Experimental analysis
Reagents and solvents were purchased from commercial suppliers unless otherwise specified. All reactions were carried out under an air atmosphere. Anhydrous solvents were purified and dried following standard procedures. Purification was generally done by flash column chromatography on brand silica gel (200–300 mesh size). Thin-layer chromatography (TLC) analysis was performed on brand precoated, glass-backed silica gel plates. Nuclear magnetic resonance (NMR) spectra were recorded on a 400-MHz Bruker spectrometer (400 MHz for 1H NMR, 100 MHz for 13C NMR). Chemical shifts (1H and 13C) are given in ppm relative to the residual solvent peak (CDCl3, 7.26 ppm, 77.0 ppm, respectively). High-resolution mass spectra (HRMS) were obtained on a Thermo Fisher LC-LTQ-Orbitrap XL spectrometer. For more information about chemical spectra, please see the supplemental material.
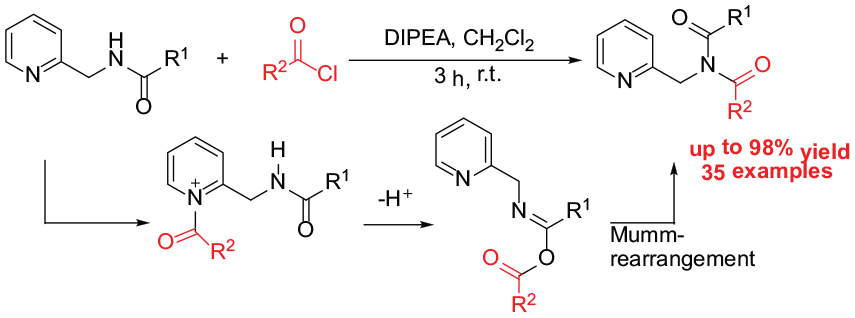
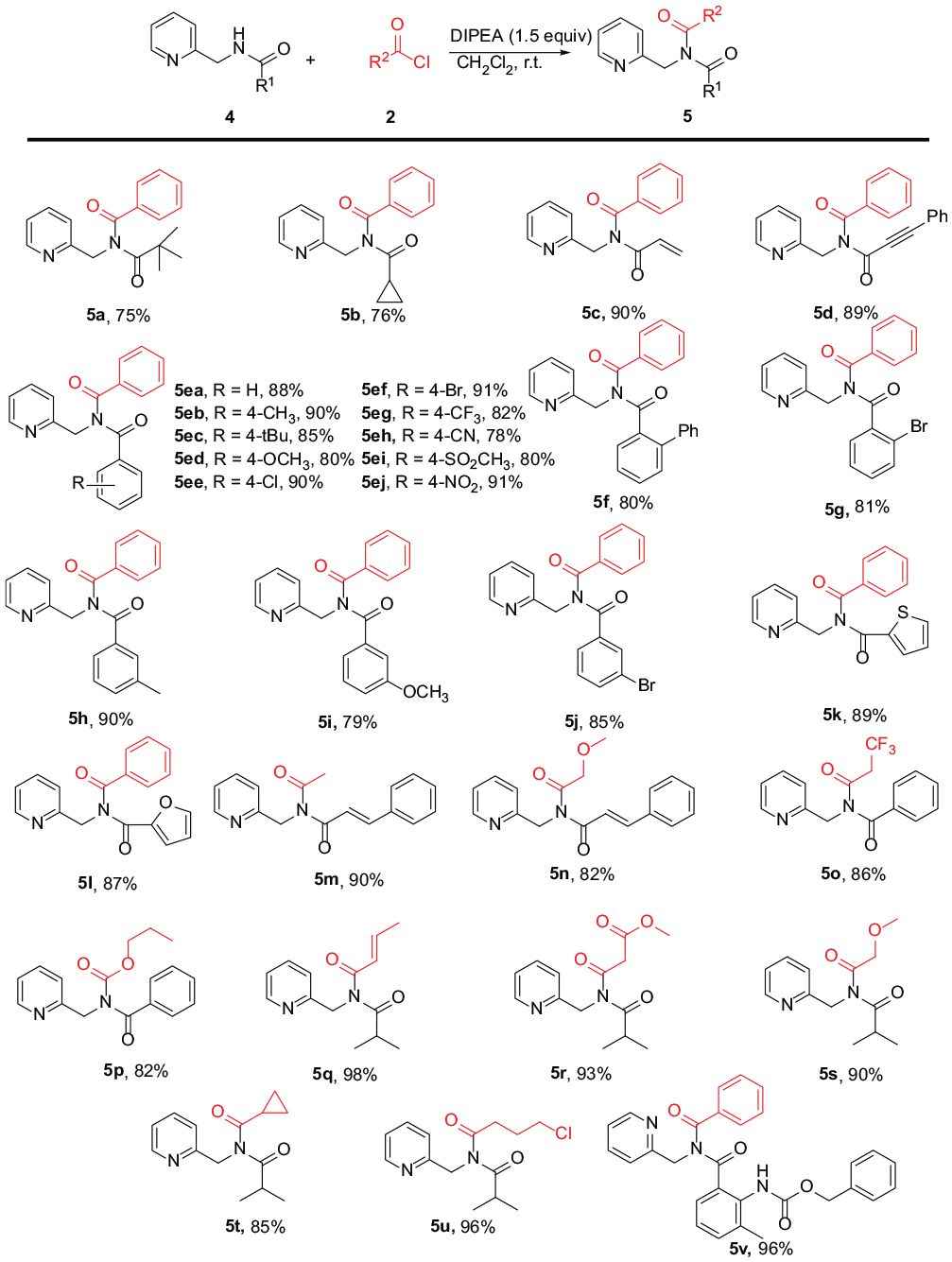
General procedure for the synthesis of imides 5. To a mixture of amide 4 (0.2 mmol) and DIPEA (0.3 mmol) in CH2Cl2 (2 mL) was added acyl chloride 2 (0.26 mmol) dropwise at 0 °C. The reaction mixture was stirred at room temperature for 3 h. After completion of the reaction, the mixture was poured into water (20 mL) and extracted with CH2Cl2 (3 × 20 mL). The combined organic layers were dried with anhydrous Na2SO4. After removal of the solvents in vacuo, the obtained crude product was further purified by column chromatography on silica gel, eluting with a mixture of petroleum and ethyl acetate (5:1) to give the desired products.
