Abstract
Heterogeneous tandem cyclization of 2-alkynylbenzamides with ammonium acetate is achieved in acetonitrile at 85 °C using a magnetic nanoparticles-immobilized bipy-gold(III) complex and AgSbF6 as catalysts to afford a variety of 1-aminoisoquinoline derivatives in moderate to high yields. This heterogeneous gold catalyst can be easily recovered from the reaction mixture by simply applying an external magnetic field and can be recycled at least seven times without any apparent loss of catalytic activity.
Introduction
1-Aminoisoquinolines are a class of important N-heterocycles and exhibit a wide range of biological activities. For instance, they are inhibitors of thrombin, factor Xa, and rho kinase-1, and they demonstrate activity toward cancers, tumors, malaria, and Parkinson’s disease.1–4 There is a high demand for efficient syntheses of 1-aminoisoquinoline derivatives for high-throughput drug screening due to their excellent biological profiles. The most common method for the preparation of 1-aminoisoquinolines involves the nucleophilic aromatic substitution reaction of 1-haloisoquinolines with amino compounds.5–8 One main drawback of this method is that harsh reaction conditions are required such as the use of strong bases, high reaction temperatures, or high pressures for the preparation of either the target products or the starting materials. In recent years, several alternative approaches have been developed for the construction of 1-aminoisoquinolines. These methods include the domino electrophilic cyclization reactions of 2-alkynylbenzamides 9 or 2-alkynylbenzaldoximes10–12 and transition-metal-catalyzed annulations of benzamidine derivatives with internal alkynes using rhodium,13,14 ruthenium,15,16 or cobalt 17 complexes as catalysts.
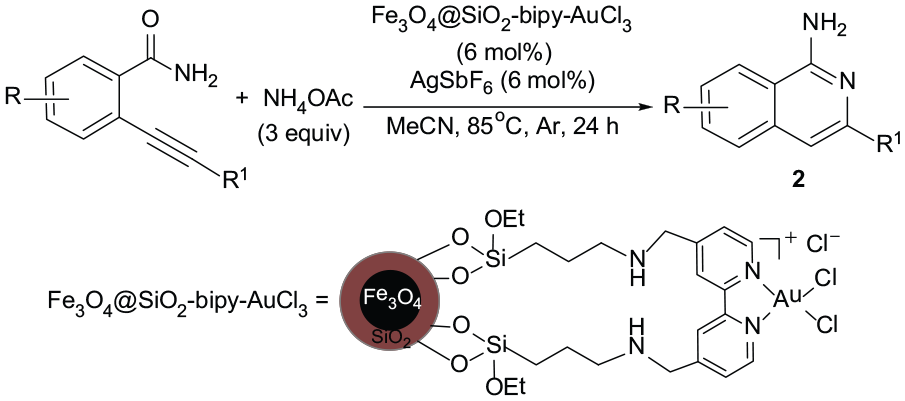
Over the past decades, homogeneous gold-catalyzed organic reactions have become highly efficient and powerful tools for the construction of valuable building blocks.18–22 Recently, gold-catalyzed synthesis of N-heterocycles such as pyrroles,23,24 indoles,25–27 oxazoles,28–30 and quinolines or isoquinolines31,32 has attracted considerable attention due to its high efficiency and mild reaction conditions. Long et al. reported the synthesis of 1-aminoisoquinolines by gold(III)-mediated domino reactions from 2-alkynylbenzamides and ammonium acetate. 33 However, homogeneous gold catalysts are expensive, not recyclable, and difficult to recover from reaction mixtures, which restrict their applications in large-scale or multistep syntheses. Recycling of homogeneous metal catalysts represents one of the most important features of green synthetic methods. Immobilization of existing homogeneous catalysts on various solid supports appears to be a logical solution to this problem. 34 Furthermore, heterogeneous catalysis can minimize the waste generated from reaction work-up, thus developing green and sustainable chemical processes. 35 However, to our knowledge, no examples of the heterogeneous gold-catalyzed synthesis of isoquinolines have been reported until now.
To address the problems of catalyst recovery and recycling, magnetic nanoparticles-supported catalysts have proven to be advantageous because their magnetic separation is an attractive alternative to conventional filtration as it avoids loss of the catalyst and improves the reusability.36–38 Recently, we described the preparation of a magnetic nanoparticle-supported bipy-gold(III) complex (Fe3O4@SiO2-bipy-AuCl3) and its application to the oxidative α-cyanation of tertiary amines. 39 To further expand the application range of this heterogeneous gold catalyst, herein we report a heterogeneous gold(III)-catalyzed tandem cyclization reaction of 2-alkynylbenzamides with ammonium acetate by using Fe3O4@SiO2-bipy-AuCl3 and AgSbF6 as the catalysts leading to 1-aminoisoquinolines in moderate to good yields. The heterogeneous gold(III) catalyst could be easily recovered from the reaction mixture by simply applying an external magnet, and its catalytic activity remains almost unchanged even after recycling seven times.
