Abstract
Amorphous MoS2 spheres with diameters ranging from 150 nm to 1.5 μm are successfully prepared following a reliable one-pot hydrothermal route using a homemade autoclave and inexpensive reactants, at a low temperature (160 °C) and a short reaction time (12 h). The obtained material is comprehensively characterized by X-ray diffraction, scanning-electron microscopy, energy-dispersive X-ray spectroscopy, atomic force microscopy, and infrared spectroscopy. The X-ray diffraction pattern reveals the amorphous structure of the prepared sample without stacking layers, whereas scanning-electron microscopy and atomic force microscopy images indicate the formation of spheres with regular shape and morphology.

Introduction
Transition-metal sulfides, especially molybdenum disulfide (MoS2), are scientifically and technologically important materials and are widely used in applications in electrochemistry, 1 lubrication,2,3 and catalysis,4,5 thanks to their graphene-like structure, intrinsic band gap, and high mobility. Molybdenum disulfide has a hexagonal layer structure which consists of a layer of molybdenum atoms sandwiched between two layers of sulfur atoms, and connected by weak van der Waals forces. 6 This layered structure is particularly important for solid lubrication and as an additive for lubricating oils and greases.
With sustainable development of nanotechnologies, new routes have been developed for the synthesis and the preparation of MoS2 materials on nanoscale via new engineering solutions. It has been well proven that nanosized MoS2 presents better properties than the conventional bulk material. In the lubrication field, MoS2 nanomaterial has better tribological properties, both in friction reduction and wear resistance, than microsized and bulk material. In fact, MoS2 nanoparticles as solid lubricants or additives present several advantages, thanks to their extremely small size that permits delivery to the tribological contacts; they are often efficient at ambient temperature, and therefore no induction period is necessary to obtain satisfactory tribological results. 7 Also, in catalysis, the nanosized MoS2 catalyst exhibits completely new behavior and demonstrates high activity for the hydrodesulfurization (HDS) of dibenzothiophene in the presence of H2S, in contrast to the behavior of the classic conventional MoS2 catalysts that suffer severe inhibition due to the presence of H2S in the HDS feedstock. 8
Therefore, in the last decade, significant efforts have been devoted toward the preparation of MoS2 on the nanometer scale with very specific morphology and size. A variety of synthetic methods have been reported, such as solid-gas or gas phase reactions, 9 electron-beam irradiation activation and arc discharge, 10 and solvothermal synthesis.11,12 The resulting materials present different morphologies such as nanotubes, fullerene-like, nanospheres, and so on.
Note that most of these preparation methods present drawbacks, mainly associated with the use of highly sophisticated devices and toxic gases, as well as high temperatures and pressures. Therefore, simpler alternative methods, based on solution synthesis routes, were found to be more appropriate for the synthesis of nanopowders, specifically relating to the needed for lower temperatures during their synthesis, shorter incubation times, the need for less complicated equipment, and so on.
One of the most promising synthetic techniques is without doubt the solvothermal-hydrothermal process that is widely used for developing soft processing in materials science. This process is based on the use of chemical reactions taking place in a solvent (aqueous or nonaqueous) under high pressure and mild temperature conditions for developing new materials with a diversity of morphology. Also, the solvothermal-hydrothermal process can be used to grow nanosized crystalline materials without annealing treatment, which allows for an important savings in energy. 11
Likewise, and for environmental requirements, the hydrothermal method is more interesting because of the use of water as the solvent, which is environmentally safe and cheaper than other solvents. In fact, the properties of water under hydrothermal conditions can be significantly changed. Under high temperature and pressure hydrothermal conditions, the surface tension, density, and viscosity of water are lower, and the vapor pressure and ion product are higher, giving adequate space for the growth of new materials.