N-Acetyl-N-(pyridin-2-ylmethyl)benzamide (3a): colorless oil; yield: 47.8 mg, (94%). 1H NMR (400 MHz, CDCl3): δ 8.51 (d, J = 4.7 Hz, 1H), 7.70 (d, J = 8.1 Hz, 2H), 7.62 (t, J = 7.1 Hz, 1H), 7.52 (t, J = 7.4 Hz, 1H), 7.42 (t, J = 7.6 Hz, 2H), 7.22 (d, J = 7.8 Hz, 1H), 7.17–7.11 (m, 1H), 5.09 (s, 2H), 2.26 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.3, 173.5, 156.5, 149.2, 136.6, 135.7, 132.2, 128.6, 128.5, 122.1, 121.4, 50.8, 26.2; HRMS (ESI+): m/z [M + Na]+ calcd for C15H14N2O2Na: 277.0947; found: 277.0944.
N-Acetyl-N-(thiophen-2-ylmethyl)benzamide (3b): colorless oil; yield: 33.7 mg, (65%). 1H NMR (400 MHz, CDCl3): δ 7.60–7.53 (m, 3H), 7.45 (t, J = 7.6 Hz, 2H), 7.21 (dd, J = 5.0, 1.4 Hz, 1H), 6.93–6.87 (m, 2H), 5.15 (s, 2H), 2.10 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 173.9, 173.0, 139.3, 135.7, 132.6, 128.9, 128.5, 127.3, 126.5, 125.8, 44.0, 26.5; HRMS (ESI+): m/z [M + Na]+ calcd for C14H13NO2SNa: 282.0559; found: 282.0569.
N-Acetyl-N-(furan-2-ylmethyl)benzamide (3c): colorless oil; yield: 33.5 mg, (69%). 1H NMR (400 MHz, CDCl3): δ 7.59 (d, J = 8.0 Hz, 2H), 7.54 (t, J = 7.4 Hz, 1H), 7.44 (t, J = 7.6 Hz, 2H), 7.29 (s, 1H), 6.27 (s, 1H), 6.20 (d, J = 3.1 Hz, 1H), 4.96 (s, 2H), 2.17 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.0, 172.8, 150.4, 142.1, 135.5, 132.5, 128.8, 128.4, 110.4, 108.4, 42.3, 26.1; HRMS (ESI+): m/z [M + Na]+ calcd for C14H13NO3Na: 266.0788; found: 266.0782.
N-Acetyl-N-(2-(pyridin-2-yl)ethyl)benzamide (3d): colorless oil; yield: 36.5 mg, (68%). 1H NMR (400 MHz, CDCl3): δ 8.43 (d, J = 4.2 Hz, 1H), 7.57 (td, J = 7.65,1.32 Hz, 1H), 7.51–7.47 (m, 3H), 7.40 (t, J = 7.6 Hz, 2H), 7.15–7.07 (m, 2H), 4.19 (t, J = 6.9 Hz, 2H), 3.11 (t, J = 6.9 Hz, 2H), 2.15 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.4, 173.3, 158.5, 149.1, 136.6, 135.4, 132.3, 128.7, 128.5, 123.8, 121.6, 46.3, 37.1, 25.9; HRMS (ESI+): m/z [M + Na]+ calcd for C16H16N2O2Na: 291.1104; found: 291.1121.
