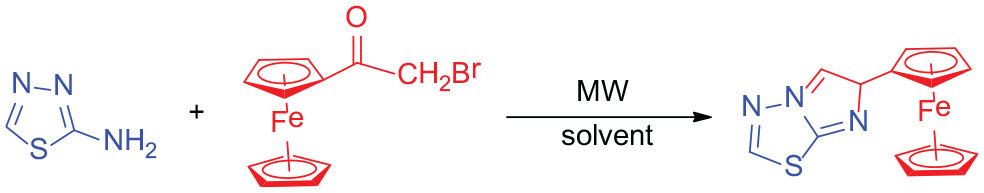Abstract
Ferrocenyl imidazolo[2,1-b]-1,3,4-thiadiazoles are synthesized using 2-amino-5-substituted-1,3,4-thiadiazole and α-bromoacetyl ferrocene as substrates under microwave-assisted and catalyst-free conditions. The structures are characterized by infrared, 1H nuclear magnetic resonance, 13C nuclear magnetic resonance, mass spectrometry, and elemental analysis. Compared with conventional methods, the present protocol has the advantages of being rapid, efficient, environmentally friendly, and low cost. The results of biological activity studies showed that the products displayed better activities than those of 2-amino-5-substituted-1,3,4-thiadiazole. The biological activities of compounds in which R = phenyl were better than those with alkyl groups. Compounds with electron-withdrawing groups on the aryl moiety showed increased biological activities.

Introduction
Imidazolo[2,1-b]-1,3,4-thiadiazole derivatives were first discovered in 1952. 1 The near-planar and molecular rigid structure conferred them with distinctive physicochemical and biological properties. Significant progress has been made with these compounds in the field of pharmaceutical applications with continuous in-depth studies of this heterocyclic system in biology and chemistry. In the design process of modern drugs, it was discovered that imidazo[2,1-b]-1,3,4-thiadiazole derivatives display selective inhibition toward several receptors and enzymes, thus making them interesting as lead targets for potential drugs.
A large number of imidazo[2,1-b]-1,3,4-thiadiazole derivatives have been found to show various biological activities, including antituberculosis,2,3 antibacterial,4–6 anticancer,7,8 and anti-inflammatory.9,10 The imidazo[2,1-b]-1,3,4-thiadiazole moiety is relatively stable but examples in which the 2,5 and 6 positions contain a C–H bond can undergo electrophilic substitution, 7 nucleophilic substitution, nitration, 11 and the Vilsmeier–Haack reaction. 12 Imidazo[2,1-b]-1,3,4-thiadiazoles can also coordinate with metals to form complexes 13 or be used in its ring-opening reactions 11 under specific conditions.
Up to now, imidazo[2,1-b]-1,3,4-thiadiazoles are mostly obtained by ring-forming reactions of 1,3,4-thiadiazole compounds and α-haloketones. The traditional method involves refluxing a mixture of a 1,3,4-thiadiazole and α-haloketone in an alcohol solvent for several hours (intermolecular reaction) followed by intramolecular cyclization to give imidazo[2,1-b]-1,3,4-thiadiazoles. With the development of different organic synthesis technologies, ionic liquid-catalyzed 14 and microwave-assisted reactions 8 have also been applied for the synthesis of imidazo[2,1-b]-1,3,4-thiadiazoles. Compared with traditional methods, these approaches improve the reaction efficiency.
In recent years, our group has focused on the development of green organic synthesis technologies,15–19 and herein, we have developed a microwave-assisted method to synthesize imidazo[2,1-b]-1,3,4-thiadiazoles (Scheme 1). Importantly, the present protocol did not need any catalyst, and the reaction efficiency was very high under the given conditions. A series of novel ferrocenyl imidazo[2,1-b]-1,3,4-thiadiazoles, which were characterized by infrared (IR), 1H nuclear magnetic resonance (NMR), 13C NMR, mass spectrometry (MS), and elemental analysis, were obtained. Furthermore, the biological activities of the prepared compounds were explored.

The synthesis of ferrocenyl imidazo[2,1-b]-1,3,4-thiadiazoles
Results and discussion
Synthetic mechanism
The ferrocenyl imidazo[2,1-b]-1,3,4-thiadiazoles were synthesized by an intermolecular nucleophilic substitution reaction and an intramolecular nucleophilic addition reaction of 2-amino-5-substituted-1,3,4-thiadiazoles and α-bromoacetyl ferrocene. First, the secondary amine was formed by nucleophilic substitution of α-bromoacetyl ferrocene and 2-amino-1,3,4-thiadiazole. An intramolecular nucleophilic addition reaction then yielded the intermediate product. Finally, ferrocenyl imidazo[2,1-b]-1,3, 4-thiadiazoles

The mechanism for synthesis of ferrocenyl imidazo[2,1-b]-1,3,4-thiadiazoles.
