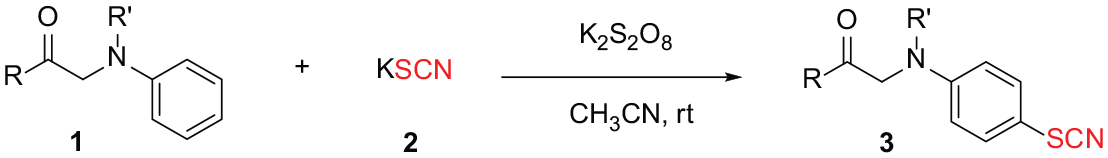Abstract
A procedure for K2S2O8-mediated thiocyanation of α-amino carbonyl compounds has been developed for the synthesis of aromatic thiocyanates. A series of α-amino carbonyl compounds have been investigated, and the desired products are obtained in 74%–93% yields. This strategy has the advantages of simple reaction conditions without use of a transition-metal catalyst, high regioselectivity, and high efficiency. Moreover, we found that arylamine thiocyanates can also be obtained from α-amino carbonyl compounds and potassium thiocyanate in the presence of CoCl2·6H2O, I2, and dimethyl sulfoxide through the cleavage of the C–N bond. To explore the reaction mechanism, we designed several control experiments and proposed a possible mechanism using the experimental results and related literature reports.

Introduction
As an important structural unit, the α-amino carbonyl fragment exists in numerous natural products and pharmaceutical compounds,1–7 and it is a versatile precursor for the synthesis of various heterocyclic compounds.8–14 Glycine, the simplest α-amino carbonyl compound, is one of the essential amino acids in the human body. Besides, amoxicillin and clopidogrel demonstrate the application of α-amino carbonyl compounds in medicine and provide resistance to some diseases. α-Amino carbonyl compounds are involved in many reactions, the most important among which are the reactions with nucleophiles, which include alcohols, 15 thiols, 15 amines,16,17 phosphites,18,19 nitromethane, 20 ketones, 21 1,3-dicarbonyl compounds,22–24 (hetero) arenes,25–31 and others.
To the best of our knowledge, there are no reports on the thiocyanation of α-amino carbonyl compounds. Organic thiocyanates are well known in the area of organosulfur chemistry and have become a hot research field in organic synthetic chemistry, since they are not only the substructures or building blocks of bioactive natural products32–35 but also versatile synthetic precursors for the synthesis of sulfur-containing compounds such as thiols,36,37 thioethers,38,39 and disulfides.40,41 Therefore, it is worthwhile to develop a simple and efficient method to achieve the thiocyanation of α-amino carbonyl compounds for the synthesis of organic thiocyanates.
As a continuation of our research on α-functionalized carbonyl compounds,17,20,42–48 herein, we describe a straightforward synthesis of aromatic thiocyanates through K2S2O8-mediated thiocyanation of α-amino carbonyl compounds using KSCN as the thiocyanation reagent (Scheme 1A). Moreover, we can prepare arylamine thiocyanates from α-amino carbonyl compounds and KSCN in the presence of CoCl2·6H2O, I2, and dimethyl sulfoxide (DMSO) through the cleavage of the C–N bond (Scheme 1B). The α-amino carbonyl compounds can be easily fabricated from ketones and amines49,50 and are used as raw materials for the synthesis of a wide range of compounds. 51

Thiocyanation of α-amino carbonyl compounds.
Results and discussion
We began the investigation using 2-(methyl(phenyl)amino)-1-phenylethan-1-one (
Optimization of reaction conditions. a

Reaction conditions:
Isolated yields based on
At 50 °C.
At 80 °C.
Bold figure represents the best conditions.
With the optimal conditions in hand, we then investigated the substrate scope of the transformation (Table 2). First, we screened the substituents on the aromatic ring of the 1-arylethanone moiety. For the substrates bearing electron-withdrawing substituents on the aromatic ring, including F–, Cl–, I–, CF3–, CN–, and NO2– at the p-position, the protocol was applicable, and the corresponding products were obtained in moderate to good yields (
Reaction conditions:
Isolated yields based on
Moreover, we explored the reactivity of α-amino carbonyl compounds having substituents on the nitrogen atom. The reaction can proceed smoothly when other alkyl groups are attached to the nitrogen atom, such as ethyl, n-butyl, allyl, and benzyl (
Furthermore, we found that arylamine thiocyanates can also be obtained from α-amino carbonyl compounds and KSCN. After screening, the catalyst system CoCl2·6H2O (10 mol%)/I2 (2 equiv.)/DMSO was shown to be the best combination. Then, we investigated the substrate scope of this transformation (Table 3). For the carbonyl moiety, regardless of whether an electron-donating or electron-withdrawing group was attached to the benzene ring, the product
Reaction conditions:
Isolated yields based on
To explore the reaction mechanism, several control experiments were carried out (Scheme 2). When the radical inhibitor TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy) was added to the reaction under the optimal conditions, the product

