Abstract
Organosulfur compounds with unsymmetrical S–S bonds are usually called unsymmetrical disulfides and are widely used in the biological, medicinal, and chemical fields. Their versatility has guided the development of various new methods for the synthesis of disulfides. In recent years, the synthesis of disulfides by cross-dehydrogenation of thiols has attracted much attention due to its high atomic economy. Herein, this review summarizes progress toward the synthesis of unsymmetrical disulfides under chemical oxidation, electrooxidation, or photocatalysis by cross-dehydrogenation of thiols.

Introduction
Disulfides exist in many biologically active natural products1–2 and are widely employed in the pharmaceutical industry,3–7 materials sciences, 8 and so on. Unsymmetrical disulfides have attracted significant attention because of their role in stabilizing protein structures 9 and for their promising use in the clinical application of tumor therapies.10,11 Thus effective methods for the construction of unsymmetrical disulfides are increasingly important.
A number of methods have been reported for the construction of unsymmetrical disulfides. For example, thiocyanates,12,13 alkyl halides,14,15 and thioacetates16,17 can be used as sulfur sources to build disulfide bonds. However, these methods often rely on more complicated processes. The method of synthesizing unsymmetrical disulfides by employing commercially available thiols is relatively simple and has high atom economy. The synthesis of unsymmetrical disulfides predominantly includes nucleophilic substitution of the thiol18–26 and exchange with a disulfide bond.27–31 Another method relies on an oxidative dehydrogenation coupling reaction of S–H/S–H bonds. The former two methods require pre-functionalization of the thiol and multi-step reactions. This makes the study of the latter method more attractive. This review therefore focuses on the synthesis of unsymmetrical disulfides via the oxidative cross-dehydrogenation of thiols.
Oxidative cross-dehydrogenation of thiols
Sodium tellurite as the oxidant
Sodium tellurite is commonly used in organic synthesis. In 1991, Suzuki et al.
32
reported that sodium tellurite, under phase-transfer conditions, allows for the synthesis of unsymmetrical disulfides via thiols with high selectivity under mild conditions (Scheme 1). Under their reported reaction conditions, sodium tellurite is used as an oxidant and can instantly oxidize aryl thiols and benzyl thiols to their corresponding disulfides. The oxidation of primary thiols and secondary thiols is slow, while tertiary thiols are unreactive. Suzuki’s group explored the limitations of the reaction and found that the outstanding ability of sodium tellurite to distinguish between thiol types is reflected by an efficient one-step synthesis of a number of unsymmetrical disulfides. For example, when a 9:10 mixture of benzyl thiol and tert-butyl thiol is reacted with Na2TeO3 in the presence of tetrabutylammonium hydroxide under biphasic conditions for 36 h, benzyl tert-butyl disulfide (

