Abstract
Two new isostructural lanthanide(III) coordination polymers based on an unreported zwitterionic ligand, namely, [Ln(ox)(L)]n (ox = oxalate, HL = N,N'-dipropionic acid imidazolium, Ln = Eu or La), are synthesized under hydrothermal conditions and characterized by single-crystal X-ray diffraction, infrared spectroscopy, powder X-ray diffraction, and thermogravimetric analysis. The fluorescence properties of the europium coordination polymer are investigated. In addition, the europium-based coordination polymer is utilized for specific sensing of UO22+ ions, showing high selectivity, a fast response time (8 min) and high sensitivity with noticeable quenching (Ksv = 6.19 × 104 M−1) and limit of detection of 1.95 µM.
Two new LnCPs were synthesized under hydrothermal conditions and were characterized adequately. The LnCP

Introduction
Lanthanide coordination polymers (LnCPs, sometimes including lanthanide organic frameworks (LnOFs)) have attracted considerable attention from a scientific and technological point of view due to their distinctive optical, electronic, and magnetic properties.1–3 Many lanthanide ions, particularly Eu(III) and Tb(III) ions, can act as the emissive centers to afford types of phosphors, 4 which have opened many opportunities in growing fields of social and economic impact, for example, as luminescent materials, in medical diagnostics, in environmental sciences and in cell biology.5–8 They have also been extensively used in many practical fields such as lighting and displays. Luminescent lanthanide ions or LnCPs possess several virtues, such as strong emission bands for f–f transitions generated via the “antenna effect”, 9 wide spectral range, broad range lifetimes, high quantum yields, and so on. 10 Due to their unique luminescence properties, LnCPs have potential applications as fluorescent probes, which have been extensively studied for selectively probing nitro explosives, toxic organic solvents, and heavy-metal ions.11–13
Well known as the key raw material in the nuclear energy industry and other important industrial applications, radioactive and chemically toxic uranium mainly exists in its hexavalent state with very high solubility in solution and is widely distributed in different environments such as various forms of water, air, dust, soil, and rock.14,15 The chemical toxicity and radioactivity of uranium poses a significant threat to biological systems, leading to irreversible damage.16,17 Therefore, it is vital to develop effective, quick, and simple analytical methods for detecting UO22+ in different samples.
There are a few reports describing the use of LnCPs to detect or uptake UO22+ at low concentrations and with high sorption capacity and high sensitivity by fluorescence quenching.18–23 One of the most important features for sensing uranyl cations is the abundance of open Lewis basic sites.
18
Encouraged by these earlier reports, we have synthesized two new LnCPs based on a novel zwitterionic N,N'-dipropionic acid imidazolium ligand as a more flexible organic linker, together with oxalic acid under hydrothermal conditions. The products, LnCPs, [Ln(ox)(L)]n (Ln = Eu,
Results and discussion
The crystal structures of complexes 1 and 2
The crystallographic data are shown in Table 1 for
Crystallographic data for

a R1 = ∑||Fo|―|Fc||/∑|Fo|, b wR2 = {∑[w(Fo2―Fc2)2]/∑[w(Fo2)2]}1/2.

(a) The coordination environment of the Ln ion, with displacement ellipsoids at the 50% probability level. [Symmetry codes: (i) x, 0.5-y, -0.5+z; (ii) 1-x, 1-y, -z; (iii) x, y, -1+z; (iv) 1-x, 1-y, 1-z.] (b) The binuclear [Ln2(ox)4(COO)4] SBU of [Ln(ox)(L)]n. (c) The 2D structure of [Ln(ox)(L)]n along the bc plane. H atoms have been omitted for clarity.
IR spectra
From Supplemental Figure S1, it is found that in the IR spectra of zwitterionic ligand HL (HL = N,N'-dipropionic acid imidazolium),
Powder X-ray diffraction (PXRD)
The PXRD patterns for
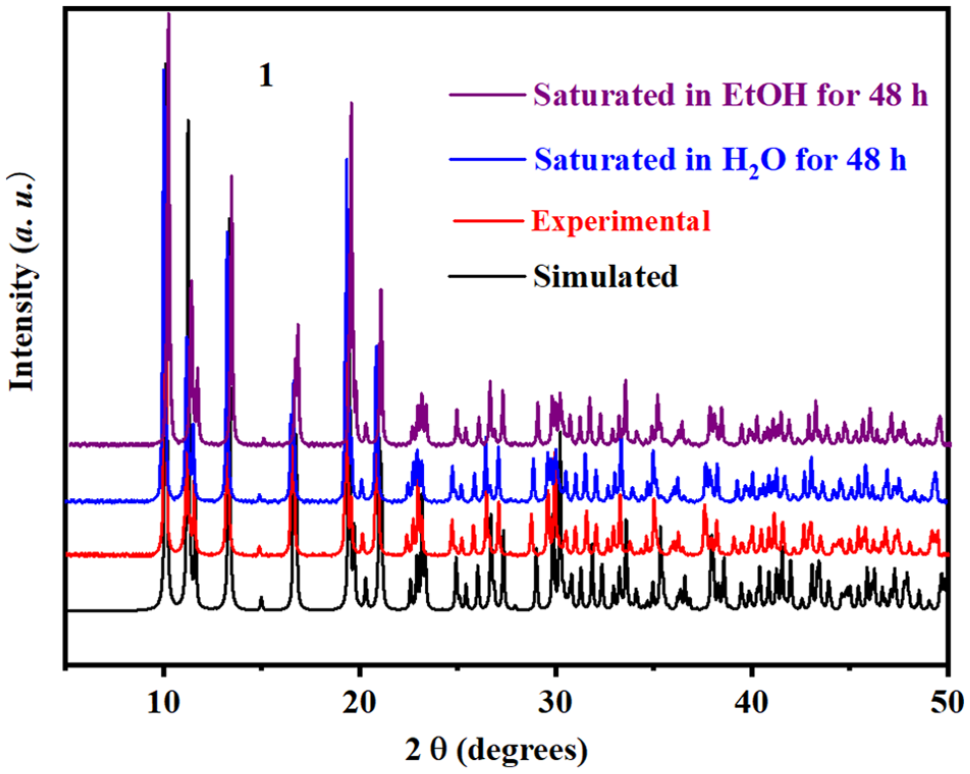
The experimental, simulated, saturated in H2O for 48 h and saturated in EtOH for 48 h PXRD patterns for LnCP
Thermogravimetric analysis (TGA)
Because LnCPs
Fluorescence properties
The fluorescence spectrum of

