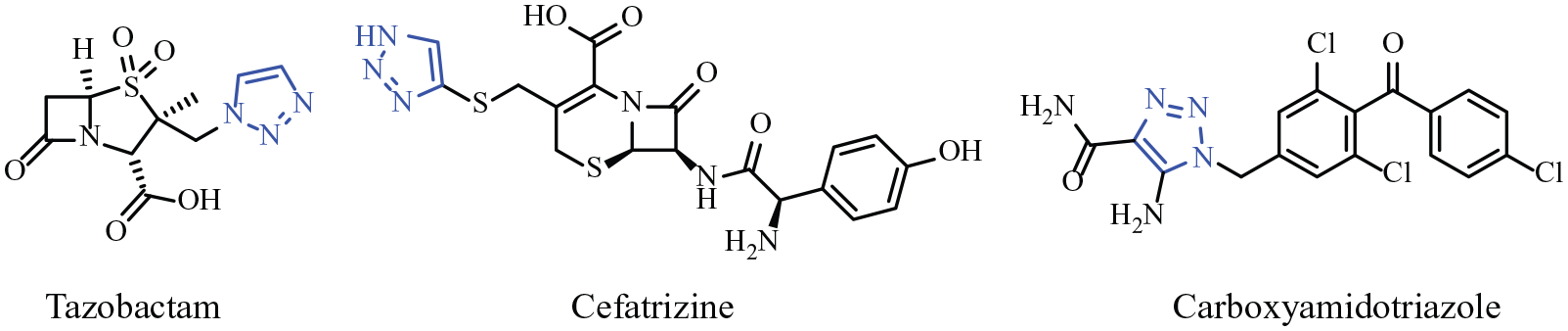
A few biologically active compounds containing 1,2,3-triazole moiety.
Prompted by these observations on the pharmacological significance of triazole derivatives, and in order to discover new intermediates for the synthesis of antioxidant agents, this research describes the synthesis of new O-alkyltriazolylphenylaldehyde/ketone and chalcone derivatives. The preparation of new triazolated compounds as synthetic intermediates, is currently under investigation in our laboratory for the investigation of their potential pharmacological activities.
Results and discussion
Chemistry
The goal of our synthesis was to introduce a propargyl and thus triazole group to 3-methoxy-4-hydroxybenzaldehyde or 3-allyl-4-hydroxyacetophenone derivatives in order to prepare the desired O-substituted chalcone moieties and to investigate their antioxidant profiles. O-Substituted phenolic and chalcone derivatives are an important class of compounds which are known to possess good biological activities.
The synthetic routes to the desired compounds are outlined in Schemes 1–3. Initially, 4-O-substituted phenolic derivatives

Synthesis of 4-O-substituted phenolic derivatives

Synthesis of O-substituted 4-O-methyltriazolylphenylaldehyde

Synthesis of 4-O-methyltriazolyl-chalcone derivatives
Azide derivatives
The intermediates, 4-O-methyltriazolylphenylaldehyde
Biologically, pyrroles tend to construct the key structure of porphyrin rings, which act as an active moiety in chlorophyll, vitamin B12, or bile pigments. 29 Pyrrole and its derivatives are widely used as intermediates in synthesis of pharmaceuticals, agrochemicals, dyes, photographic chemicals, perfumes, and other organic compounds. They are also the versatile molecules, which are active against the various diseases as antiviral, anticonvulsant, and antiinflammatory. 29 Otherwise, pyrrole and its derivatives exhibit wide range of biological activities such as anticancer and antioxidant activity. 30 In addition, the pyrrole-containing heterocyclic compounds have attracted attention particularly as antimicrobial agents. 29 These studies confirmed that pyrrole ring is a good pharmacophore group for the design of bioactive molecules.29,30
Thus, under these biological interests of pyrrole derivatives, our group recently developed a synthetic methodology of O-alkyl substituted heterocyclic-chalcones and evaluated their antioxidant profile.