Synthesis of unsymmetrical disulfides using sodium tellurite as the oxidant.
Iodine as the oxidant or mediator
N-Iodosuccinimide (NIS) can also act as a catalyst in the synthesis of unsymmetrical disulfides. In 2014, Yuan et al.
33
synthesized unsymmetrical aryl tertiary alkyl disulfides. In this reaction, 2-mercaptobenzothiazole
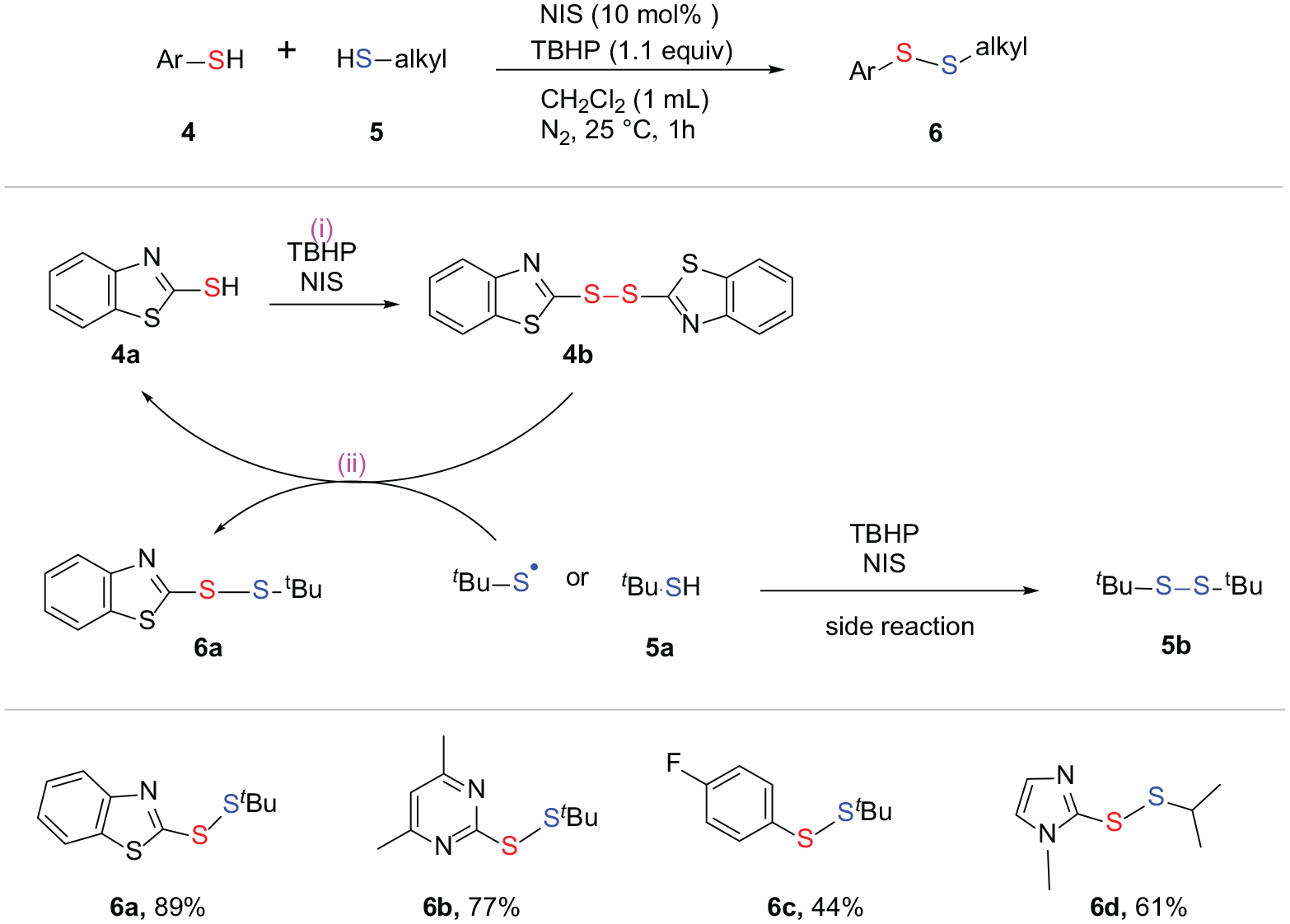
Synthesis of unsymmetrical disulfides with NIS as the catalyst.
Water plays an important role in many organic reactions.34,35 In 2019, Parida et al.
36
reported the use of iodine as a catalyst and 4-dimethylaminopyridine (DMAP)/water as a reaction promoter, in order to synthesize unsymmetrical organic disulfides in EtOH-H2O (2:1) as the solvent via an umpolung method (Scheme 3). This methodology is suitable for the synthesis of unsymmetrical diaryl disulfides (
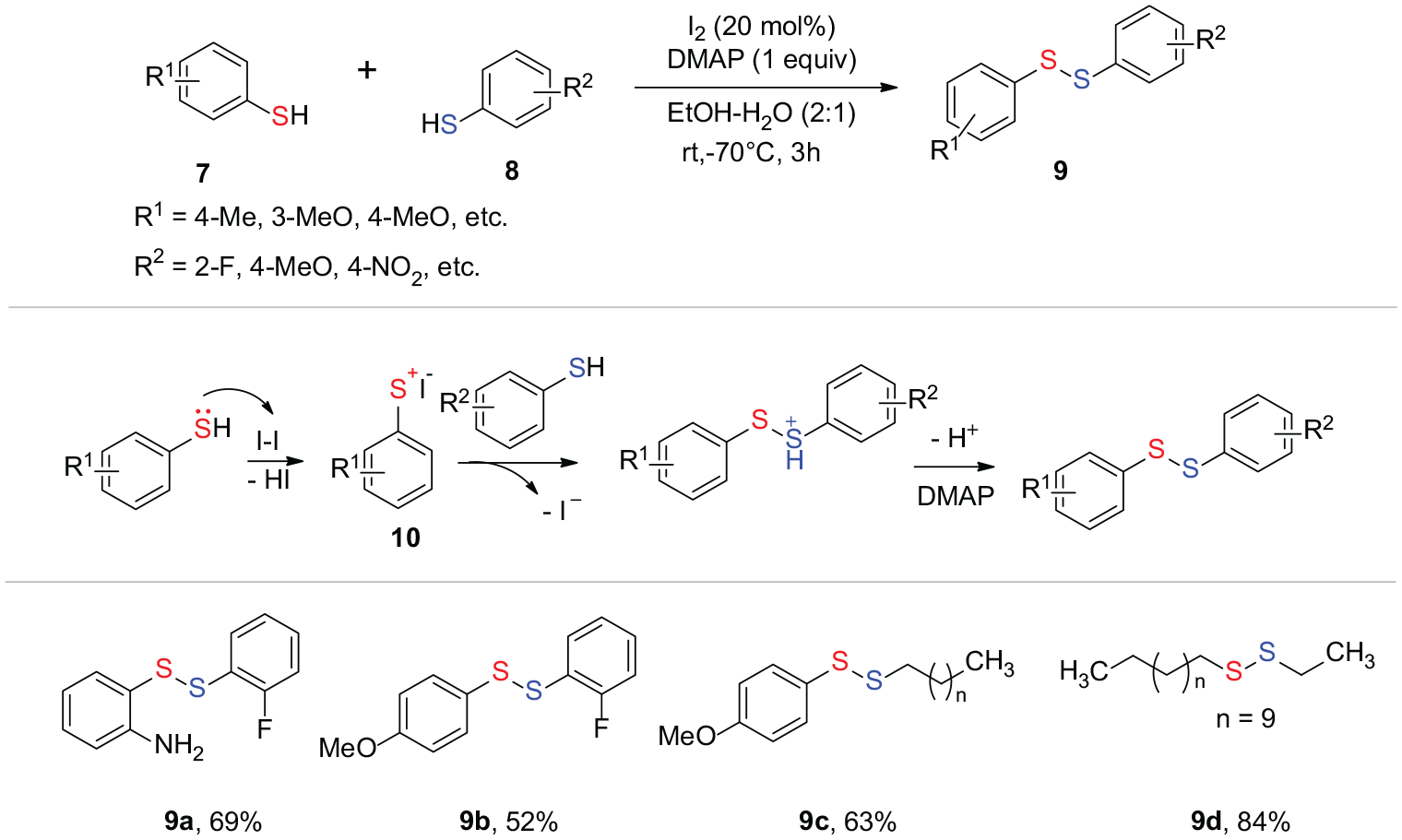
Synthesis of unsymmetrical disulfides with I2 as the catalyst.
Trichloroisocyanuric acid as the oxidant
Trichloroisocyanuric acid (TCCA) is often used as a chlorinating agent and oxidizing agent in synthesis.
37
In 2016, Yang et al.
38
used TCCA as the oxidant to construct unsymmetrical disulfides under mild conditions. Unsymmetrical aromatic-aromatic disulfides and aromatic-aliphatic disulfides can be obtained in good yields with a reaction time of only 5 min. A reasonable mechanism has been proposed as shown in Scheme 4. After adding TCCA to the thiol, two sulfur intermediates