N-Pivaloyl-N-(pyridin-2-ylmethyl)benzamide (5a): colorless oil; yield: 44.4 mg, (75%). 1H NMR (400 MHz, CDCl3): δ 8.54 (d, J = 4.7 Hz, 1H), 7.80 (d, J = 7.1 Hz, 2H), 7.62 (t, J = 6.9 Hz, 1H), 7.51 (t, J = 6.7 Hz, 1H), 7.41 (t, J = 7.4 Hz, 2H), 7.22–7.12 (m, 2H), 4.92 (s, 2H), 1.31 (s, 9H); 13C NMR (100 MHz, CDCl3): δ 187.4, 174.8, 156.5, 149.2, 136.6, 134.6, 132.3, 129.0, 128.6, 122.2, 121.6, 52.6, 43.4, 28.5; HRMS (ESI+): m/z [M + Na]+ calcd for C18H20N2O2Na: 319.1417; found: 319.1424.
N-(Cyclopropanecarbonyl)-N-(pyridin-2-ylmethyl)benzamide (5b): colorless oil; yield: 42.6 mg, (76%). 1H NMR (400 MHz, CDCl3): δ 8.53 (d, J = 4.4 Hz, 1H), 7.80–7.75 (m, 2H), 7.64 (td, J = 7.7, 1.7 Hz, 1H), 7.52 (t, J = 7.4 Hz, 1H), 7.43 (t, J = 7.5 Hz, 2H), 7.28 (d, J = 7.85, 1H), 7.15 (dd, J = 7.2, 5.2 Hz, 1H), 5.21 (s, 2H), 1.59 (ddd, J = 12.4, 7.8, 4.6 Hz, 1H), 1.07–1.00 (m, 2H), 0.65 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 177.9, 173.9, 156.9, 149.3, 136.6, 136.2, 132.2, 129.0, 128.5, 122.1, 121.4, 50.7, 18.3, 11.7; HRMS (ESI+): m/z [M + Na]+ calcd for C17H16N2O2Na: 303.1104; found: 303.1115.
N-Acryloyl-N-(pyridin-2-ylmethyl)benzamide (5c): colorless oil; yield: 47.9 mg, (90%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.7 Hz, 1H), 7.72 (d, J = 8.2 Hz, 2H), 7.64 (t, J = 7.7 Hz, 1H), 7.53 (t, J = 7.5 Hz, 1H), 7.41 (t, J = 7.6 Hz, 2H), 7.29 (d, J = 7.8 Hz, 1H), 7.18–7.12 (m, 1H), 6.31–6.17 (m, 2H), 5.51 (dd, J = 9.1, 2.7 Hz, 1H), 5.21 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 173.7, 169.2, 156.4, 149.3, 136.6, 135.8, 132.5, 130.8, 129.1, 128.8, 128.6, 122.2, 121.5, 50.6; HRMS (ESI+): m/z [M + Na]+ calcd for C16H14N2O2Na: 289.0947; found: 289.0951.
N-(3-Phenylpropioloyl)-N-(pyridin-2-ylmethyl)benzamide (5d): colorless oil; yield: 60.5 mg, (89%). 1H NMR (400 MHz, CDCl3): δ 8.55 (d, J = 4.9 Hz, 1H), 7.88 (d, J = 7.5 Hz, 2H), 7.67 (t, J = 7.7 Hz, 1H), 7.53 (t, J = 7.4 Hz, 1H), 7.45 (t, J = 7.5 Hz, 2H), 7.35 (t, J = 7.8 Hz, 2H), 7.24 (m, 2H), 7.21–7.13 (m, 1H), 7.09 (d, J = 7.7 Hz, 2H), 5.34 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 173.4, 156.1, 155.0, 149.5, 136.6, 136.0, 132.7, 132.6, 130.6, 129.7, 128.5, 128.3, 122.3, 121.5, 119.5, 95.9, 82.7, 49.9; HRMS (ESI+): m/z [M + Na]+ calcd for C22H16N2O2Na: 363.1104; found: 363.1113.
N-Benzoyl-N-(pyridin-2-ylmethyl)benzamide (5ea): colorless oil; yield: 55.6 mg, (88%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.7 Hz, 1H), 7.66 (td, J = 7.7, 1.4 Hz, 1H), 7.54 (d, J = 7.3 Hz, 4H), 7.39 (d, J = 7.8 Hz, 1H), 7.25 (t, J = 7.20, 3H), 7.14 (t, J = 7.5 Hz, 5H), 5.34 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 174.2, 156.6, 149.3, 136.6, 136.5, 131.7, 129.1, 128.1, 122.3, 121.8, 51.4; HRMS (ESI+): m/z [M + Na]+ calcd for C20H16N2O2Na: 339.1104; found: 311.1111.
N-Benzoyl-4-methyl-N-(pyridin-2-ylmethyl)benzamide (5eb): colorless oil; yield: 59.3 mg, (90%). 1H NMR (400 MHz, CDCl3): δ 8.53 (d, J = 4.9 Hz, 1H), 7.67 (t, J = 7.7 Hz, 1H), 7.56 (d, J = 7.4 Hz, 2H), 7.47 (d, J = 7.9 Hz, 2H), 7.40 (d, J = 7.8 Hz, 1H), 7.25 (d, J = 7.0 Hz, 1H), 7.17 (t, J = 7.9 Hz, 3H), 6.96 (d, J = 7.9 Hz, 2H), 5.34 (s, 2H), 2.23 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.3, 174.2, 156.7, 149.3, 142.5, 136.6, 136.5, 133.6, 131.6, 129.3, 129.0, 128.8, 128.0, 122.2, 121.8, 51.6, 21.4; HRMS (ESI+): m/z [M + Na]+ calcd for C21H18N2O2Na: 353.1260; found: 353.1262.