Compounds
Biological studies
The antioxidant activity of compounds
Antioxidant activity of the prepared compounds and vitamin C as determined by the DPPH radical scavenging assay.

Values are mean ± standard deviation (SD) of six independent experiments; values with the same letter are not significantly different (p < 0.05; ANOVA).
Chemicals are considered to have high or significant anti-oxidant capacity when their IC50 value is below 10 μg/mL, moderate antioxidant capacity for IC50 values between 10 and 20 μg/mL and low antioxidant capacity if the IC50 value is >20 μg/mL.
36
Thus, compound
Conclusion
In conclusion, we have developed an efficient method for the synthesis of new O-substituted triazolylphenylaldehyde/ketone compounds as interesting intermediates for the preparation of chalcone derivatives in order to explore their antioxidant profiles. Also, this synthesis, which will be the key subject of our further research, offers significant improvements over existing procedures and thus helps to facilitate the synthesis of a variety of O-alkyltriazole compounds with potentially high synthetic and biological utilities. Further studies on O-substituted phenolic and chalcone derivatives as model intermediates for the synthesis of antioxidant lead compounds are currently under investigation in our laboratory.
Experimental section
General
Melting points were determined using open-glass capillaries on a Gallenkamp 8093/08/224 melting point apparatus which are uncorrected. IR spectra were determined with a Perkin Elmer FT-IR spectrophotometer. Nuclear magnetic resonance (NMR) spectra were recorded using a Bruker or Varian spectrometers (400 MHz for 1H NMR and 100 MHz for 13C NMR). The chemical shifts are reported in parts per million (ppm) downfield from tetramethylsilane (TMS), which was used as the internal standard. Elemental analyses were recorded on a LECO 932 CHNS elemental analyzer. Thin-layer chromatography (TLC) was carried out using Merck silica gel 60 F-254 plates (layer thickness = 0.25 mm), and all solvents were distilled prior to use.
All the azide derivates
General procedure for the synthesis of 4-allyloxy-3-methoxybenzaldehyde (1a )
To vanillin (
General procedure for the synthesis of 3-methoxy-4-O-propargylbenzaldehyde (1b ) and 3-allyl-4-O-propargylacetophenone (1c )
To a stirring mixture of vanillin (
3-Methoxy-4-prop-2-ynyloxy-benzaldehyde (1b ):
White solid; m.p. 85–87 °C (lit.9 86 °C); yield 92%,;Rf = 0.40; (hexane-EtOAc 4:1 v/v); 1H NMR (400 MHz, CDCl3): δ 9.87 (s, 1H), 7.47 (br d, J = 8.2 Hz, 1H), 7.44 (br s, 1H), 7.15 (d, J = 8.2 Hz, 1H), 4.86 (d, J = 1.8 Hz, 2H), 3.94 (s, 3H), 2.58 (t, J = 1.8 Hz, 1H); 13C NMR (100 MHz, CDCl3): δ 191.0, 152.3, 150.2, 131.1, 126.3, 112.8, 109.7, 77.7, 76.9, 56.8, 56.2.
1-(3-allyl-4-propargyloxyphenyl)ethanone (1c ):
White solid; m.p. 47–48 °C; yield 84%; Rf = 0.19; (hexane-EtOAc 9:1 v/v); IR (CHCl3): 3962, 3502, 3293, 3078, 2920, 2579, 2122, 2039, 1846, 1682, 1639, 1600,1496, 1454, 1424, 1360, 1318, 1267, 1190, 1132,1077, 1023, 979, 924, 814, 682, 657, 595, 579 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.85 (dd, J = 8.6, 2.0 Hz, 1H), 7.80 (d, J = 2.0 Hz, 1H), 7.00 (d, J = 8.6 Hz, 1H), 6.06–5.86 (m, 1H), 5.10–5.06 (m, 2H), 4.78 (d, J = 2.1 Hz, 2H), 3.42 (br d, J = 6.6 Hz, 2H), 2.55–2.54 (m, 4H); 13C NMR (100 MHz, CDCl3): δ 197.0, 159.1, 135.9, 130.8, 130.4, 129.4, 128.6, 116.2, 111.1, 78.0, 76.0, 56.0, 34.2, 26.4; Anal. Calcd for C14H14O2: C, 78.48; H, 6.59; found: C, 78.52; H, 6.62%.