Results and discussion
The magnetic nanoparticles-immobilized bipy-gold(III) complex (Fe3O4@SiO2-bipy-AuCl3) was synthesized by a simple two-step procedure, as presented in Scheme 1. 39 First, the condensation reaction of silica-coated Fe3O4 (Fe3O4@SiO2) with 4,4′-bis[3-(triethoxysilyl)propylaminomethyl]-2,2′-bipyridine (BTESBPY) in toluene at 110 °C for 24 h provided the bipyridine-modified magnetic nanoparticles (Fe3O4@SiO2-bipy). The Fe3O4@SiO2-bipy was then reacted with AuCl3 in methanol at reflux for 24 h to afford the magnetic nanoparticles-immobilized bipy-gold(III) complex (Fe3O4@SiO2-bipy-AuCl3) as brown nanoparticles; the gold content was found to be 0.62 mmol g−1 based on inductively coupled plasma atomic emission spectroscopy (ICP-AES) analysis.
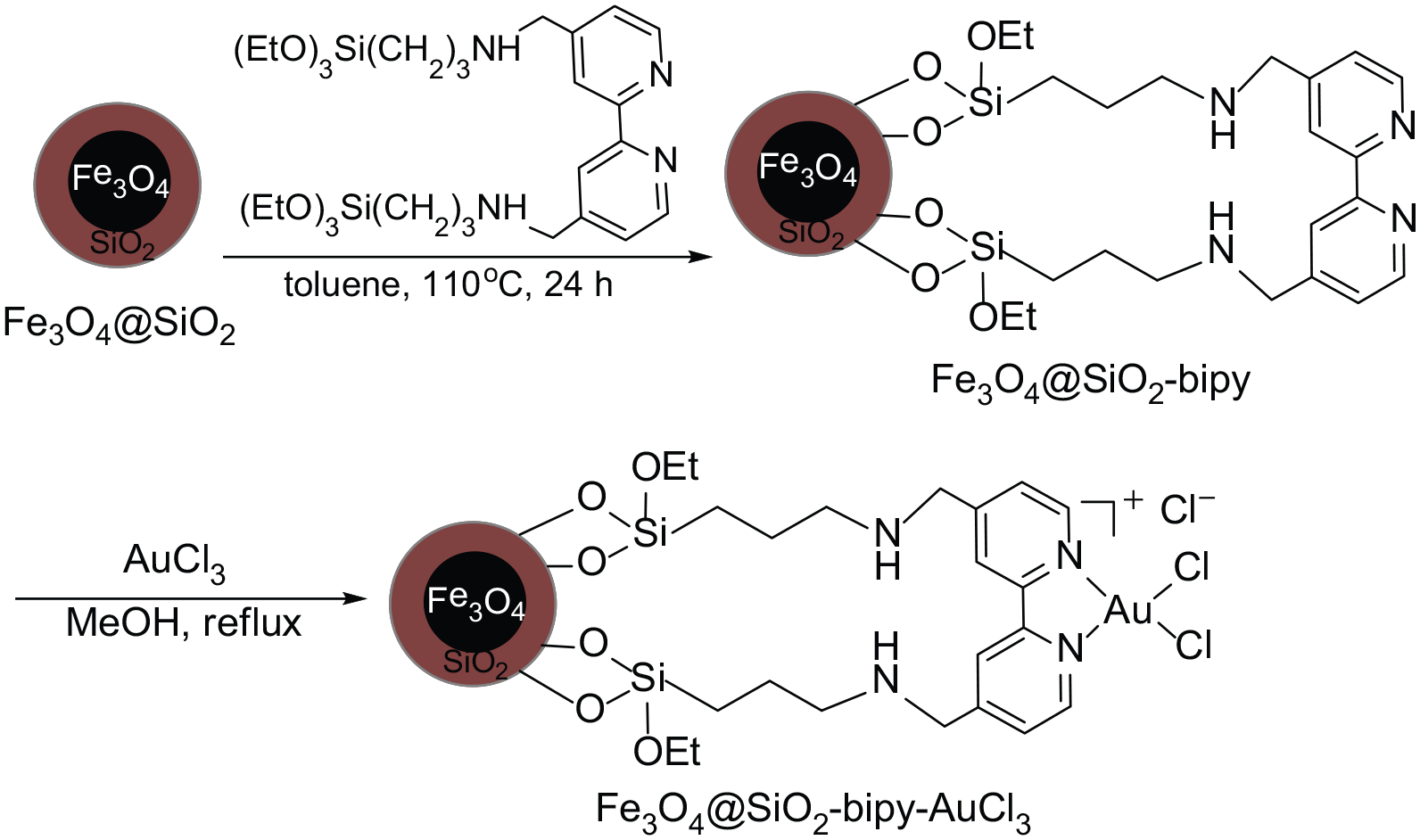
Preparation of the Fe3O4@SiO2-bipy-AuCl3 complex.