N-Benzoyl-4-(tert-butyl)-N-(pyridin-2-ylmethyl)benzamide (5ec): colorless oil; yield: 63.3 mg, (85%). 1H NMR (400 MHz, CDCl3): δ 8.54 (d, J = 4.4 Hz, 1H), 7.67 (t, J = 7.7 Hz, 1H), 7.53 (d, J = 7.5 Hz, 2H), 7.49 (d, J = 8.2 Hz, 2H), 7.40 (d, J = 7.8 Hz, 1H), 7.22 (t, J = 7.2 Hz, 1H), 7.17–7.12 (m, 5H), 5.35 (s, 2H), 1.19 (s, 9H); 13C NMR (100 MHz, CDCl3): δ 174.4, 174.2, 156.7, 155.3, 149.4, 136.6, 133.5, 131.4, 129.1, 129.0, 128.0, 125.0, 122.2, 121.8, 51.4, 34.9, 30.9; HRMS (ESI+): m/z [M + Na]+ calcd for C24H24N2O2Na: 395.1730; found: 395.1725.
N-Benzoyl-4-methoxy-N-(pyridin-2-ylmethyl)benzamide (5ed): colorless oil; yield: 55.4 mg, (80%). 1H NMR (400 MHz, CDCl3): δ 8.59 (d, J = 4.9 Hz, 1H), 7.72 (t, J = 7.6 Hz, 1H), 7.63 (dd, J = 8.4, 2.5 Hz, 4H), 7.46 (d, J = 7.8 Hz, 1H), 7.33 (t, J = 7.2 Hz, 1H), 7.23 (q, J = 7.7 Hz, 3H), 6.73 (d, J = 8.6 Hz, 2H), 5.40 (s, 2H), 3.79 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.1, 173.7, 162.4, 156.7, 149.3, 136.5, 131.5, 131.4, 128.9, 128.6, 128.1, 122.1, 121.7, 113.4, 55.3, 51.6; HRMS (ESI+): m/z [M + Na]+ calcd for C21H18N2O3Na: 369.1210; found: 369.1218.
N-Benzoyl-4-chloro-N-(pyridin-2-ylmethyl)benzamide (5ee): colorless oil; yield: 63.0 mg, (90%). 1H NMR (400 MHz, CDCl3): δ 8.51 (d, J = 4.9 Hz, 1H), 7.67 (t, J = 7.7 Hz, 1H), 7.56 (d, J = 7.6 Hz, 2H), 7.52 (d, J = 8.3 Hz, 2H), 7.37 (d, J = 7.9 Hz, 1H), 7.30 (t, J = 7.4 Hz, 1H), 7.22–7.11 (m, 5H), 5.33 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 174.0, 173.3, 156.3, 149.4, 137.8, 136.6, 136.3, 135.0, 132.0, 130.4, 129.1, 128.4, 128.3, 122.3, 121.8, 51.4; HRMS (ESI+): m/z [M + Na]+ calcd for C20H15ClN2O2Na: 373.0714; found: 373.0712.
N-Benzoyl-4-bromo-N-(pyridin-2-ylmethyl)benzamide (5ef): colorless oil; yield: 71.7 mg, (91%). 1H NMR (400 MHz, CDCl3): δ 8.51 (d, J = 4.4 Hz, 1H), 7.67 (td, J = 7.7, 1.8 Hz, 1H), 7.58–7.53 (m, 2H), 7.47–7.42 (m, 2H), 7.37 (d, J = 7.8 Hz, 1H), 7.30 (m, 3H), 7.23–7.14 (m, 3H), 5.33 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 174.0, 173.4, 156.2, 149.4, 136.5, 136.2, 135.4, 132.0, 131.3, 130.5, 129.1, 128.3, 126.4, 122.3, 121.7, 51.4; HRMS (ESI+): m/z [M + Na]+ calcd for C20H15BrN2O2Na: 417.0209; found: 417.0208.
N-Benzoyl-N-(pyridin-2-ylmethyl)-4-(trifluoromethyl)benzamide (5eg): colorless oil; yield: 63.0 mg, (82%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.9 Hz, 1H), 7.69 (d, J = 7.8 Hz, 3H), 7.56 (d, J = 7.5 Hz, 2H), 7.43 (d, J = 7.7 Hz, 2H), 7.38 (d, J = 7.6 Hz, 1H), 7.33–7.25 (m, 1H), 7.19 (t, J = 6.9 Hz, 3H), 5.35 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 174.0, 173.0, 156.0, 149.4, 139.9, 136.6, 136.0, 132.8 (q, J = 32.0 Hz), 132.09, 129.2, 129.1, 128.3, 125.3 (q, J = 4.0 Hz), 123.4 (d, J = 271.0 Hz), 122.4, 121.8, 51.2; HRMS (ESI+): m/z [M + Na]+ calcd for C21H15F3N2O2Na: 407.0978; found: 407.0970.