3-Azidopyridine (4 )
(2.00 g, 21.2 mmol) was dissolved in a solution of conc. H2SO4 (2.4 mL) and H2O (14 mL). The resulting solution was cooled to 0 °C, and a solution of NaNO2 (1.76 g, 25.6 mmol) in H2O (10 mL) was added dropwise with stirring. The solution was stirred at this temperature for a further 15 min, whereupon a solution of NaN3 (2.34 g, 36.0 mmol) in H2O (8 mL) was added with vigorous stirring. The mixture was stirred for 1 h at 0 °C, then overnight at room temperature. The reaction mixture was basified with freshly saturated Na2CO3 solution and extracted with CH2Cl2 (3 × 30 mL). The combined extracts were washed with H2O (50 mL), dried over MgSO4, and the solvent removed in vacuo to provide 3-azidopyridine (
General procedure for the synthesis of the 4-O-methyltriazolylphenylaldehyde/ketone derivatives 2a , and 3a–b
A mixture of 3-methoxy-4-O-propargylbenzaldehyde (
3-Methoxy-4-[{1-(naphthalen-1-yl)-1H-1,2,3-triazol-4-yl}methoxy]benzaldehyde (2a ):
Colorless solid; m.p. 127–128 °C; yield 88%; Rf = 0.22; (hexane-EtOAc 7:3 v/v); IR (CHCl3): 3143, 3060, 2939, 2834, 1681, 1587, 1508, 1465, 1425, 1388, 1339, 1267, 1235, 1159, 1136, 1043, 1019, 999, 951, 864, 802, 773, 731, 701, 643, 591, 571 cm−1; 1H NMR (400 MHz, CDCl3): δ 9.86 (s, 1H), 8.07 (s, 1H), 8.03–7.86 (m, 2H), 7.77–7.38 (m, 7H), 7.33 (s, 1H), 5.53 (s, 2H), 3.92 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 190.9, 153.0, 150.0, 143.2, 134.1, 133.5, 130.9, 130.8, 128.6, 128.5, 128.0, 127.1, 126.7, 125.9, 125.0, 123.6, 122.2, 112.8, 109.4, 63.0, 56.0; Anal. Calcd for C21H17N3O3: C, 70.18; H, 4.77; N, 11.69; found: C, 70.51; H, 4.67; N, 11.42%.
1-[3-allyl-4-{(1-Benzyl-1H-1,2,3-triazol-4-yl)methoxy}phenyl]ethanone (3a ):
White solid; m.p. 100–102 °C; yield 86%; Rf = 0.41; (hexane-EtOAc 2:3 v/v); IR (CHCl3): 3137, 3064, 2913, 2846, 1673, 1599, 1498, 1425, 1358, 1263, 1131, 1002, 816, 736 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.83 (d, J = 8.6 Hz, 1H), 7.78 (s, 1H), 7.50 (s, 1H), 7.39–7.37 (m, 3H), 7.32–7.24 (m, 2H), 7.02 (d, J = 8.6 Hz, 1H), 5.96–5.86 (m, 1H), 5.54 (s, 2H), 5.27 (s, 2H), 5.00 (br s, 1H), 4.97 (br d, J = 8.4 Hz, 1H), 3.37 (d, J = 6.5 Hz, 2H), 2.54 (s, 3H). 13 C NMR (100 MHz, CDCl3): δ 197.0, 159.8, 144.2, 136.1, 134.4, 130.6, 130.3, 129.2, 129.0 (2C), 128.9 (2C), 128.8, 128.1, 122.6, 116.1, 110.9, 62.4, 54.3, 34.4, 26.4; Anal. Calcd for C21H21N3O2: C, 72.60; H, 6.09; N, 12.10; found: C, 72.65; H, 5.79; N, 11.95%.