Optimization of the reaction conditions
The conventional method for the synthesis of 2-substituted-6-ferrocenyl-imidazo[2,1-b]-1,3,4-thiadiazoles requires a high temperature and long reaction time and is generally refluxed in an alcohol for 12–14 h. In addition, due to the limitations, the overall yield was low, the probability of side reactions is greatly increased, and the by-products generated are difficult to remove.
Heating in a microwave oven transfers electromagnetic energy to the inside of a heated substance.
20
The frictional collisions between molecules generate thermal energy to increase the temperature of the substance. Using microwave-assisted synthesis, the energy is directly applied to the internal system of the reacting molecules, thereby making the energy utilization more effective to improve the reaction efficiency, greatly accelerate the reaction speed, shorten the reaction time, and reduce the occurrence of side reactions. Microwave-assisted synthesis can greatly reduce the reaction time (about 10 min) and also lowers the reaction temperature,
21
which effectively reduces the incidence of side reactions. At the same time, it has the advantages of convenient operation and low cost. A comparison between the conventional and microwave-assisted methods for the synthesis of compounds
Comparison of the synthesis of
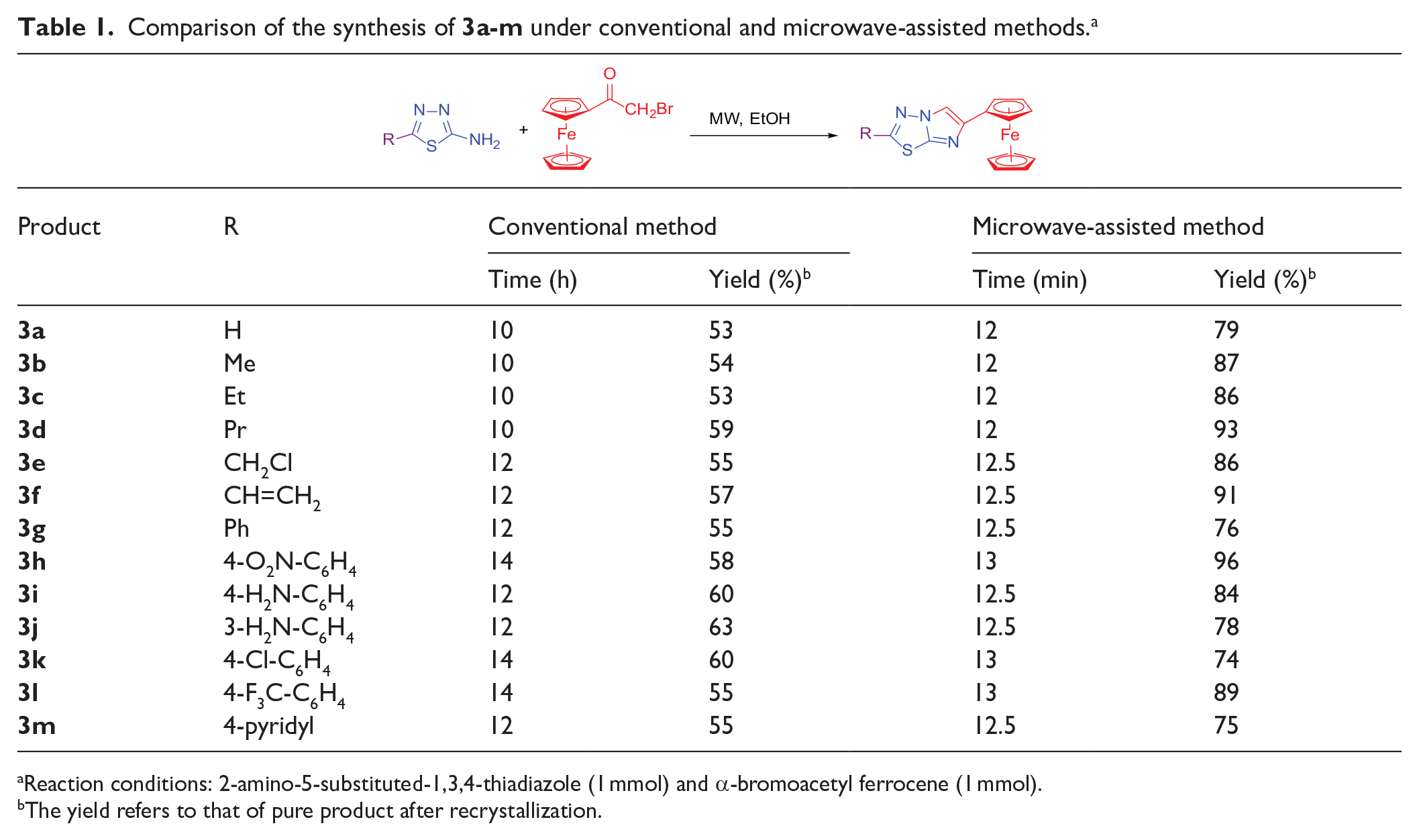
Reaction conditions: 2-amino-5-substituted-1,3,4-thiadiazole (1 mmol) and α-bromoacetyl ferrocene (1 mmol).