N-Benzoyl-4-cyano-N-(pyridin-2-ylmethyl)benzamide (5eh): colorless oil; yield: 53.2 mg, (78%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.7 Hz, 1H), 7.72–7.65 (m, 3H), 7.58–7.53 (m, 2H), 7.46 (d, J = 8.3 Hz, 2H), 7.37 (d, J = 7.8 Hz, 1H), 7.32 (t, J = 7.4 Hz, 1H), 7.23–7.17 (m, 3H), 5.35 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 173.7, 172.5, 155.7, 149.3, 140.6, 136.8, 135.8, 132.3, 131.8, 129.3, 129.1, 128.5, 122.5, 121.8, 117.8, 114.6, 51.1; HRMS (ESI+): m/z [M + Na]+ calcd for C21H15N3O2Na: 364.1056; found: 364.1060.
N-Benzoyl-4-(methylsulfonyl)-N-(pyridin-2-ylmethyl)benzamide (5ei): colorless oil; yield: 63.1 mg, (80%). 1H NMR (400 MHz, CDCl3): δ 8.53 (d, J = 4.7 Hz, 1H), 7.79–7.67 (m, 5H), 7.57 (d, J = 7.3 Hz, 2H), 7.38 (d, J = 7.8 Hz, 1H), 7.31 (t, J = 7.4 Hz, 1H), 7.20 (t, J = 7.8 Hz, 3H), 5.36 (s, 2H), 2.93 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 173.8, 172.5, 155.7, 149.3, 142.4, 141.5, 136.8, 135.9, 132.3, 129.7, 129.2, 128.4, 127.2, 122.5, 121.8, 51.1, 44.2; HRMS (ESI+): m/z [M + Na]+ calcd for C21H18N2O4SNa: 417.0879; found: 417.0881.
N-Benzoyl-4-nitro-N-(pyridin-2-ylmethyl)benzamide (5ej): colorless oil; yield: 65.7 mg, (91%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.5 Hz, 1H), 8.02 (d, J = 8.6 Hz, 2H), 7.74 (d, J = 8.6 Hz, 2H), 7.69 (t, J = 7.7 Hz, 1H), 7.58 (d, J = 7.8 Hz, 2H), 7.37 (d, J = 7.8 Hz, 1H), 7.31 (t, J = 7.4 Hz, 1H), 7.20 (t, J = 7.7 Hz, 3H), 5.36 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 173.7, 172.3, 155.7, 149.4, 148.9, 142.3, 136.7, 135.8, 132.4, 129.8, 129.2, 128.5, 123.2, 122.5, 121.8, 51.2; HRMS (ESI+): m/z [M + Na]+ calcd for C20H15N3O4Na: 384.0955; found: 384.0956.
N-Benzoyl-N-(pyridin-2-ylmethyl)-[1,1′-biphenyl]-2-carboxamide (5f): colorless oil; yield: 40.5 mg, (80%). 1H NMR (400 MHz, CDCl3): δ 8.47 (d, J = 4.5 Hz, 1H), 7.56 (td, J = 7.7, 1.7 Hz, 1H), 7.43–7.40 (m, 3H), 7.39–7.33 (m, 3H), 7.27–7.22 (m, 4H), 7.21 (d, J = 11.7 Hz, 1H), 7.18–7.16 (m, 1H), 7.15–7.12 (m, 1H), 7.12–7.07 (m, 3H), 4.87 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 174.4, 172.9, 156.4, 149.3, 139.7, 139.4, 136.6, 136.0, 135.8, 131.6, 130.3, 129.8, 129.8, 128.7, 128.6, 128.5, 127.9, 127.2, 122.2, 121.7, 51.0; HRMS (ESI+): m/z [M + Na]+ calcd for C26H20N2O2Na: 415.1417; found: 415.1429.
N-Benzoyl-2-bromo-N-(pyridin-2-ylmethyl)benzamide (5g): colorless oil; yield: 63.9 mg, (81%). 1H NMR (400 MHz, CDCl3): δ 8.55 (d, J = 4.8 Hz, 1H), 7.67 (t, J = 7.8 Hz, 1H), 7.57 (d, J = 7.6 Hz, 2H), 7.46 (d, J = 7.6 Hz, 1H), 7.39 (d, J = 7.9 Hz, 1H), 7.28 (t, J = 8.45, 2H), 7.23–7.16 (m, 3H), 7.10 (t, J = 7.5 Hz, 1H), 7.01 (t, J = 7.8 Hz, 1H), 5.35 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 174.0, 171.1, 156.2, 149.4, 137.8, 136.5, 136.4, 133.3, 131.6, 131.3, 130.3, 128.6, 128.1, 126.8, 122.3, 121.9, 120.8, 50.5; HRMS (ESI+): m/z [M + Na]+ calcd for C20H15BrN2O2Na: 417.0209; found: 417.0205.