Recently, the hydrothermal method was used to prepare new MoS2 nanomaterials with very special properties. The application of hydrothermal method was first published by Li et al. 13 for the synthesis of MoS2 nanowires with diameters of 4 nm and lengths of 50 nm by using MoO3 and Na2S as precursors in HCl solution at 260 °C for 12 h in the presence of an additive (CH3CH2ONa or NH4Cl). They demonstrated that the growth of MoS2 nanowires as well as their morphology and size depend on the additive nature, the concentration of precursors, and the reaction temperature. Therefore, several studies have described the preparation of molybdenum disulfide nanomaterials following the hydrothermal method, a process considered as environmentally friendly. Y Wang et al. 14 have prepared different morphologies of MoS2 under hydrothermal conditions related to pressure in the range of 0.1–3.5 Mpa at 200 °C. Also, Tian et al. have synthesized amorphous MoS2 nanospheres, with an average diameter of 30 nm, through a two-step hydrothermal reaction. The first step is the redox reaction and the second is the sulfurizing reaction at 120 °C for 12 h. 15
Also, Tang et al. 3 have prepared flower-like MoS2 microspheres with a mean diameter of about 1 μm through a Pluronic F-127 (ethylene oxide/propylene oxide block copolymer) assisted hydrothermal method using a 100-mL Teflon-lined stainless steel autoclave at 180 °C for 24 h. Also, Lu et al. 16 obtained MoS2 nanoflowers by one-step hydrothermal fabrication using polypyrrole as a template. Guogang and co-workers 17 have prepared flower-like hollow microspheres by a hydrothermal method with the assistance of a surfactant (pluronic F-127). The authors assumed that the reaction time and the surfactant concentration played an important role in the final growth morphology. Furthermore, Zhang and co-workers 18 have reported the preparation of highly dispersive 3D flower-like MoS2 microspheres with a diameter of about 4 μm following a hydrothermal approach. The experimental results suggest that the reaction time played an important role in controlling the morphology of the obtained final structures.
In fact, the hydrothermal technique for the preparation of MoS2 materials offers several advantages with respect to highly controlled diffusivity in a strong solvent medium in a closed system (autoclave), as well as the energy saving, the use of larger volume equipment, better nucleation control, higher dispersion and rates of reaction, better shape control, and lower temperature operations. 19 The technique allows the formation of particles with high purity, high crystallinity, and high-quality mono-dispersion with controlled physical and chemical characteristics which, today, are in great demand in the fields of industry and research.
However, the use of templates or surfactants for better control of the size and shape17,20,21 represents a major drawback of this technique, because of the hazardous nature of these components and the harmful gas released during the reaction process.
In this work, we describe a successful, simple, and facile one-pot hydrothermal method under mild conditions (subcritical conditions) to prepare a mixture of nano and microsized amorphous MoS2 spheres without using a template or surfactant. The obtained material has been studied in detail by comprehensive techniques that permit structural and morphological characterizations.
Results and discussion
Figure 1 shows the X-ray diffraction (XRD) patterns of the prepared MoS2 and the bulk hexagonal MoS2. It can be seen from the pattern of the prepared sample the absence of any well-defined peak with only the presence of a broad diffraction peak indicating the disordered phase nature of the obtained material. The latter peak envelope beginning at about 2θ = 30° and ends at 2θ = 40° with a maximum approximately located at the 100 position assigned to bulk 2H-MoS2. Another broad peak, with very low intensity, was also observed and can be attributed to the 110 position of 2H-MoS2 (JCPDS card No. 37-1492, molybdenite). This XRD pattern indicates that the prepared material is of amorphous structure. 12 Note that the absence of the 002 diffraction in the XRD of the obtained material indicates that no stacking of layers has taken place. 22