The yield refers to that of pure product after recrystallization.
From Table 1, it can be seen clearly that when R was an alkyl group, the reaction time was shorter than that when R was an aryl group. The electron-donating effect of the alkyl group increased the electron cloud density on the N of the -NH2, which favored the reaction for the nucleophilic substitution reaction with α-bromoacetyl ferrocene; hence the reaction time was shorter. In contrast, the reaction time when R was an electron-withdrawing aryl group was longer, and the nucleophilic substitution reaction with α-bromoacetyl ferrocene was more difficult.
Next, the effect of the catalyst on the yield of ferrocenyl imidazo[2,1-b]-1,3,4-thiadiazole (
The effect of catalysts on the yield of

Reaction conditions: 2-amino-1,3,4-thiadiazole (1 mmol) and α-bromoacetyl ferrocene (1 mmol).
The yield refers to that of pure product after recrystallization.
The effect of the solvent on the yield of
Effect of the solvent on the yield of

DMF: dimetyl formamide; DMSO: dimethyl sulfoxide.
Reaction conditions: 2-amino-1,3,4-thiadiazole (1 mmol) and α-bromoacetyl ferrocene (1 mmol).
The yield refers to that of pure product after recrystallization.
Biological activity
The synthesized compounds in the present study were screened in vitro for antibacterial and antifungal activity against various gram-positive and gram-negative bacteria, and fungal strains using the agar cup-plate method.21,22 Tetracycline and fluconazole were used as standard drugs for antibacterial and antifungal activity, respectively, at concentrations of 3 g L−1. The zones of inhibition for
Antibacterial and antifungal activity (concentration: used 3 mg mL−1 in DMSO) of

DMSO: dimethyl sulfoxide.
Zone of inhibition (mm): <20, weak; 20–25, moderate; >25, significant.
As can be seen from Table 4, the activities of
Conclusion
In summary, ferrocenyl imidazo[2,1-b]-1,3,4-thiadiazoles have been synthesized by a microwave-assisted method under catalyst-free conditions. The results showed that this method had the advantages of being rapid, gives high yields, adopts a simple method, and is environmentally friendly. The antibacterial and antifungal activity has also been studied with all the synthesized compounds showing interesting biological activities. Among them, aryl-substituted compounds were more active than alkyl-substituted examples. Compounds with electron-withdrawing aryl groups gave enhanced activities, and all compounds have better biological activity than the starting 2-amino-5-substituted-1,3,4-thiadiazoles.
Experimental
Materials and method
All chemicals were analytical grade and were used without further purification. The melting points were determined in an open glass capillary using a Titan TY1-0004 and are uncorrected. IR spectra (400–4000 cm−1) were recorded on a Bruker Vector-22 FT-IR spectrometer with samples prepared as KBr pellets. The 1H NMR and 13C NMR spectra were recorded on a Bruker Avance-400 MHz spectrometer in DMSO-d6 with tetramethylsilane (TMS) as an internal standard. MS were recorded on a Waters Ultima Global Spectrometer (ESI). The C, H, and N analyses were performed with a Fisons EA 1108 (CHNSO) elemental analyzer. Microwave reaction was carried out in a Honghu XH-MC-1 microwave reactor.