Synthesis of unsymmetrical disulfides with TCCA as the catalyst.
H2O2 as the oxidant
H2O2 is an ideal reagent in green chemistry. Hydrogen peroxide often acts as an oxidant in the synthesis of disulfides. In 1996, Marchand-Brynaert et al.
39
reported the coupling of cysteamine and 3-mercaptopropionic acid using hydrogen peroxide as the oxidant for the synthesis of unsymmetrical disulfide
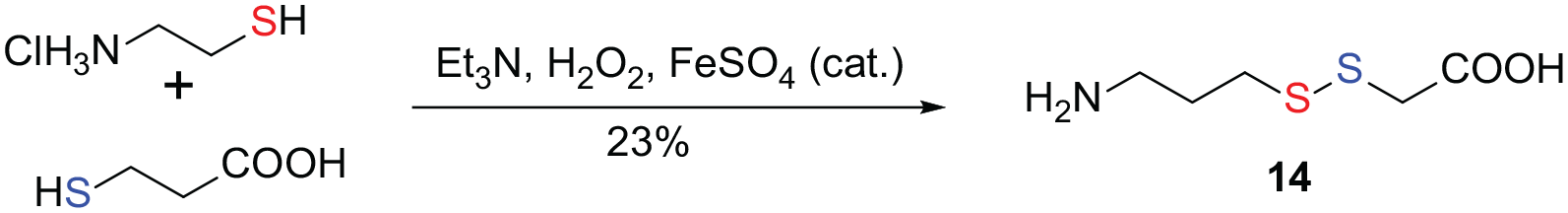
Synthesis of unsymmetrical disulfide
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) as the oxidant
Choosing the appropriate oxidant is important for the preparation of unsymmetrical disulfides via the oxidative S–H/S–H cross-coupling method. After investigating many oxidants (such as DDQ, ammonium cerium nitrate, pyridinium chlorochromate (PCC), potassium ferricyanide, and manganese acetate), in 2011, Vandavasi et al.
40
disclosed DDQ as an oxidant to efficiently synthesize unsymmetrical disulfides (Scheme 6). Under the optimum conditions consisting of dichloromethane as the solvent, a reaction temperature of 0 °C and a short reaction time of 5 min, the desired products