1-[3-allyl-4-{(1-(pyridin-3-yl)-1H-1,2,3-triazol-4-yl)methoxy}phenyl]ethanone (3b ):
White solid; m.p. 97–98 °C; yield 71%; Rf = 0.22; (hexane-EtOAc 2:3 v/v); IR (CHCl3): 3417, 3137, 3076, 3008, 1673, 1598, 1498, 1359, 1263, 1248, 1130, 1018, 806 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.99 (br s, 1H), 8.68 (d, J = 4.6 Hz, 1H), 8.13 (br s, 1H), 8.11–8.10 (m, 1H), 7.82 (dd, J = 8.6, 2.1 Hz, 1H), 7.76 (d, J = 2.1 Hz, 1H), 7.48 (dd, J = 8.2, 4.6 Hz, 1H), 7.04 (d, J = 8.6 Hz, 1H), 5.94 (ddt, J = 16.8, 10.7, 6.6 Hz, 1H), 5.36 (s, 2H), 5.06–5.03 (m, 1H), 5.01 (dd, J = 10.7, 1.5 Hz, 1H), 3.39 (d, J = 6.6 Hz, 2H), 2.51 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 197.2, 159.8, 150.4, 145.3, 141.8, 136.2, 133.7, 130.9, 130.7, 129.3, 129.2, 128.4, 124.5, 121.1, 116.4, 111.1, 62.3, 34.5, 26.6; Anal. Calcd for C19H18N4O2: C, 68.25; H, 5.43; N, 16.76; found: C, 68.58; H, 5.44; N, 16.33%.
General procedure for the synthesis of 4-O-methyltriazolyl-chalcone derivatives 4a–e
A solution of KOH (0.5 g, 7.24 mmol) in H2O (5 mL) was slowly added dropwise to a mixture of substituted aryl/heteroaryl-carbaldehyde (2 mmol) and 3-allyl-4-O-methyltriazolyl-acetophenone (2 mmol) dissolved in EtOH (10–20 mL). The reaction mixture was stirred at 40–42 °C for 39–80 h, under a nitrogen atmosphere and monitored by TLC. The precipitate formed was filtered, dried and purified by silica gel column chromatography using a mixture hexane-EtOAc to provide
(E)-1-(3-allyl-4-((1-benzyl-1H-1,2,3-triazol-4-yl)methoxy)phenyl)-3-(4-(allyloxy)-3-methoxyphenyl)prop-2-en-1-one (4a ):
Yellow solid; m.p. 115–117 °C; yield 90%; Rf = 0.58; (hexane-EtOAc 3:2 v/v); IR (CHCl3): 3137, 3070, 2937, 1653, 1598, 1509, 1463, 1423, 1310, 1255, 1165, 1135, 1032, 997, 923, 804, 725, 554 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.03 (dd, J = 8.6, 2.2 Hz, 1H), 7.97 (d, J = 2.2 Hz, 1H), 7.85 (d, J = 15.6 Hz, 1H), 7.64 (s, 1H), 7.54–7.50 (m, 1H), 7.50–7.45 (m, 3H), 7.45–7.34 (m, 2H), 7.32–7.23 (m, 2H), 7.17 (d, J = 8.6 Hz, 1H), 7.01 (d, J = 8.3 Hz, 1H), 6.20 (ddd, J = 20.4, 10.6, 5.4 Hz, 1H), 6.05 (ddt, J = 18.2, 9.5, 6.6 Hz, 1H), 5.65 (s, 2H), 5.54 (dd, J = 20.4, 1.1 Hz, 1H), 5.43 (dd, J = 10.6, 1.1 Hz, 1H), 5.40 (s, 2H), 5.12 (br d, J = 0.9 Hz, 1H), 5.10–5.08 (m, 1H), 4.78 (d, J = 5.4 Hz, 2H), 4.06 (s, 3H), 3.51 (d, J = 6.6 Hz, 2H); 13C NMR (100 MHz, CDCl3): δ 188.9, 159.6, 150.3, 149.6, 144.2, 144.1, 136.2, 134.4, 132.8, 131.6, 130.6, 129.2 (2C), 129.1, 128.9, 128.8, 128.3, 128.1 (2C), 122.7, 122.6, 120.0, 118.4, 116.0, 113.0, 111.1, 110.7, 69.8, 62.4, 56.1, 54.3, 34.4; Anal. Calcd for C32H31N3O4: C, 73.68; H, 5.99; N, 8.06; found: C, 73.44; H, 5.93; N, 7.74%.
