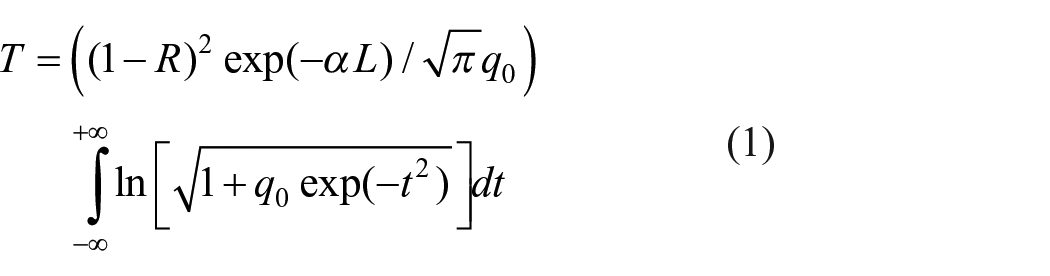Abstract
Mixed ligand complexes of cobalt and palladium containing triphenylphosphine and a hydrazone derived from furfural and hydrazine hydrate have been designed, synthesized, evaluated, and characterized from their spectral properties, elemental analysis, and magnetic susceptibility measurements. The spectral techniques suggest that the complexes exhibit square planar geometry. The monomeric properties of the complexes are evaluated from their magnetic susceptibility values. The complexes were subjected to z-scan analysis for third-order non-linear optical measurements. Non-linear transmission measurements performed using laser pulses at 532 nm in nanosecond indicate that the complexes may show good potential as optical limiters.

Introduction
The expansion of innovative and simple materials that display non-linear optical (NLO) activities is of prime importance in laser technology. Materials derived from specific molecules that have considerable potential for fabrication of a device and its commercialization are of significant interest in industrial and academic research laboratories. 1 The investigation of metal–organic compounds as NLO materials is making its way toward many applications including satellite communications. Novel NLO materials are crucial to the improvement of optoelectronic technologies and devices used in communications, ultra-fast electro-optical information processing, high-density data storage, light variation, switching, and so on. Since the mid-1980s, these materials have continued to be at the forefront of research activities in the field of laser optics. Still, the challenge remains for scientists to design and prepare new non-linear optically active materials which are required for practical NLO applications in the form of optoelectronic devices.2,3
Many organic and inorganic materials have been comprehensively used for NLO applications.4–9 For a NLO material used for optical devices, optimal dipole moments of an active chromophore and matrices should be present.10,11 Also, the other criteria of the molecule being non-symmetric should be satisfied. A derivative based on ferrocene exhibiting NLO properties was reported for the first time that belonged to the class of metal–organic compounds. 12 In organic compounds with a metal, increased optical activity would be observed. This is due to two reasons. First, the high level of the p conjugated charge transfer and second, the growth in the extent of conjugation that will have a pivotal role on the delocalization of p-electrons in the presence of excitation light. Hence, as a result, the χ 3 and β values are enhanced with the different types of bonds in the molecule. 13
In recent years, organometallic and coordination complexes have been widely studied for their NLO properties. The combined properties of flexibility and thermal stability of organic materials and the mechanical strength of inorganics make these compounds potentially show NLO activities. Metal complexes (organometallic and coordination), in particular, signify themselves as a trending class of materials in this area of research. Due to the larger variety of structures and their stability under normal environmental conditions, metal–organic compounds serve as better NLO materials compared to organic molecules. Also, metal complexes have a range of electronic properties because of the electron-deficient central metal atom surrounded by electron-donating organic moieties. 14 In 1987, a report by Green et al. described the second-harmonic generation (SHG) efficiency on a derived ferrocene complex that proved it to be an excellent type. 9 Subsequently, the utilization of metal complexes as NLO materials has attracted the attention of many researchers. Consequently, a systematic exploration has been performed on various new metal complexes in this field. Initial and more topical literature on the studies of metal complexes and their NLO activities indicate the scope of research in this field. Noticeably, the NLO properties of several organometallic compounds have been given substantial attention. 15
The following section of the paper reports the design, synthesis, and NLO measurements of metal M [M = Co(II) and Pd(II)] complexes of a mixed ligand system containing triphenylphosphine and a hydrazone derived from furfural and hydrazine. The synthesis of the ligand (

Synthesis of the ligand.