Synthesis of unsymmetrical disulfides
In 2013, inspired by Wang, Smith et al. 41 envisaged employing DDQ for the synthesis of unsymmetrical glycosyl disulfides. Seven unsymmetrical glycosyl disulfides have been synthesized directly from the corresponding glycosyl thiols by oxidation with DDQ (Scheme 7). The coupling of α-thiols and β-thiols proceeded smoothly, giving the moderate yields (32%–78%) of the cross-coupling products. The homo-coupling of thiols was also obtained, and the mechanism of the reaction was not revealed in their work.

Synthesis of unsymmetrical glycosyl disulfide
DMSO as the oxidant
In 2012, Miller et al. 42 used dithiol as a substrate and DMSO as a mild oxidant to synthesize an unsymmetrical disulfide peptide. However, the yield of the reaction was low (20% of the theoretical yield) (Scheme 8).
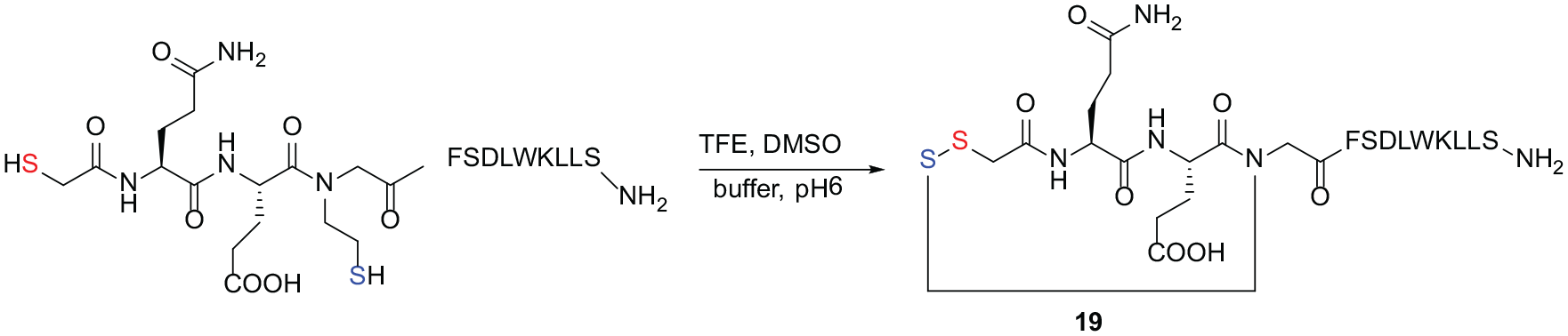
Synthesis of an unsymmetrical glycosyl disulfide
Diethyl azodicarboxylate as the oxidant
Diethyl azodicarboxylate (DEAD) can also be used as an oxidant for thiol couplings for the synthesis of unsymmetrical glycosyl disulfides.
43
Various aromatic and heterocyclic thiols

