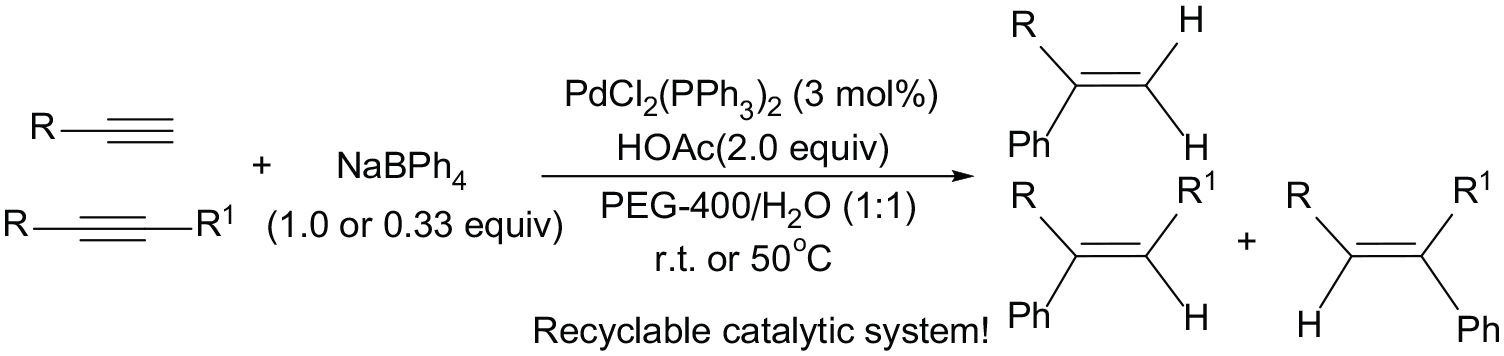
Abstract
A stable and efficient PdCl2(PPh3)2/PEG-400/H2O catalytic system for the hydrophenylation reaction of alkynes has been developed. In the presence of 3 mol% PdCl2(PPh3)2 and 2 equiv. of HOAc, the hydrophenylation of both terminal and internal alkynes with sodium tetraphenylborate proceeded smoothly in a mixture of PEG-400 and water at room temperature or 50 °C to afford a variety of phenyl-substituted alkenes in moderate to high yields. The isolation of the products was easily performed by extraction with petroleum ether, and the PdCl2(PPh3)2/PEG-400/H2O system could be readily recycled and reused six times without apparent loss of catalytic activity.
Introduction
Styrenes are important structural motifs extensively presented in pharmaceuticals, natural or synthetic products, and functional materials.1–5 Traditionally, styrenes are prepared by the Wittig 6 or Peterson olefinations 7 and insertions of alkynes into organometallic reagents,8–10 which are waste-intensive and require prefunctionalized starting materials. Catalytic methods such as the olefin metathesis11,12 and Heck coupling reactions 13 are more atom-economic but also employ prefunctionalized arenes as substrates. In addition, C–H vinylations of the Fujiwara–Moritani type have provided an efficient route to styrenes, but stoichiometric oxidants are required.14–16
In recent years, catalytic hydroarylation of alkynes has attracted considerable interest because it allows for the atom-economical construction of functionalized alkenes from relatively simple arenes and alkynes.17–19 The hydroarylations of alkynes catalyzed by transition metal complex or Lewis acid through C–H bond activation of arenes have been widely investigated, but most research has mainly focused on the electron-rich arenes, and the regiocontrol of the reaction is still difficult, thereby providing a mixture of alkene derivatives.20–23 Recently, the employment of arylboron compounds in hydroarylation reactions drew considerable attention because of their ready availability, stability, and high functional group compatibility.24–28 Hayashi et al. 29 reported hydroarylation of alkynes with arylboronic acids, and the reaction has been expanded to the arylative cyclization of aldehydes and alkyne-tethered alkenes.30,31 Sodium tetraphenylborate is a stable and inexpensive phenylating reagent and has been widely used in organic synthesis. Palladium-catalyzed hydrophenylation of alkenes 32 and alkynes 33 with sodium tetraphenylborate has been reported to proceed smoothly under mild conditions. However, the hydrophenylation reaction generally proceeds in the presence of a homogeneous palladium catalyst such as Pd(OAc)2 or PdCl2(PPh3)2, which makes the recovery of the expensive palladium catalyst tedious if not impossible and might cause unacceptable palladium contamination of the product. Therefore, from the standpoint of green and sustainable chemistry, the development of recyclable and reusable palladium catalytic systems for these important organic transformations is highly desirable.