Synthesis of the complexes.
Results and discussion
Electronic spectral studies
The UV-Vis spectra of the complexes in DMF are depicted in Figure 1, exhibiting bands in the region between 250 and 500 nm. The electronic transitions due to the intraligand interactions appear between 260 and 350 nm, being more toward the visible region. A relatively low intensity band is seen in the range 380–495 nm due to the forbidden d → d transition. Other cobalt and palladium complexes exhibiting similar electronic spectral performance have been described in the literature.16–18

UV spectra of complexes
FTIR spectral studies
The Fourier transform infrared spectrum of the ligand displays an intense sharp peak around 1610–1620 cm−1, assigned to the ν(C=N) vibration. As a result of complex formation as the nitrogen atom shares its pair of the electrons with the electron-deficient metal atom, this peak shifts to a lower wavenumber.16,17,19,20 The band in the region 1315–1330 cm−1, assigned to ν(C−O) in the free ligand, is shifted to a higher wavenumber in the complexes suggesting the coordination of oxygen to the metal ion.8,16,17 The 550- and 470-cm−1 bands in the complexes are assigned to ν(M−O) and ν(M−N) frequencies, respectively.16,17 The FTIR spectra are provided in the Supporting Information (Supplemental Figure S1).
NMR spectral studies
The 1 H NMR spectra of the complexes exhibit multiplets around 6.9–7.9 ppm which are assigned to the protons of the phenyl groups present in triphenylphosphine (Supplemental Figures S2 and S5). A sharp peak at around 8.5 ppm in the complex has been assigned to azomethine proton of the hydrazone where the nitrogen of −C=N− is participating in complex formation.21–26 The 1H NMR spectra of the complexes are provided in the Supporting Information. In the 13 C NMR spectra of the complexes (Supplemental Figures S3 and S6), resonances were observed in the 151- to 155-ppm series was due to the azomethine carbon. The chemical shifts observed in the regions 145–150, 162–165, and 142–145 ppm are due to C–N, C–O, and C–P, respectively. 16 Also, additional information was derived from the 13 C NMR spectra of complexes, which showed the existence of carbons with six different chemical environments (130–135 ppm). The three quaternary carbons arising from the triphenylphosphine aromatic units were in the same magnetic environments. 16 The 31 P NMR spectra exhibited singlets at 22.3 and 23.1 ppm (Supplemental Figures S4 and S7), which suggests that one triphenylphosphine molecule coordinates with the metal ion in the complexes.16,17
Magnetic susceptibility measurements
The complexes were subjected to magnetic susceptibility measurements to obtain further information on their structures. The magnetic susceptibility measurements showed that the complexes are diamagnetic in nature and confirmed a tetra-coordinated configurations. 26
NLO measurements
The open-aperture z-scan technique, developed by Sheik Bahae et al., 27 is widely used to measure the non-linear transmission of light through samples. This technique allows the determination of the non-linear absorption (NLA) coefficient. A Gaussian laser beam is focused spatially through the sample using a lens. The beam is propagated in the direction of the z-axis, and the focal point is chosen from the origin, z = 0. At the origin, the laser beam, which has maximum energy density, will be reduced proportionally to each side of the positive and negative values of the focal point. The experiment proceeds by the sample being placed in the laser beam at different positions relative to the focus (various values of z). This allows the measurement the light transmission. The intensity-dependent transmission of the sample can thus be measured since the sample experiences dissimilar laser intensities at each position. These data give the way to calculate the NLA coefficient.
Using an Nd:YAG laser, at a wavelength of 532 nm, laser pulses at 5-nanosecond intervals were performed to irradiate the samples in 1-mm cuvettes. This provides the SHG of the crystal. The third-order NLO properties were assessed at concentrations and the solvent same as that in UV-Vis studies (i.e. 0.01 M and DMF). DMF was selected as the solvent to increase the solubility and also to minimize the overlap between the absorption spectra of the metal complexes and the wavelength of the z-scan measurement. Most importantly, DMF does not show any NLO response and hence ensures that the absorption obtained is that of the solute. 28 Both complexes, CoL and PdL, showed good non-linear transmission. At higher intensities, the transmission in both samples was reduced (Figures 3 and 4), indicating that these samples may conduct as optical limiters. It is a known fact that optical limiters find uses in protecting light-sensitive devices like cameras from accidental exposure to intense lasers. Optical limiters also play a similar and very important role in protecting human eyes in these situations.
It was observed that the absorption process of a two-photon type gives the best fit to the obtained open-aperture z-scan data. The data obtained for a two-photon absorption process are numerically fitted to the non-linear transmission equation, 29 given by
where T is the transmission; L is the length; R is the surface reflectivity of the sample; α is the linear absorption coefficient; q0 = β(1 − R)I0Leff, where β is the two-photon absorption coefficient; I0 is the on-axis peak intensity; and Leff = 1 − exp(−αl/α).
A knife-edge measurement reveals that the laser beam is Gaussian spatially (Figure 2). The diameter of the beam (full width at half maximum) was found to be 7.82 mm (by calculation). The Rayleigh range in our experiment was 1.8 mm.