The 2-amino-5-substituted-1,3,4-thiadiazoles and α-bromoacetyl ferrocene were prepared as reported.23,24
General procedure for conventional synthesis of the ferrocenyl imidazolo[2,1-b]-1,3,4-thiadiazoles 3
A mixture of 2-amino-5-substituted-1,3,4-thiadiazole (1 mmol), α-bromoacetyl ferrocene (1 mmol), and ethanol (5 mL) was refluxed for 10–12 h. After completion (monitored by thin-layer chromatography (TLC)), the solvent was removed by distillation and the obtained crude product was then purified by column chromatography using ethyl acetate hexane (3:1) to obtain the product
General procedure for microwave-assisted catalyst-free synthesis of ferrocenyl imidazolo [2,1-b]-1,3,4-thiadiazoles 3
2-Amino-5-substituted-1,3,4-thiadiazole (1 mmol), α-bromoacetyl ferrocene (1 mmol), and ethanol (5 mL) were mixed uniformly in a dry crucible and then subjected to microwave irradiation of 700 W for 30 s. The crucible was removed from the microwave, ethanol (5 mL) was added, and then subjected to microwave irradiation; this process was repeated for 20–25 times until the reaction was complete (monitored by TLC). Then, the reaction was allowed to cool to room temperature. Water of 10 mL was added and the mixture was neutralized to pH 7–8 with saturated sodium carbonate solution. The obtained solid was filtered, washed with water, and dried. The crude product was recrystallized in dimetyl formamide (DMF) to afford
6-Ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3a): Orange-red solid; m.p. 278–280 °C; yield 79%. IR (KBr): 3081, 3013, 1556, 1427, 1141, 1063 cm−1. 1H NMR (CDCl3, 400 MHz): δ 4.23 (s, 5H), 4.59 (t, 2H, J = 7.2 Hz), 4.84 (t, 2H, J = 7.2 Hz), 8.05 (s, 1H), 8.56 (s, 1H). 13C NMR (101 MHz, DMSO-d6): δ 155.3, 147.9, 138.2, 125.1, 57.7. ESI-MS: m/z = 310.06 [M + H]+. Anal. calcd. for C14H11FeN3S: C, 54.39; H, 3.59; N, 13.59; found: C, 54.45; H, 3.87; N, 13.63.
2-Methyl-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3b): Brown solid; m.p. 256–258 °C; yield 87%. IR (KBr): 3093, 2965, 1533, 1375, 1194, 1070 cm−1. 1H NMR (CDCl3, 400 MHz): δ 2.74(s, 3H), 4.24 (s, 5H), 4.58 (t, 2H, J = 4.0 Hz), 4.79 (t, 2H, J = 6.8 Hz), 8.04 (s, 1H).13C NMR (101 MHz, DMSO-d6): δ 149.9, 145.2, 137.6, 127.7, 60.2, 18.3. ESI-MS: m/z = 324.08 [M + H]+. Anal. calcd. for C15H13FeN3S: C, 55.74; H, 4.05; N, 13.00; found: C, 55.70; H, 4.11; N, 13.13.
2-Ethyl-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3c): Brown solid; m.p. > 300 °C; yield 86%. IR (KBr): 3076, 2953, 1563, 1406, 834 cm−1. 1H NMR (CDCl3, 400 MHz): δ 1.35 (t, 3H, J = 7.2 Hz), 2.88 (q, 2H, J = 6.8 Hz), 4.28 (s, 5H), 4.64 (t, 2H, J = 8.0 Hz), 4.89 (t, 2H), 8.05 (s, 1H). 13C NMR (101 MHz, DMSO-d6): δ 170.2, 147.2, 140.6, 120.6, 62.7, 23.1, 11.0. ESI-MS: m/z = 338.10 [M + H]+. Anal. calcd. for C16H15FeN3S: C, 56.99; H, 4.48; N, 12.46; found: C, 57.07; H, 4.55; N, 12.49.
2-n-Propyl-6-ferrocenyl-imidazolo[2,1-b]-1,3,4-thiadiazole (3d): Brown solid; m.p. > 300 °C; yield 93%. IR (KBr): 3064, 2979, 2928, 1588, 1390, 1093 cm−1. 1H NMR (CDCl3, 400 MHz): δ 1.01 (t, 3H, J = 7.2 Hz), 1.80 (m, 2H, J = 7.2 Hz), 2.87 (t, 2H, J = 8.0 Hz), 4.22 (s, 5H), 4.60 (t, 2H, J = 3.2 Hz), 4.91 (t, 2H, J = 6.8 Hz), 8.02 (s, 1H). 13C NMR (101 MHz, DMSO-d6): δ 177.9, 149.0, 142.3, 120.4, 62.1, 40.2, 22.1, 10.6. ESI-MS: m/z = 352.11 [M + H]+. Anal. calcd. for C17H17FeN3S: C, 58.13; H, 4.88; N, 11.96; found: C, 58.19; H, 4.82; N, 11.91.