N-Benzoyl-3-methyl-N-(pyridin-2-ylmethyl)benzamide (5h): colorless oil; yield: 59.4 mg, (90%). 1H NMR (400 MHz, CDCl3): δ 8.53 (d, J = 4.7 Hz, 1H), 7.67 (t, J = 7.6 Hz, 1H), 7.54 (d, J = 7.7 Hz, 2H), 7.40 (d, J = 7.8 Hz, 1H), 7.35 (d, J = 4.5 Hz, 1H), 7.33 (s, 1H), 7.24 (d, J = 7.3 Hz, 1H), 7.16 (t, J = 7.6 Hz, 3H), 7.04 (d, J = 4.9 Hz, 2H), 5.35 (s, 2H), 2.21 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.4, 174.3, 156.7, 149.4, 137.9, 136.6, 136.6, 136.3, 132.4, 131.6, 129.7, 129.0, 128.1, 128.0, 126.3, 122.3, 121.8, 51.5, 21.1; HRMS (ESI+): m/z [M + Na]+ calcd for C21H18N2O2Na: 353.1260; found: 353.1257.
N-Benzoyl-3-methoxy-N-(pyridin-2-ylmethyl)benzamide (5i): colorless oil; yield: 54.7 mg, (79%). 1H NMR (400 MHz, CDCl3): δ 8.54 (d, J = 4.6 Hz, 1H), 7.67 (t, J = 7.6 Hz, 1H), 7.56 (d, J = 7.7 Hz, 2H), 7.40 (d, J = 7.9 Hz, 1H), 7.30–7.24 (m, 1H), 7.21–7.13 (m, 4H), 7.07 (dd, J = 14.7, 6.7 Hz, 2H), 6.79 (d, J = 8.1 Hz, 1H), 5.35 (s, 2H), 3.70 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.2, 174.1, 159.2, 156.6, 149.3, 137.7, 136.7, 136.5, 131.7, 129.2, 129.1, 128.1, 122.3, 121.8, 121.6, 118.4, 113.7, 55.4, 51.4; HRMS (ESI+): m/z [M + Na]+ calcd for C21H18N2O3Na: 369.1210; found: 269.1214.
N-Benzoyl-3-bromo-N-(pyridin-2-ylmethyl)benzamide (5j): colorless oil; yield: 67.0 mg, (85%). 1H NMR (400 MHz, CDCl3): δ 8.53 (d, J = 4.5Hz, 1H), 7.69 (m, 2H), 7.55 (d, J = 7.5 Hz, 2H), 7.49 (d, J = 7.4 Hz, 1H), 7.36 (t, J = 8.3 Hz, 2H), 7.29 (t, J = 7.29, 1H), 7.23–7.15 (m, 3H), 7.02 (t, J = 7.8 Hz, 1H), 5.34 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 174.0, 172.7, 156.2, 149.4, 138.4, 136.6, 136.3, 134.3, 132.0, 131.9, 129.6, 129.0, 128.3, 127.5, 122.3, 122.1, 121.7, 51.3; HRMS (ESI+): m/z [M + Na]+ calcd for C20H15BrN2O2 417.0209; found: 417.0214.
N-Benzoyl-N-(pyridin-2-ylmethyl)thiophene-2-carboxamide (5k): colorless oil; yield: 57.3 mg, (89%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.7 Hz, 1H), 7.71–7.61 (m, 3H), 7.44–7.36 (m, 3H), 7.32 (t, J = 7.3 Hz, 1H), 7.24 (t, J = 7.32, 2H), 7.20–7.14 (m, 1H), 6.81 (t, J = 4.4 Hz, 1H), 5.35(s, 2H), 13C NMR (100 MHz, CDCl3): δ 173.5, 167.5, 156.5, 149.1, 139.5, 136.8, 136.2, 133.3, 133.0, 131.8, 129.0, 128.3, 127.2, 122.3, 121.8, 51.8; HRMS (ESI+): m/z [M + Na]+ calcd for C18H14N2O2SNa: 345.0668; found: 345.0678.
N-Benzoyl-N-(pyridin-2-ylmethyl)furan-2-carboxamide (5l): colorless oil; yield: 53.3 mg, (87%). 1H NMR (400 MHz, CDCl3): δ 8.51 (d, J = 4.7 Hz, 1H), 7.66 (dd, J = 15.9, 7.7 Hz, 3H), 7.40 (d, J = 7.7 Hz, 1H), 7.33 (t, J = 6.1 Hz, 1H), 7.29–7.19 (m, 3H), 7.17–7.11 (m, 1H), 6.95 (s, 1H), 6.22 (s, 1H), 5.32 (s, 2H); 13C NMR (100 MHz, CDCl3): δ 173.3, 162.9, 156.6, 149.4, 147.9, 145.3, 136.6, 136.0, 131.8, 128.7, 128.1, 122.2, 121.4, 118.8, 112.3, 51.3; HRMS (ESI+): m/z [M + Na]+ calcd for C18H14N2O3Na: 329.0897; found: 329.0902.