(E)-1-(3-allyl-4-((1-benzyl-1H-1,2,3-triazol-4-yl)methoxy)phenyl)-3-(1-(3,5-dichlorophenyl)-1H-pyrrol-2-yl)prop-2-en-1-one (4b ):
Pale brown solid; m.p. 128–130 °C; yield 88%; Rf = 0.26; (hexane-EtOAc 7:3 v/v); IR (CHCl3): 3476, 3069, 1650, 1600, 1589, 1573, 1497, 1464, 1433, 1354, 1320, 1272, 1252, 1136, 1049, 1003, 917, 858, 807, 725, 681, 575 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.83 (dd, J = 8.6, 2.2 Hz, 1H), 7.76 (d, J = 2.2 Hz, 1H), 7.52 (d, J = 15.3 Hz, 1H), 7.50 (br s, 1H), 7.43 (br t, J = 1.8 Hz, 1H), 7.38–7.37 (m, 1H), 7.36 (d, J = 1.8 Hz, 2H), 7.31–7.24 (m, 4H), 7.21 (d, J = 15.3 Hz, 1H), 7.03 (d, J = 8.6 Hz, 1H), 6.98–6.92 (m, 2H), 6.41–6.38 (m, 1H), 5.91 (ddt, J = 17.5, 11.0, 6.6 Hz, 1H), 5.53 (s, 2H), 5.27 (s, 2H), 4.99 (br s, 1H), 4.98–4.94 (m, 1H), 3.37 (d, J = 6.6 Hz, 2H); 13C NMR (100 MHz, CDCl3): δ 188.4, 159.8, 144.5, 141.0, 136.4, 136.0 (2C), 134.7, 131.7, 130.9, 130.7, 129.4 (2C), 129.3, 129.1, 128.9 (2C), 128.6, 128.3, 127.2, 125.3, 122.8, 118.9 (2C), 116.2 (2C), 113.9, 111.7, 111.3, 62.6, 54.5, 34.7; Anal. Calcd for C32H26Cl2N4O2: C, 67.49; H, 4.60; N, 9.84; found: C, 67.35; H, 4.63; N, 9.61%.