As green organic synthesis is attracting more and more attention, the use of eco-friendly, green solvents is highly desirable. To address the recyclability of metal catalysts and environmental concerns, a convenient and efficient way is to anchor the catalyst in a liquid phase by dissolving it into a nonvolatile and nonmixing liquid, such as poly(ethylene glycols) (PEGs)34–36 and ionic liquids. 37 Generally, ionic liquids require a complicated preparative procedure, and their environmental safety is still being debated because the toxicity and environmental burden data are unknown for most of the ionic liquids. By contrast, PEGs are easily available and cheap, thermally stable, biodegradable, recoverable, and nontoxic liquid polymers which can be used as efficient media for eco-friendly and safe chemical reactions. Recently, PEGs have been widely utilized as green solvents for the palladium-catalyzed carbon–carbon bond-forming reactions such as the Heck coupling, 38 the Suzuki coupling,39–41 the homo-coupling and cross-coupling of aryl halides, 42 the direct arylation of 1,2,3-triazoles with aryl bromides, 43 the Hiyama coupling, 44 carbonylative Suzuki coupling, 45 carbonylative Sonogashira coupling, 46 and the homo-coupling of arylboronic acids 47 with easy recyclability of the solvents and Pd catalysts. However, to the best of our knowledge, no palladium-catalyzed hydrophenylation reaction of alkenes or alkynes with sodium tetraphenylborate in PEGs has been described until now. Herein, we report the application of PdCl2(PPh3)2/PEG-400/H2O system as a highly efficient and reusable catalytic medium for the hydrophenylation of alkynes with sodium tetraphenylborate. This affords a variety of phenyl-substituted alkenes in moderate to high yields. The developed methodology shows important practical advantages deserving special note.
Results and discussion
In our initial screening experiments, the hydrophenylation of 1,2-diphenylacetylene (
Reaction of 1,2-diphenylacetylene (
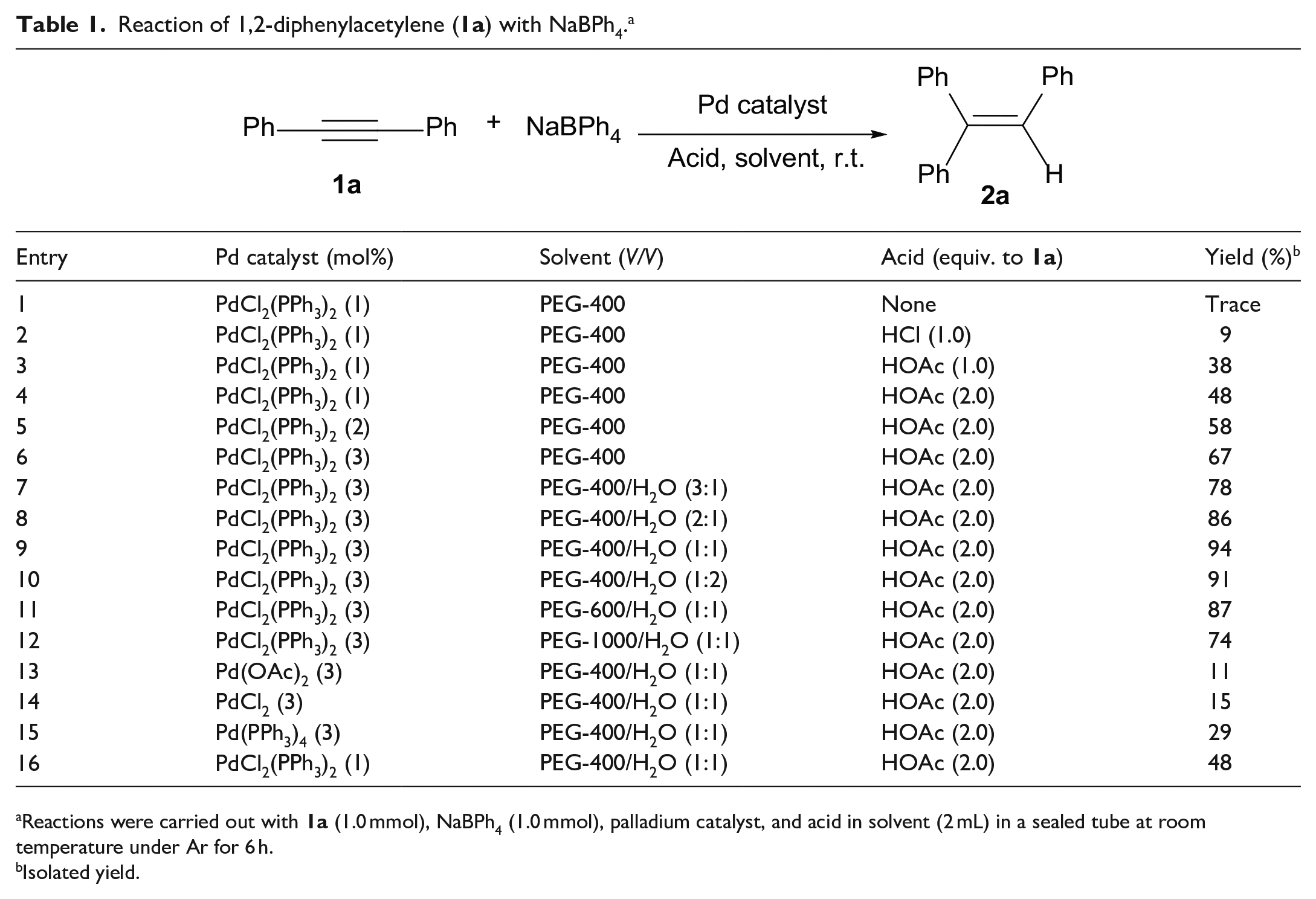
Reactions were carried out with
Isolated yield.