N-Acetyl-N-(pyridin-2-ylmethyl)cinnamamide (5m): colorless oil; yield: 50.4 mg, (90%). 1H NMR (400 MHz, CDCl3): δ 8.56 (d, J = 4.9 Hz, 1H), 7.76 (d, J = 15.5 Hz, 1H), 7.67 (d, J = 6.36 Hz, 1H), 7.52 (s, 2H), 7.37 (s, 3H), 7.28 (d, J = 13.6 Hz, 2H), 7.20 (s, 1H), 5.14 (s, 2H), 2.53 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 173.8, 169.4, 156.7, 149.4, 145.2, 136.9, 134.7, 130.4, 128.8, 128.3, 122.5, 121.7, 120.5, 49.5, 26.3; HRMS (ESI+): m/z [M + Na]+ calcd for C17H16N2O2Na: 303.1104; found: 303.1115.
N-(2-Methoxyacetyl)-N-(pyridin-2-ylmethyl)cinnamamide (5n): colorless oil; yield: 50.9 mg, (82%). 1H NMR (400 MHz, CDCl3): δ 8.57 (d, J = 4.7 Hz, 1H), 7.76 (d, J = 15.5 Hz, 1H), 7.67 (td, J = 1.9, 7.6 Hz, 1H), 7.51 (dd, J = 6.6, 2.9 Hz, 2H), 7.37 (dd, J = 5.1, 1.8 Hz, 3H), 7.32 (d, J = 7.9 Hz, 1H), 7.26 (d, J = 15.5 Hz, 2H), 7.21 (dd, J = 7.1, 5.3 Hz, 1H), 5.18 (s, 2H), 4.66 (s, 2H), 3.49 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 174.0, 169.1, 156.3, 149.4, 146.3, 137.0, 134.4, 130.6, 128.9, 128.4, 122.7, 122.2, 119.3, 74.6, 59.3, 48.8; HRMS (ESI+): m/z [M + Na]+ calcd for C18H18N2O3Na: 333.1210; found: 333.1214.
N-(Pyridin-2-ylmethyl)-N-(3,3,3-trifluoropropanoyl)benzamide (5o): colorless oil; yield: 55.4 mg, (86%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.6 Hz, 1H), 7.71–7.67 (m, 2H), 7.64 (td, J = 7.7, 1.6 Hz, 1H), 7.54 (t, J = 7.5 Hz, 1H), 7.43 (t, J = 7.6 Hz, 2H), 7.18 (t, J = 6.4 Hz, 2H), 5.05 (s, 2H), 3.74 (q, J = 9.9 Hz, 2H); 13C NMR (100 MHz, CDCl3): δ 174.1, 166.9 (t, J = 3.0 Hz), 155.3, 149.2, 136.8, 134.4, 132.5, 128.8, 128.4, 123.7 (d, J = 276.0 Hz), 122.5, 121.6, 51.3, 41.9 (q, J = 30.0 Hz); HRMS (ESI+): m/z [M + Na]+ calcd for C16H13F3N2O2Na: 345.0821; found: 345.0825.
Propyl Benzoyl(pyridin-2-ylmethyl)carbamate (5p): colorless oil; yield: 48.8 mg, (82%). 1H NMR (400 MHz, CDCl3): δ 8.56 (d, J = 4.2 Hz, 1H), 7.70–7.62 (m, 3H), 7.48 (t, J = 7.3 Hz, 1H), 7.41 (t, J = 7.5 Hz, 2H), 7.30 (d, J = 7.8 Hz, 1H), 7.20–7.13 (m, 1H), 5.18 (s, 2H), 3.92 (t, J = 6.6 Hz, 2H), 1.27 (h, J = 7.1 Hz, 2H), 0.58 (t, J = 7.4 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 173.0, 156.9, 155.1, 149.4, 136.9, 136.6, 131.3, 128.1, 127.8, 122.2, 121.0, 68.7, 50.4, 21.5, 10.0; HRMS (ESI+): m/z [M + Na]+ calcd for C17H18N2O3Na: 321.1210; found: 321.1225.
(E)-N-Isobutyryl-N-(pyridin-2-ylmethyl)but-2-enamide (5q): colorless oil; yield: 48.3 mg, (98%). 1H NMR (400 MHz, CDCl3): δ 8.52(d, J = 4.8 Hz, 1H), 7.63 (t, J = 7.6 Hz, 1H), 7.17 (d, J = 7.3 Hz, 2H), 7.07–6.98 (m, 1H), 6.48 (d, J = 14.9, 1H), 5.04 (s, 2H), 3.32 (p, J = 6.1, 5.6 Hz, 1H), 1.89 (d, J = 6.9 Hz, 3H), 1.15 (d, J = 6.6 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 181.5, 169.5, 156.9, 149.3, 144.9, 136.8, 125.2, 122.3, 121.2, 49.2, 34.8, 19.5, 18.3; HRMS (ESI+): m/z [M + Na]+ calcd for C14H18N2O2Na: 269.1260; found: 269.1256.