(E)-1-(3-allyl-4-((1-benzyl-1H-1,2,3-triazol-4-yl)methoxy)phenyl)-3-(1-(4-(trifluoromethyl)phenyl)-1H-pyrrol-2-yl)prop-2-en-1-one (4c ):
Pale yellow solid; m.p. 140–142 °C; yield 80%; Rf = 0.17; (hexane-EtOAc 7:3 v/v); IR (CHCl3): 3098, 3068, 2936, 1650, 1600, 1536, 1498, 1454, 1357, 1326, 1299, 1249, 1170, 1129, 1067, 1036, 1003, 918, 888, 850, 818, 728, 629, 604, 460 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.82 (dd, J = 8.6, 2.2 Hz, 1H), 7.78 (d, J = 2.2 Hz, 1H), 7.76 (d, J = 3.6 Hz, 2H), 7.58 (d, J = 15.4 Hz, 1H), 7.50 (s, 1H), 7.46 (d, J = 8.4 Hz, 2H), 7.39–7.35 (m, 3H), 7.28 (br d, J = 2.5 Hz, 1H), 7.24 (d, J = 15.4 Hz, 1H), 7.03 (d, J = 3.6 Hz, 2H), 7.02 (d, J = 8.6 Hz, 1H), 6.98 (dd, J = 3.7, 1.4 Hz, 1H), 6.43 (dd, J = 3.7, 3.1 Hz, 1H), 5.96–5.85 (m, 1H), 5.54 (s, 2H), 5.27 (s, 2H), 4.99 (br s, 1H), 4.98–4.94 (m, 1H), 3.37 (d, J = 6.5 Hz, 2H); 13C NMR (100 MHz, CDCl3): δ 188.0, 159.6, 144.2, 141.9, 136.1, 134.4, 131.8, 131.5, 130.7, 130.4 (2C), 129.2 (2C), 129.1, 128.9, 128.7, 128.1 (2C), 126.9, 126.8, 126.6 (2C), 126.5 (q, J = 272.7 Hz, CF3), 125.1, 122.5, 118.3, 116.0, 113.5, 111.4, 111.1, 62.4, 54.3, 34.4; 19F NMR (376 MHz, CDCl3): δ −62.88 (s, 3F, CF3); Anal. Calcd for C33H27F3N4O2: C, 69.71; H, 4.79; N, 9.85; found: C, 69.32; H, 4.69; N, 9.51%.
(E)-1-(3-allyl-4-((1-benzyl-1H-1,2,3-triazol-4-yl)methoxy)phenyl)-3-(1-(p-tolyl)-1H-pyrrol-2-yl)prop-2-en-1-one (4d ):
Pale yellow solid; m.p. 100–101 °C; yield 68%; Rf = 0.57; (hexane-EtOAc 13:7 v/v); IR (CHCl3): 3132, 3065, 2975, 2923, 2874, 1648, 1600, 1586, 1516, 1498, 1455, 1359, 1326, 1309, 1248, 1136, 1038, 1003, 917, 824, 728, 629, 606, 570, 465 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.80 (dd, J = 8.5, 1.9 Hz, 1H), 7.74 (d, J = 1.9 Hz, 1H), 7.59 (d, J = 15.4 Hz, 1H), 7.49 (br s, 1H), 7.38–7.35 (dd, J = 5.5, 4.1 Hz, 3H), 7.29–7.25 (br t, J = 6.3 Hz, 4H), 7.20 (d, J = 8.2 Hz, 2H), 7.15 (d, J = 15.4 Hz, 1H), 7.00 (d, J = 8.5 Hz, 2H), 6.93 (br d, J = 3.2 Hz, 1H), 6.38–6.35 (m, 1H), 5.96–5.84 (m, 1H), 5.53 (s, 2H), 5.26 (s, 2H), 4.99 (br s, 1H), 4.97–4.94 (m, 1H), 3.36 (d, J = 6.5 Hz, 2H), 2.43 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 188.3, 159.4, 144.3, 138.1, 136.5, 136.2, 134.4, 132.7, 131.8, 130.8, 130.4, 130.0 (2C), 129.2 (2C), 128.9, 128.8, 128.6, 128.1 (2C), 127.5, 126.3 (2C), 122.5, 117.4, 115.9, 112.9, 111.0, 110.5, 62.4, 54.3, 34.4, 21.1; Anal. Calcd for C33H30N4O2: C, 77.02; H, 5.88; N, 10.89; found: C, 77.41; H, 5.78; N, 10.82%.