The fluorescence spectrum of
Fluorescence probing studies
To investigate the influence of different metal ions on the fluorescence of

(a) Bar chart obtained for LnCP
Conclusion
Two new LnCPs were synthesized under hydrothermal conditions using an unreported zwitterionic ligand and oxalate as co-ligands. The two title compounds were characterized adequately with highly thermal stability. Single-crystal measurement revealed that the two LnCPs are 2D isostructural constructed by [Ln2(ox)4(COO)4] SBUs. The LnCP
Experimental
Materials and methods
All of the reagents are commercially available and were used without further purification. All of the metal salts used are nitrate salts. Other reagents and solvents were purchased from commercial sources and were used without further purification. Elemental analyses (C, H and N) were carried out using a Vario EL III elemental analyzer. The IR spectrum was recorded as a KBr pellet in the range 4000–400 cm−1 on a Smart Omni-Transmission spectrometer. PXRD data were recorded on a Philips X-Pert-MPD diffractometer with CuKα (λ = 1.5406 Å) radiation in the 2θ range (5–50o) at a rate of 5o min−1. Thermogravimetric analysis was carried out on a Mettler TGA/SDTA 851 thermal analyzer in the temperature range 298–1073 K under an N2 flow with a heating rate of 10 K min−1. The fluorescence spectrum of
Synthesis of ligand (HL)
The new zwitterionic ligand HL (Supplemental Scheme S1) was synthesized according to the reference with slight modification. 29 At room temperature, 3-aminopropanoic acid (8.9 g, 100 mmol) was mixed with 40% glyoxal (7.32 g, 50 mmol, w/w in water) and stirred under air. After 15 min, the reaction solution became cloudy and dark brown. After stirring for 2 h, the mixture was filtered, and washed with ice-cold water (3 × 15 mL). Following drying under vacuum, a yellow-brown powder was obtained, yield 56.8% (based on γ-aminobutyric acid), which was characterized by IR spectroscopy (Supplemental Figure S1).
Synthesis of [Ln(ox)(L)]n (1 and 2 )
A mixture of Ln(NO3)3·6H2O (1 mmol, Ln = Eu, 446 mg,
Single-crystal structure determination
The X-ray diffraction data for
Selected bond lengths (Å) for compounds

Symmetry codes:
Fluorescence probing measurement
Finely ground crystalline material of
Supplemental Material
CaiB – Supplemental material for Exploiting a new europium(III) coordination polymer based on a zwitterionic ligand as a fluorescent probe for uranyl cations
Supplemental material, CaiB for Exploiting a new europium(III) coordination polymer based on a zwitterionic ligand as a fluorescent probe for uranyl cations by Bin Cai, Yu-Ning Meng, Meng-En Zhu and Youming Yang in Journal of Chemical Research
Supplemental Material
checkcif – Supplemental material for Exploiting a new europium(III) coordination polymer based on a zwitterionic ligand as a fluorescent probe for uranyl cations
Supplemental material, checkcif for Exploiting a new europium(III) coordination polymer based on a zwitterionic ligand as a fluorescent probe for uranyl cations by Bin Cai, Yu-Ning Meng, Meng-En Zhu and Youming Yang in Journal of Chemical Research
Supplemental Material
Supporting_Information_3 – Supplemental material for Exploiting a new europium(III) coordination polymer based on a zwitterionic ligand as a fluorescent probe for uranyl cations
Supplemental material, Supporting_Information_3 for Exploiting a new europium(III) coordination polymer based on a zwitterionic ligand as a fluorescent probe for uranyl cations by Bin Cai, Yu-Ning Meng, Meng-En Zhu and Youming Yang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Key Scientific and Technological Research Projects in Henan Province (192102210028) and the National Natural Science Foundation of China (51602358, 51564017, and 51774155).
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