Having achieved satisfactory results in the hydrophenylation of 1,2-diphenylacetylene with NaBPh4, then, various internal and terminal alkynes were examined to explore the scope of substrates under the optimized reaction conditions and the results are listed in Table 2. As expected, symmetrical internal alkynes
Reactions were carried out with alkyne (1.0 mmol), NaBPh4 (1.0 mmol), HOAc (2.0 mmol), and PdCl2(PPh3)2 (0.03 mmol) in a mixture of PEG-400 (1 mL) and H2O (1 mL) in a sealed tube at room temperature under Ar for 6 h.
Isolated yield.
Determined by GC.
Sodium tetraarylborates are usually prepared by the reaction of arylmagnesium bromides with trimethyl borate, followed by hydrolysis. To explore the scope of the borates, we prepared sodium tetrakis(4-methylphenyl)borate via a complicated procedure starting from 4-bromotoluene, magnesium turnings, and trimethyl borate. The reaction of ethyl but-2-ynoate
To explore the possibility for further efficient transfer of phenyl groups in NaBPh4, we examined the reaction of
Reactions were carried out with alkyne (1.0 mmol), NaBPh4 (0.33 mmol), HOAc (2.0 mmol), and PdCl2(PPh3)2 (0.03 mmol) in a mixture of PEG-400 (1 mL) and H2O (1 mL) in a sealed tube at 50 °C under Ar for 12 h.
Isolated yield.
Determined by GC.
It was reported that the reaction of NaBPh4 with HOAc could produce Ph3B, benzene, and NaOAc, and Ph3B could react with H2O further to form di- and monophenylboronic acid.
32
Thus, we also studied the reactivity of arylboronic acids with alkynes at room temperature (Scheme 1). As shown in Scheme 1, the palladium-catalyzed hydroarylation reaction of internal alkynes with various arylboronic acids also took place smoothly at room temperature in PEG-400/H2O (1:1) to give the corresponding trisubstituted aryl alkenes in moderate yields. We next carried out the reaction of 1,2-diphenylacetylene

Palladium-catalyzed hydroarylation reaction of alkynes with arylboronic acids in PEG-400/H2O (1:1).
A possible mechanism for the palladium-catalyzed hydrophenylation of alkynes with NaBPh4 is illustrated in Scheme 2. First, oxidative addition of the C–B bond of Ph3B formed in situ from reaction of NaBPh4 with HOAc to (Ph3P)2Pd(0) produces intermediate

Proposed catalytic cycle.