Methyl 3-Oxo-3-(N-(pyridin-2-ylmethyl)isobutyramido)propanoate (5r): colorless oil; yield: 51.7 mg, (93%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 4.8 Hz, 1H), 7.67 (t, J = 7.6 Hz, 1H), 7.29 (d, J = 7.8 Hz, 1H), 7.19 (t, J = 5.0 Hz, 1H), 5.12 (s, 2H), 3.93 (s, 2H), 3.73 (s, 3H), 3.05 (p, J = 6.6 Hz, 1H), 1.10 (d, J = 6.6 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 181.1, 169.2, 168.0, 156.3, 149.3, 137.1, 122.5, 121.2, 52.3, 48.7, 46.0, 34.2, 19.2; HRMS (ESI+): m/z [M + Na]+ calcd for C14H18N2O4Na: 301.1159; found: 301.1158.
N-(2-Methoxyacetyl)-N-(pyridin-2-ylmethyl)isobutyramide (5s): colorless oil; yield: 45.0 mg, (90%). 1H NMR (400 MHz, CDCl3): δ 8.50 (d, J = 4.7 Hz, 1H), 7.64 (td, J = 7.7, 1.7 Hz, 1H), 7.23–7.15 (m, 2H), 5.08 (s, 2H), 4.56 (s, 2H), 3.45 (s, 3H), 3.13 (hept, J = 6.7 Hz, 1H), 1.12 (d, J = 6.7 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 181.0, 174.1, 156.2, 149.3, 136.9, 122.5, 121.5, 74.7, 59.2, 48.2, 34.2, 19.3; HRMS (ESI+): m/z [M + Na]+ calcd for C13H18N2O3Na: 273.1210; found: 273.1218.
N-Isobutyryl-N-(pyridin-2-ylmethyl)cyclopropanecarboxamide (5t): colorless oil; yield: 41.8 mg, (85%). 1H NMR (400 MHz, CDCl3): δ 8.53 (d, J = 3.8 Hz, 1H), 7.66 (td, J = 7.7, 1.7 Hz, 1H), 7.18 (dd, J = 7.6, 4.0 Hz, 2H), 5.16 (s, 2H), 3.41 (p, J = 6.7 Hz, 1H), 2.21–2.15 (m, 1H), 1.17 (d, J = 6.7 Hz, 6H), 1.10–1.06 (m, 2H), 0.92–0.87 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 181.5, 177.8, 157.1, 149.2, 137.0, 122.4, 121.1, 49.2, 35.1, 19.6, 15.8, 10.5; HRMS (ESI+): m/z [M + Na]+ calcd for C14H18N2O2Na: 269.1260; found: 269.1259.
4-Chloro-N-isobutyryl-N-(pyridin-2-ylmethyl)butanamide (5u): colorless oil; yield: 54.2 mg, (96%). 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 33.8 Hz, 1H), 7.65 (t, J = 7.6 Hz, 1H), 7.17 (d, J = 7.3 Hz, 2H), 5.06 (s, 2H), 3.60 (t, J = 6.2 Hz, 2H), 3.27 (p, J = 6.6 Hz, 1H), 2.98 (t, J = 6.8 Hz, 2H), 2.13 (p, J = 6.56 Hz, 2H), 1.15 (d, J = 6.6 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 181.3, 175.7, 156.6, 149.3, 136.9, 122.5, 121.2, 48.8, 44.2, 35.1, 34.9, 27.7, 19.4; HRMS (ESI+): m/z [M + Na]+ calcd for C14H19ClN2O2Na: 307.0998; found: 307.0997.
Benzyl (2-(Benzoyl(pyridin-2-ylmethyl)carbamoyl)-6-methyl-phenyl)carbamate (5v): colorless oil; yield: 92.0 mg (96%). 1H NMR (400 MHz, CDCl3): δ 8.28 (d, J = 4.9 Hz, 1H), 7.72 (dd, J = 8.2, 1.3 Hz, 2H), 7.43 (td, J = 7.7, 1.6 Hz, 1H), 7.34 (d, J = 7.4 Hz, 1H), 7.29–7.21 (m, 5H), 7.16–7.07 (m, 4H), 7.04–6.96 (m, 2H), 6.86 (d, J = 6.6 Hz, 2H), 5.06 (s, 2H), 4.91 (s, 2H), 2.24 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 171.2, 166.2, 155.8, 154.2, 148.7, 137.0, 135.7, 134.8, 134.5, 133.3, 133.2, 131.5, 128.5, 128.4, 128.2, 128.2, 127.7, 126.0, 122.1, 121.6, 69.2, 49.8, 19.0; HRMS (ESI+): m/z [M + Na]+ calcd for C29H25N3O4Na: 502.1737; found: 502.1746.