The magnetic nanoparticles-immobilized bipy-gold(III) complex (Fe3O4@SiO2-bipy-AuCl3) was then used as the catalyst for the tandem cyclization reactions of 2-alkynylbenzamides with ammonium acetate. In our initial screens, the cyclization of 2-(phenylethynyl)benzamide (
Optimization of the reaction conditions. a

DCE: 1,2-dichloroethane; DME: 1,2-dimethoxyethane; THF: tetrahydrofuran.
All reactions were performed using
Isolated yield.
Two equivalents of NH4OAc were used.
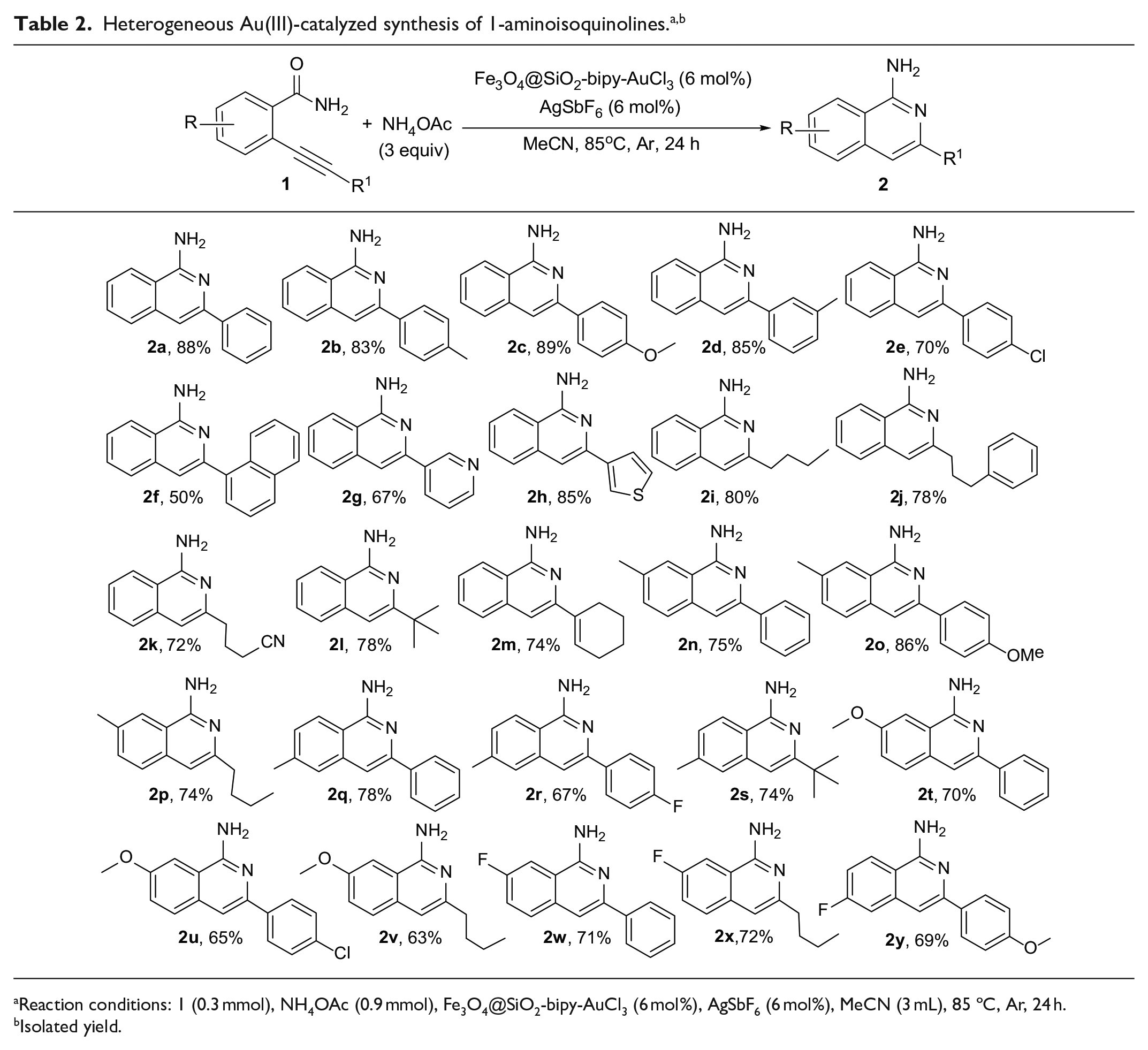
Having optimized the reaction conditions, we next examined the scope of this heterogeneous gold(III)-catalyzed cyclization reaction using a variety of 2-alkynylbenzamides as the substrates and the results are summarized in Table 2. The cyclization reactions of 2-alkynylbenzamides
Reaction conditions: 1 (0.3 mmol), NH4OAc (0.9 mmol), Fe3O4@SiO2-bipy-AuCl3 (6 mol%), AgSbF6 (6 mol%), MeCN (3 mL), 85 ºC, Ar, 24 h.