To evaluate the reusability of the solvent and the catalyst, the hydrophenylation reaction of 1,2-diphenylacetylene (1 mmol) with NaBPh4 (1 mmol) was investigated in the presence of PdCl2(PPh3)2 (3 mol%) and HOAc (2.0 equiv.) in PEG-400/H2O (V/V = 1:1, 2 mL) at room temperature. As shown in Table 4, we were pleased to observe that the PdCl2(PPh3)2/PEG-400/H2O system could be recycled and reused up to six times without apparent loss of catalytic activity. After initial experimentation, the reaction mixture was extracted with petroleum ether (3 × 10 mL), and the PdCl2(PPh3)2/PEG-400/H2O system was then subjected to a second run of the reaction by charging with the same starting materials (1,2-diphenylacetylene, NaBPh4, and HOAc) without addition of PdCl2(PPh3)2. In addition, the leaching of palladium in the product was also determined, and inductively coupled plasma (ICP) analysis of the crude petroleum ether extract after the first cycle showed that palladium content was less than 0.80 ppm.
Recyclability of PdCl2(PPh3)2/PEG-400/H2O system. a

Reaction conditions:
Isolated yield.
In conclusion, an efficient and recyclable catalytic system for the palladium-catalyzed hydrophenylation reaction of alkynes with sodium tetraphenylborate has been developed. In the presence of 3 mol% PdCl2(PPh3)2 and HOAc, the hydrophenylation reaction of a variety of alkynes with NaBPh4 proceeded smoothly at room temperature in PEG-400/H2O (1:1) to afford the corresponding phenyl-substituted alkenes in moderate to high yields. Furthermore, the PdCl2(PPh3)2/PEG-400/H2O system could be recycled up to six times without apparent loss of catalytic activity. The present protocol will serve as an efficient and green way to prepare a variety of phenyl-substituted alkenes. Currently, further efforts to extend the application of the system in other palladium-catalyzed organic transformations are underway in our laboratory.
Experimental
All reagents were purchased from different commercial sources and used as received without further purification unless otherwise indicated. All reactions were carried out under Ar in a sealed reaction tube with magnetic stirring and the solvents needed to be de-gassed. 1H NMR spectra were recorded on a Bruker Avance 400 (400 MHz) spectrometer with tetramethylsilane (TMS) as an internal standard using CDCl3 as the solvent. 13C NMR spectra were recorded on a Bruker Avance 400 (100 MHz) spectrometer using CDCl3 as the solvent. HRMS spectra were recorded on a quadrupole–time of flight Bruker MicroTOF-Q II mass spectrometer equipped with an electrospray ionization (ESI) and atmospheric pressure chemical ionization (APCI) source. Palladium content was determined with inductively coupled plasma atom emission Atomscan16 (ICP-AES, TJA Corporation).
General procedure for palladium-catalyzed hydrophenylation of alkynes with NaBPh4 in PEG-400/H2O (1:1)
A mixture of alkyne
The residue (the aqueous layer) of the extraction was heated to 60 °C in vacuum for 20 min to remove the residual petroleum ether, and then subjected to a second run of the reaction by charging with the same starting materials (alkyne, NaBPh4, and HOAc) without addition of PdCl2(PPh3)2 under identical conditions. After the first reaction cycle, the crude petroleum ether extract was subjected to ICP-AES analysis before any further purification.
1,1,2-Triphenylethene (
(E)-3-phenylhex-3-ene (
(E)-4-phenyloct-4-ene (
(E)-5-phenyldec-5-ene (
(Z)-dimethyl 2-phenylmaleate (
(E)-methyl 3-phenylbut-2-enoate (
(E)-ethyl 3-phenylbut-2-enoate (
(E)-ethyl 3-phenyloct-2-enoate (
Ethyl 3,3-diphenylacrylate (
(E)-1,2-diphenylpropene (
1,1-Diphenylpropene (
2-Phenyl-1-hexene (
2-Phenyl-1-octene (
1,1-Diphenylethene (
2,5-Diphenyl-1-pentene (
5-Chloro-2-phenyl-1-pentene (
5-Cyano-2-phenyl-1-pentene (
General procedure for palladium-catalyzed hydroarylation of alkynes with ArB(OH)2 in PEG-400/H2O (1:1)
A mixture of alkyne
(Z)-dimethyl 2-p-tolylmaleate (
(E)-ethyl 3-p-tolylbut-2-enoate (
(E)-ethyl 3-(4-methoxyphenyl)but-2-enoate (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the National Natural Science Foundation of China (no. 21462021) and the Natural Science Foundation of Jiangxi Province of China (no. 20161BAB203086).