(E)-1-(3-allyl-4-((1-(pyridin-3-yl)-1H-1,2,3-triazol-4-yl)methoxy)phenyl)-3-(1-(3,5-dichlorophenyl)-1H-pyrrol-2-yl)prop-2-en-1-one (4e ):
Yellow solid; m.p. 160–162 °C; yield 90%; Rf = 0.33; (hexane-EtOAc 3:2 v/v); IR (CHCl3): 3126, 1586, 1460, 1401, 1351, 1250, 1124, 1046, 806 cm−1; 1H NMR (400 MHz, CDCl3): δ 9.01 (d, J = 2.4 Hz, 1H), 8.76–8.69 (m, 1H), 8.14 (ddd, J = 8.3, 2.4, 1.5 Hz, 1H), 8.10 (br s, 1H), 7.86 (dd, J = 8.6, 2.1 Hz, 1H), 7.79 (d, J = 2.1 Hz, 1H), 7.52 (d, J = 15.4 Hz, 1H), 7.50 (br t, J = 3.9 Hz, 2H), 7.43 (br t, J = 1.8 Hz, 1H), 7.25 (d, J = 1.8 Hz, 1H), 7.20 (d, J = 15.4 Hz, 1H), 7.08 (d, J = 8.6 Hz, 1H), 6.98–6.92 (m, 2H), 6.41–6.37 (m, 1H), 5.99 (ddt, J = 16.7, 10.3, 6.4 Hz, 1H), 5.40 (br s, 2H), 5.09–5.05 (m, 1H), 5.03 (br d, J = 1.6 Hz, 1H), 3.45 (d, J = 6.4 Hz, 2H); 13C NMR (100 MHz, CDCl3): δ 188.3, 159.6, 150.4, 145.4, 141.8, 140.9, 136.3, 136.0 (2C), 133.8, 131.9, 131.8, 130.9, 130.8, 129.3, 129.0, 128.6, 128.4, 127.2, 125.3, 124.5, 121.0, 118.8 (2C), 116.4, 113.9, 111.7, 111.2, 62.4, 34.7; Anal. Calcd for C30H23Cl2N5O2: C, 64.76; H, 4.17; N, 12.59; found: C, 63.90; H, 4.37; N, 12.12%.
Biological studies
Evaluation of the antioxidant activity: DPPH radical scavenging assay
The free radical scavenging activity of the compounds was evaluated as described by Mensor et al. 37 Briefly, the test samples were dissolved in pure dimethylsulfoxide (DMSO, Sigma-Aldrich, St Quentin Fallavier, France) and mixed with a DPPH (Sigma) solution (0.3 mM) in EtOH. Various concentrations (1, 5, 10, 50, 100, 250, 500, and 1000 µg/mL) of each sample were tested. After 30 min at room temperature, the absorbance was measured at 517 nm and converted into percentage of antioxidant activity. Ascorbic acid (vitamin C) was used as a standard control. Each assay was repeated thrice in duplicate and the results recorded as the mean of the six experiments (Table 1). The inhibition ratio (%) was calculated as follows: % inhibition = [(Absorbance of control − Absorbance of test sample)/Absorbance of control] × 100. The IC50 value is the concentration of a sample required to scavenge 50% of the DPPH free radical and was calculated from a calibration curve by linear regression. 38 One-way analysis of variance (ANOVA) at the 95% confidence level was used for statistical analysis.
Supplemental Material
Supporting_NMR_data – Supplemental material for Synthesis and structural characterization of novel O-substituted phenolic and chalcone derivatives with antioxidant activity
Supplemental material, Supporting_NMR_data for Synthesis and structural characterization of novel O-substituted phenolic and chalcone derivatives with antioxidant activity by Bathélémy Ngameni, Musa Erdoğan, Victor Kuete, Erdin Dalkılıç, Bonaventure T Ngadjui and Arif Daştan in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B.N. and A.D. are grateful for the financial support from TÜBİTAK-BİDEB 2221-Funding of Visiting Scientists on Sabbatical Leave Fellowship Program to the Department of Chemistry, Ataturk University, Erzurum, Turkey. The authors also thank the Department of Chemistry, Ataturk University, for the important support with the experimental equipment.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