Control experiments.
Based on these experimental results and previous reports,52–60 a possible mechanism for the thiocyanation of α-amino carbonyl compounds is proposed (Scheme 3). Initially, +SCN is formed from the KSCN by the oxidant and adds to the substrate

Possible mechanism.
Conclusion
In conclusion, we have demonstrated a novel procedure for the synthesis of aromatic thiocyanates through K2S2O8-mediated thiocyanation of α-amino carbonyl compounds. This strategy has the advantages of simple reaction conditions without the use of a transition-metal catalyst, high regioselectivity, and high efficiency. Moreover, arylamine thiocyanates can also be obtained from α-amino carbonyl compounds and potassium thiocyanate in the presence of cobalt catalyst, I2, and DMSO through the cleavage of the C–N bond. These reactions expand further the use of α-amino carbonyl compounds in organic synthesis. Studies to gain insights into the reaction mechanism and application of the protocol for the synthesis of bioactive molecules are currently underway.
Experimental section
General information
Commercially available reagents were purchased from commercial suppliers and used without further purification. Reactions were monitored by thin-layer chromatography (TLC). Flash column chromatography was performed over silica gel (200–300 mesh). 1H and 13C NMR spectra were recorded on a 400 MHz NMR plus spectrometer using residue solvent peaks as internal standards. High-resolution mass spectra were obtained using a GCT time of flight (TOF) instrument with the electron ionization (EI) source. Melting points are uncorrected.
General procedure for the synthesis of product 3
To a sealed tube were added α-amino carbonyl compound
2-(Methyl(4-thiocyanatophenyl)amino)-1-phenylethan-1-one (
1-(4-Fluorophenyl)-2-(methyl(4-thiocyanatophenyl)amino)ethan-1-one (
1-(4-Chlorophenyl)-2-(methyl(4-thiocyanatophenyl)amino)ethan-1-one (
1-(4-Iodophenyl)-2-(methyl(4-thiocyanatophenyl)amino)ethan-1-one (
2-(Methyl(4-thiocyanatophenyl)amino)-1-(4-(trifluoromethyl)phenyl)ethan-1-one (
4-(N-Methyl-N-(4-thiocyanatophenyl)glycyl)benzonitrile (
2-(Methyl(4-thiocyanatophenyl)amino)-1-(4-nitrophenyl)ethan-1-one (
1-(3-Chlorophenyl)-2-(methyl(4-thiocyanatophenyl)amino)ethan-1-one (
1-(3-Bromophenyl)-2-(methyl(4-thiocyanatophenyl)amino)ethan-1-one (
1-(2-Chlorophenyl)-2-(methyl(4-thiocyanatophenyl)amino)ethan-1-one (
2-(Methyl(4-thiocyanatophenyl)amino)-1-(p-tolyl)ethan-1-one (
1-(4-Methoxyphenyl)-2-(methyl(4-thiocyanatophenyl)amino)ethan-1-one (
2-(Methyl(4-thiocyanatophenyl)amino)-1-(m-tolyl)ethan-1-one (
2-(Methyl(4-thiocyanatophenyl)amino)-1-(naphthalen-2-yl)ethan-1-one (
2-(Ethyl(4-thiocyanatophenyl)amino)-1-phenylethan-1-one (
2-(Butyl(4-thiocyanatophenyl)amino)-1-phenylethan-1-one (
2-(Allyl(4-thiocyanatophenyl)amino)-1-phenylethan-1-one (
2-(Benzyl(4-thiocyanatophenyl)amino)-1-phenylethan-1-one (
2-(Methyl(3-methyl-4-thiocyanatophenyl)amino)-1-phenylethan-1-one (
Ethyl N-methyl-N-(4-thiocyanatophenyl)glycinate (
2-(Methyl(4-thiocyanatophenyl)amino)acetonitrile (
General procedure for the synthesis of product 4
To a sealed tube were added α-amino carbonyl compound
N-Methyl-4-thiocyanatoaniline (
N,3-Dimethyl-4-thiocyanatoaniline (
3-Chloro-N-methyl-4-thiocyanatoaniline (
N,2-dimethyl-4-thiocyanatoaniline (
2-Chloro-N-methyl-4-thiocyanatoaniline (
N-Ethyl-4-thiocyanatoaniline (
Supplemental Material
Supporting_information_1 – Supplemental material for Thiocyanation of α-amino carbonyl compounds for the synthesis of aromatic thiocyanates
Supplemental material, Supporting_information_1 for Thiocyanation of α-amino carbonyl compounds for the synthesis of aromatic thiocyanates by Niannian Yi, Mingjing Ouyang, Huimin Liu, Miao Yan, Xiaoyong Wen, Yi Xiong and Bing Yi in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the National Natural Science Foundation of China (No. 21772035) and the Provincial Natural Science Foundation of Hunan (No. 2019JJ50104).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.