Synthesis of unsymmetrical glycosyl disulfides
O2 as the oxidant
Oxygen is cheap and readily available, and it is environmentally friendly. In 2017, Dou et al.
44
synthesized unsymmetrical disulfides via the catalytic aerobic oxidation of thiol cobalt phthalocyanine (Scheme 10). The products resulting from the cross-coupling of two electron-rich aromatic thiols can be obtained in yields of 91%–94%. Aromatic thiols can also react with aliphatic thiols, providing higher yields of the corresponding products

Co-catalyzed synthesis of unsymmetric disulfides
In 2019, Song et al.
45
demonstrated a novel K2CO3 catalyzed aerobic oxidative cross-coupling of thiols for the preparation of unsymmetrical disulfides

K2CO3-catalyzed synthesis of unsymmetric disulfides
Electro-oxidative cross-dehydrogenation of thiols
In 2018, Lei et al.
46
developed a novel S–H/S–H electrooxidation cross-coupling of aryl mercaptan with alkyl mercaptan, resulting in hydrogen evolution, to prepare unsymmetrical disulfides (Scheme 12). The ratio of aryl mercaptan to alkyl mercaptan was 1:1, and various heterocyclic mercaptans and thiophenols were suitable substrates for this transformation. In the control experiments, when using dimer

Electro-oxidative synthesis of unsymmetric disulfides.
Photocatalyzed cross-dehydrogenation of thiols
Photocatalytic oxidation is another green and effective method for the synthesis of unsymmetrical disulfides in the absence of extra oxidants. Dethe et al.
47
developed and used tris(2-phenylpyridine)iridium (Ir(ppy)3) as a photocatalyst to dehydrogenate mercaptan to the corresponding unsymmetrical disulfide. This methodology can generate high yields with a photocatalyst loading of only 0.5 mol% under white LED light, and has good functional group tolerance (nitro, methoxy, ester and carboxyl functionalities are all tolerated). Scheme 13 shows a reasonable mechanism for the reaction. First, Ir(ppy)3 is excited by visible light to produce Ir(Ⅲ)*. Ir(Ⅲ)* gains electrons from R1SH/R2SH which is reduced to Ir(Ⅱ), and at the same time R1SH/R2SH loses protons to form R1S/R2S. These radicals are replaced by a second thiol to form disulfide radical anions

Photocatalytic synthesis of unsymmetrical disulfides.
Conclusion and future prospects
In the past decades, many novel strategies for the synthesis of various unsymmetrical disulfides via the cross-dehydrogenation of thiols have been developed. These unsymmetrical disulfides are important precursors of promising compounds for use in the pharmaceutical industry and materials field. Although numerous effective methods for the synthesis of unsymmetrical disulfides have been established, more environmentally green, simpler, and efficient strategies for the construction of unsymmetrical disulfides are still necessary. In particular, it is highly desirable to find alternative sulfur sources48,49 to help broaden the substrate scope.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Natural Science Foundation of China (21762018 and 21961014), the program of Qingjiang Excellent Young Talents of Jiangxi University of Science and Technology (JXUSTQJBJ2018003), and the Science and the Technology Innovation Outstanding Young Talents Program of Jiangxi Province (20192BCBL23009).