Isolated yield.
To confirm the heterogeneous nature of this reaction, we focused on the reaction of 2-(phenylethynyl)benzamide (
A possible mechanism for this heterogeneous gold(III)-catalyzed synthesis of 1-aminoisoquinolines

Proposed catalytic cycle.
For the practical application of a supported metal catalyst, its ease of separation from the product and the ability to recycle it are important factors that have to be examined. We next studied the recycling of the Fe3O4@SiO2-bipy-AuCl3 catalyst in the reaction of 2-(phenylethynyl)benzamide (
Recycling of the Fe3O4@SiO2-bipy-AuCl3 complex. a

Reaction conditions:
Isolated yield.
In conclusion, we have developed a novel, efficient, and practical method for the construction of 1-aminoisoquinolines through the gold(III)-catalyzed cyclization reaction of 2-alkynylbenzamides with ammonium acetate using a magnetic nanoparticles-supported gold(III)-bipy complex [Fe3O4@SiO2-bipy-AuCl3] and AgSbF6 as catalysts. The reactions produced a variety of 1-aminoisoquinoline derivatives in moderate to high yields under mild conditions. Importantly, this supported gold catalyst can be easily recovered by simply fixing an external magnet near to the reaction tube and can be recycled up to eight times without any apparent loss of activity. The Fe3O4@SiO2-bipy-AuCl3 catalyst not only solves the basic problems of catalyst separation and recovery but also avoids the use of AgSbF6 in the recycling process. This makes our protocol facile, economical, and environmentally benign.
Experimental
All reagents were used as received without further purification unless otherwise indicated. 2-Alkynylbenzamides
General procedure for the heterogeneous gold-catalyzed synthesis of 1-aminoiso-quinolines
A 20-mL Schlenk tube was charged with Fe3O4@SiO2-bipy-AuCl3 (29 mg, 0.018 mmol), AgSbF6 (6.2 mg, 0.018 mmol), NH4OAc (69 mg, 0.9 mmol), 2-alkynylbenzamide
3-Phenylisoquinolin-1-amine (
3-(p-Tolyl)isoquinolin-1-amine (
3-(4-Methoxyphenyl)isoquinolin-1-amine (
3-(m-Tolyl)isoquinolin-1-amine (
3-(4-Chlorophenyl)isoquinolin-1-amine (
3-(Naphthalen-1-yl)isoquinolin-1-amine (
3-(Pyridin-3-yl)isoquinolin-1-amine (
3-(Thiophen-3-yl)isoquinolin-1-amine (
3-Butylisoquinolin-1-amine (
3-(3-Phenylpropyl)isoquinolin-1-amine (
4-(1-Aminoisoquinolin-3-yl)butanenitrile (
3-(tert-Butyl)isoquinolin-1-amine (
3-(Cyclohex-1-en-1-yl)isoquinolin-1-amine (
7-Methyl-3-phenylisoquinolin-1-amine (
3-(4-Methoxyphenyl)-7-methylisoquinolin-1-amine (
3-Butyl-7-methylisoquinolin-1-amine (
6-Methyl-3-phenylisoquinolin-1-amine (
3-(4-Fluorophenyl)-6-methylisoquinolin-1-amine (
3-(tert-Butyl)-6-methylisoquinolin-1-amine (
7-Methoxy-3-phenylisoquinolin-1-amine (
3-(4-Chlorophenyl)-7-methoxyisoquinolin-1-amine (
3-Butyl-7-methoxyisoquinolin-1-amine (
7-Fluoro-3-phenylisoquinolin-1-amine (
3-Butyl-7-fluoroisoquinolin-1-amine (
6-Fluoro-3-(4-methoxyphenyl)isoquinolin-1-amine (
Supplemental Material
Supporting_Information_for – Supplemental material for Heterogeneous gold(III)-catalyzed tandem cyclization of 2-alkynylbenzamides with ammonium acetate toward 1-aminoisoquinolines
Supplemental material, Supporting_Information_for for Heterogeneous gold(III)-catalyzed tandem cyclization of 2-alkynylbenzamides with ammonium acetate toward 1-aminoisoquinolines by Weisen Yang, Yingying Du, Feiyan Yi and Mingzhong Cai in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the National Natural Science Foundation of China (No. 21462021) and the Natural Science Foundation of Jiangxi Province of China (No. 20161BAB203086) for financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.