Spatial profile of the laser beam (Nd:YAG).
The pattern of the open-aperture characteristics describes the intensity-dependent absorptive optical non-linearity present in the samples with a symmetric dip at z = 0 affirming large absorptive optical non-linearity at strong optical intensities. NLA in the molecules can originate due to multiple components such as on-photon molecular absorption, excitation wavelength, fluence and intensity of laser beam, and pulse duration. The different possible NLA mechanisms are saturable absorption (SA), excited-state absorption (ESA), reverse SA (RSA), 2-photon absorption (2PA), and so on. The molecule exhibiting small one-photon absorption at 532 nm, so ESA is the major process in the existing NLA.28,30–33 Furthermore, 2PA play a dominating role when molecules subjected to a strong nanosecond laser beam.34–36 The normalized dip describes the existence of RSA mechanism in the molecule. 37
The non-linear transmission curves and z-scans are shown in Figures 3 and 4. The data indicate that both complexes behave as good optical limiters, with results revealing that the NLO transmission was reduced at higher intensities. The numerically calculated values of the two-photon absorption coefficient are 5 × 10−11 m W−1 for CoL (1) and 9 × 10−12 m W−1 for PdL (2), respectively.

Non-linear optical transmission in CoL.

Non-linear optical transmission in PdL.
Conclusion
Novel cobalt and palladium complexes of a mixed ligand containing triphenylphosphine and a hydrazone derived from furfural and hydrazine have been synthesized, and structural characterization was performed by elemental analysis, IR, NMR, UV spectroscopy, and mass spectrometry. Elemental analysis confirmed the molecular formulae of the ligand and complexes. Keeping in view the favored geometries and on the basis of all the above studies, square planar structures have been put forward for both the cobalt and palladium complexes. Preliminary investigations on the NLO measurements of both complexes performed at 532 nm using nanosecond laser pulses show promising responses. Both the samples display an excellent optical-limiting capability. By virtue of being optical limiters, these complexes can possibly serve as materials with potential applications in the field of optoelectronics and photonics. Future work on the optical power limiting and optical switching studies of the complexes in the form of thin films will be reported shortly.
Experimental
Materials
Analytical grade chemicals were used for the synthesis of all the compounds. A standard procedure available in the literature was used to purify the solvents. 38 Furfural, hydrazine hydrate, and triphenylphosphine were purchased from Merck and were used without further purification. CoCl2·6H2O and PdCl2 were obtained from Spectrochem chemicals, and were used without further purification. The starting complex, [CoCl2(PPh3)2], was prepared by heating a solution of CoCl2·6H2O and triphenylphosphine in glacial acetic acid. 39 The other starting material, [PdCl2(PPh3)2], was synthesized by refluxing anhydrous PdCl2 and triphenylphosphine in tetrahydrofuran for 5 h. 40
Physical measurements
The electronic spectra of solutions of the complexes in DMF were recorded on a GBC UV-Vis double beam spectrophotometer in the 200- to 800-nm range. FTIR spectra were obtained using a Thermo Nicolet Avatar FTIR spectrometer, with samples prepared with KBr powder in the frequency range 400–4000 cm−1. The C, H, N, S, and O composition analysis was performed on a Thermo Flash elemental analyzer. NMR spectra were collected on a Bruker AMX 400 instrument with TMS as the internal standard. The magnetic moment measurements were obtained using a Sherwood Scientific instrument.
Synthesis of the hydrazone: (E)-(furan-2-ylmethylene) hydrazine (1 )
The hydrazone was prepared using a procedure reported in the literature.41,42 Hydrazine was slowly added to a methanolic solution of furfural and stirred for about 30 min at room temperature.
Synthesis of the metal complexes
Cobalt complex [CoL] (2 )
An equimolar molar ratio of CoCl2(PPh3)2 and the ligand was refluxed for 2 h in an acetone:methanol (4:1) solution. CoCl2(PPh3)2 (1 mmol, 0.65 g) and the ligand (
Palladium complex [PdL] (3 )
A mixture of [PdCl2(PPh3)2] and the ligand (
Supplemental Material
Supplementary – Supplemental material for Mixed ligand cobalt and palladium complexes containing triphenylphosphine and a hydrazone: Synthesis and application in non-linear optics
Supplemental material, Supplementary for Mixed ligand cobalt and palladium complexes containing triphenylphosphine and a hydrazone: Synthesis and application in non-linear optics by Dileep Ramakrishna in Journal of Chemical Research
Footnotes
Acknowledgements
The author would like to thank the IISc, Bangalore for the NMR analyses and RRI, Bangalore for the NLO measurements.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author received no financial support for the research, authorship, and/or publication of this paper.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.