2-Chloromethyl-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3e): Orange-red solid, m.p. 287–289 °C; yield 86%. IR (KBr): 3046, 2970, 1760, 1392, 1114 cm−1.1H NMR (CDCl3, 400 MHz): δ 4.23 (s, 5H), 4.63(t, 2H, J = 3.2 Hz), 4.75(s, 2H), 5.01 (t, 2H, J = 8.0 Hz), 8.02 (s, 1H).13C NMR (101 MHz, DMSO-d6): δ 180.2, 150.5, 144.2, 137.5, 59.9. ESI-MS: m/z = 358.99 [M + H]+. Anal. calcd. for C15H12ClFeN3S: C, 50.37; H, 3.38; N, 11.75; found: C, 50.29; H, 3.40; N, 11.64.
2-Vinyl-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3f): Brown solid; m.p. 247–248 °C; yield 91%. IR (KBr): 3117, 3032, 1628, 1523, 1181 cm−1.1H NMR (CDCl3, 400 MHz): δ 4.26 (s, 5H), 4.59 (t, 2H, J = 8.4 Hz), 4.95 (t, 2H, J = 7.2 Hz), 5.19 (d, 1H, J = 16.0 Hz), 5.62 (d, 1H, J =10.4 Hz), 6.65 (m, 1H), 8.02 (s, 1H). 13C NMR (101 MHz, DMSO-d6): δ 170.2, 144.9, 142.3, 134.7, 130.2, 121.3, 68.8. ESI-MS: m/z = 336.04 [M + H]+. Anal. calcd. for C16H13FeN3S: C, 57.33; H, 3.91; N, 12.54; found: C, 57.25; H, 4.02; N, 12.48.
2-Phenyl-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3g): Brown solid; m.p. 258–261 °C; yield 76%. IR (KBr): 3120, 3073, 1597, 1508, 1393, 1014, 770 cm−1.1H NMR (CDCl3, 400 MHz): δ 4.29 (s, 5H), 4.56 (t, 2H, J = 7.2 Hz), 4.97 (t, 2H, J = 6.8 Hz), 7.33 (m, 5H), 8.02 (s, 1H).13C NMR (101 MHz, DMSO-d6): δ 157.9, 150.8, 144.6, 138.6, 132.0, 131.2, 129.2, 127.5, 122.9, 57.9. ESI-MS: m/z = 386.10 [M + H]+. Anal. calcd. for C20H15FeN3S: C, 62.35; H, 3.92; N, 10.91; found: C, 62.27; H, 3.87; N, 10.98.
2-(4-Nitrophenyl)-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3h): Brown solid; m.p. 276–279 °C; yield 96%. IR (KBr): 3032, 1587, 1538, 1477, 1088, 740 cm−1. 1H NMR (CDCl3, 400 MHz): δ 4.24 (s, 5H), 4.59 (t, 2H, J = 8.0 Hz), 5.04 (t, 2H, J = 8.4 Hz), 7.65 (m, 4H), 8.01 (s, 1H).13C NMR (101 MHz, DMSO-d6): δ 160.2, 155.9, 148.6, 137.2, 130.7, 129.0, 122.6, 118.9, 62.7. ESI-MS: m/z = 431.08 [M + H]+. Anal. calcd. for C20H14FeN4O2S: C, 55.83; H, 3.28; N, 13.02; found: C, 55.89; H, 3.19; N, 13.10.
2-(4-Aminophenyl)-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3i): Brown solid; m.p. 258–261 °C; yield 84%. IR (KBr): 3374, 3301, 3258, 3027, 1532, 1423, 1083, 710 cm−1.1H NMR (CDCl3, 400 MHz) δ 4.27 (s, 5H), 4.59 (t, 2H, J = 7.2 Hz), 4.98 (t, 2H, J = 8.0 Hz), 6.27 (s, 2H), 7.37 (m, 4H), 8.03 (s, 1H). 13C NMR (101 MHz, DMSO-d6): δ 167.9, 155.6, 152.7, 146.9, 133.7, 127.1, 125.5, 117.6, 111.2, 109.3, 59.9. ESI-MS: m/z = 401.11 [M + H]+. Anal. calcd. for C20H16FeN4S: C, 60.01; H, 4.03; N, 14.00; found: C, 59.91; H, 4.06; N, 13.96.
2-(3-Aminophenyl)-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3j): Brown solid; m.p. 250–252 °C; yield 78%. IR (KBr): 3334, 3268, 3063, 1548, 1439, 1051, 768, 678 cm−1. 1H NMR (CDCl3, 400 MHz): δ 4.25 (s, 5H), 4.57 (t, 2H, J = 8.0 Hz), 4.76 (t, 2H, J = 6.8 Hz), 6.29 (s, 2H), 7.31 (m, 4H), 8.05 (s, 1H).13C NMR (101 MHz, DMSO-d6): δ 157.2, 132.1, 137.6, 136.4, 131.9, 128.5, 125.5, 117.7, 116.9, 110.2, 109.9, 63.7. ESI-MS: m/z = 400.10 [M + H]+. Anal. calcd. for C20H16FeN4S: C, 60.01; H, 4.03; N, 14.00; found: C, 60.04; H, 4.06; N, 14.04.
2-(4-Chlorophenyl)-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3k): Brown solid; m.p. 255–258 °C; yield 74%. IR (KBr): 3200, 3005, 1639, 1511, 1451, 1028, 833 cm−1. 1H NMR (CDCl3, 400 MHz) δ 4.28 (s, 5H), 4.56 (t, 2H, J = 3.2 Hz), 4.89 (t, 2H, J = 7.2 Hz), 7.34 (m, 4H), 8.03 (s, 1H). 13C NMR (101 MHz, DMSO-d6): δ 163.1, 155.6, 148.9, 142.1, 137.6, 133.7, 129.4, 125.0, 59.2. ESI-MS: m/z = 420.99 [M + H]+. Anal. calcd. for C20H14FeClN3S: C, 57.23; H, 3.36; N, 10.01; found: C, 57.28; H, 3.31; N, 10.08.
2-(4-Trifluoromethylphenyl)-6-ferrocenyl-Imidazolo [2,1-b]-1,3,4-thiadiazole (3l): Brown solid; m.p. > 300 °C; yield 89%. IR (KBr): 3105, 3050, 1607, 1513, 1473, 1089, 973, 830, 701 cm−1. 1H NMR (CDCl3, 400 MHz): δ 4.26 (s, 5H), 4.58 (t, 2H, J = 6.8 Hz), 5.06 (t, 2H, J = 6.8 Hz), 7.88 (m, 4H), 8.08 (s, 1H). 13C NMR (101 MHz, DMSO-d6): δ 155.9, 152.9, 142.6, 140.7, 138.0, 130.4, 122.3, 120.5, 114.7, 65.2. ESI-MS: m/z = 454.08 [M + H]+. Anal. calcd. for C21H14F3FeN3S: C, 55.65; H, 3.11; N, 9.27; found: C, 55.57; H, 3.06; N, 9.20.
2-(4-Pyridyl)-6-ferrocenyl-Imidazolo[2,1-b]-1,3,4-thiadiazole (3m): Brown solid; m.p. 286–289 °C; yield 89%. IR (KBr): 3040, 1587, 1100, 916 cm−1. 1H NMR (CDCl3, 400 MHz): δ 4.26 (s, 5H), 4.59 (t, 2H, J = 3.2 Hz), 4.99 (t, 2H, J = 7.2 Hz), 8.06 (s, 1H), 8.66 (m, 4H). 13C NMR (101 MHz, DMSO-d6): δ 166.2, 157.2, 150.6, 148.9, 138.2, 130.2, 116.9, 113.2, 70.2. ESI-MS: m/z = 387.09 [M + H]+. Anal. calcd. for C19H14FeN4S: C, 59.08; H, 3.65; N, 14.51; found: C, 59.17; H, 3.72; N, 14.50.
Supplemental Material
supporting_information_6 – Supplemental material for Ferrocenyl imidazolo[2,1-b]-1,3,4-thiadiazoles: A microwave-assisted catalyst-free synthesis, characterization, and biological activities
Supplemental material, supporting_information_6 for Ferrocenyl imidazolo[2,1-b]-1,3,4-thiadiazoles: A microwave-assisted catalyst-free synthesis, characterization, and biological activities by Yuting Liu, Lan Yang and Dawei Yin in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are thankful to the instrument analysis center of Shaanxi University of Science and Technology for Fourier-transform infrared (FTIR), 1H nuclear magnetic resonance (NMR), and elemental analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful for the financial support from the Natural Science Foundation of Shaanxi Province (2018JM7046).
Supplemental material
The IR and 1H NMR spectrums of compounds 3a-m are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.