XRD patterns of the as-prepared MoS2 and bulk 2H-MoS2.
The morphology of the sample was primarily investigated by scanning-electron microscopy (SEM). SEM images show that the sample is formed entirely of agglomerated spheres with regular shape and polydispersed in size, having an average diameter near to 1 μm (Figure 2). Chemical analysis using energy-dispersive X-ray spectroscopy (EDS) indicates only the presence of Mo and S (Figure 3). The quantification of the peaks shows that the atomic ratio of S to Mo is 1:87, a value close to that of stoichiometric MoS2.

SEM images of the obtained spheres.

EDS spectrum of the as-prepared MoS2.
A detailed characterization by atomic force microscopy (AFM) allows observation of the spherical morphology of the obtained material (Figure 4). The sample is formed as a mixture of nanospheres and microspheres, with a diameter ranging from 150 nm to 1.5 μm.

AFM images of the as-obtained MoS2 spheres.
The obtained spheres were also investigated by infrared spectroscopy (IR). Figure 5 shows the IR spectra of the obtained MoS2 and bulk 2H-MoS2, for comparison purposes. The IR spectrum of bulk MoS2 (Figure 5(a)) shows the presence of a single band at 485 cm−1 corresponding to the Mo-S stretching mode of vibration. 23 For the obtained material, the band corresponding to Mo-S is also present (Figure 5(b)) at 533 cm−1 attributed to bridging S22− species, possibly related to the linking of MoS2 sheets. 11 The IR spectra also indicate the presence of a strong IR band at 835 cm−1 corresponding to the symmetrical stretching vibrations of the cis dioxo group MoO22+. 24 Also, the IR bands at 650, 990, 1150, and 1420 cm−1 are attributed to sulfate groups. 23 These bands are commonly present in amorphous MoS2 samples as a result of sample surface oxidation with air. 25 A band at 1230 cm−1 was also observed and could be assigned to residual ammonia or ammonium ions produced from decomposition of the precursor [(NH4)6Mo7O24. (4H2O)]. 26
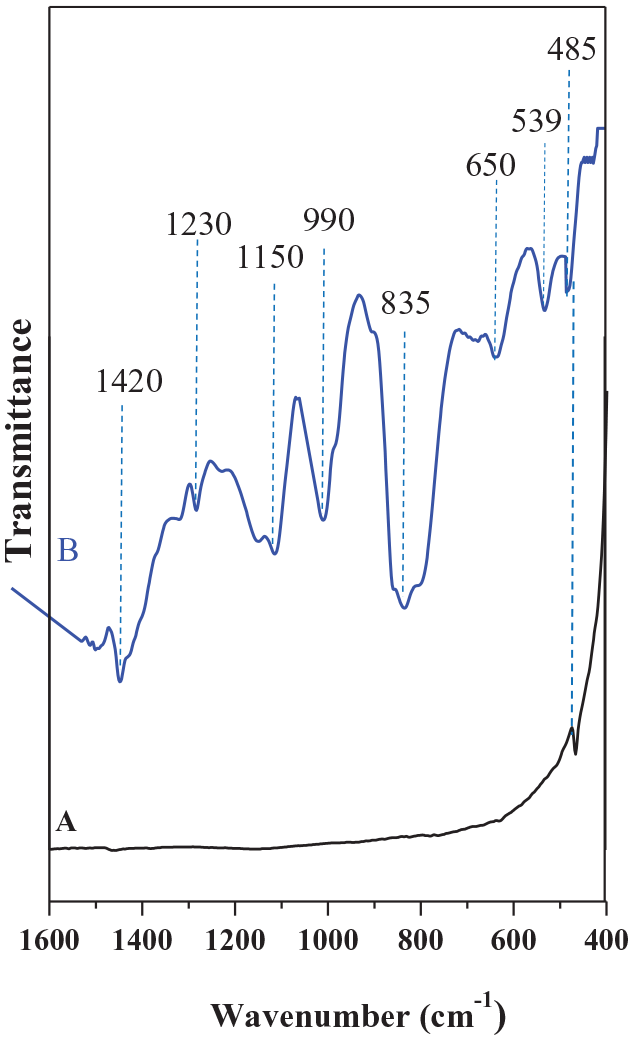
IR spectra of (a) bulk 2H-MoS2 and (b) the obtained MoS2 sample.
The preparation method, developed in the present work, allows high-purity MoS2 spheres to be obtained with regular shape following a subcritical hydrothermal approach. This brought into question the mechanisms proposed in the literature for the preparation of MoS2 nanospheres and microspheres that suggest that obtaining spherical structures depends on the nature of the templates and surfactants, ionic liquids, and ionic strength.17,20,21,27 However, based on the results obtained from this study, it is assumed that the formation of MoS2 spherical structures can be described as a sequence of three main steps: an oxidation-reduction reaction, nucleation, and germ growth. The oxidation-reduction reaction leads to the formation of MoO42− and S2− that allows MoS2 particles to be obtained through a fast nucleation process. In fact, during this step, the solution is metastable and concentration fluctuations occurring therein yield to the fast nucleation process of small particles that appear and disappear continually. Subsequently, stable particles are produced at a constant rate through the Ostwald ripening process. 28 During this phase, particles having a high surface energy will dissolve in favor of the growth of larger and stable particles. However, the size homogeneity of the MoS2 spheres is related to the reaction time. In fact, for a shorter reaction time, the growth mechanism will not be achieved by all the formed germs, and therefore, small and large particles will coexist together at the end of the reaction, which explains the lower size dispersion of the obtained spheres.
As a matter of fact, the growth of MoS2 spheres after the nucleation process depends mainly upon the reaction environment, which is related to the reaction time and pressure. Under the hydrothermal conditions, the temperature is higher than the boiling point of water and the autogenous pressure increases with the temperature. 29 This pressure increases and governs the solubility of precursors while the difference of temperature in the autoclave induces a solubility difference that allows the transport of matter. Figure 6 shows a schematic representation of the proposed hydrothermal process inspired from the Darracq et al. 29 model. The autogenous pressure in the closed autoclave, determined by its filling rate, is mainly increased by the decomposition of the ammonium precursor and hydrazine, which leads to the release of gas, and automatically accelerates the solubility of the precursors and consequently, transport of solvated species by convection phenomena from the hot zone to the cold zone of the autoclave occurs, which causes saturation of the cold zone and the beginning of sedimentation of MoS2 spheres (Figure 6).

Schematic representation of hydrothermal growth of MoS2 spheres. 29
Conclusion
MoS2 spheres with a diameter ranging from 150 nm to 1.5 μm were successfully synthesized through a one-pot, facile, reproducible, and simple hydrothermal method under mild conditions (160 °C, 12 h). The structural characterization indicates that the obtained spheres have an amorphous structure and regular shape and morphology.
A mechanism for sphere formation has been proposed and consists of nucleation and germ growth. The reaction pressure and time are assumed to be the key parameters for the good progress of the germ growth process and therefore obtaining spheres with homogeneous size distribution.
Experimental
Synthesis
All the reagents were purchased from Sigma-Aldrich and were of analytical grade and used without further purification. The typical experimental procedure was designed as follows: 1.9 g of elemental sulfur (S), 5.4 g of ammonium molybdate [(NH4)6Mo7O24. 4H2O], and 27 mL of hydrazine monohydrate (N2H4. H2O) were placed into a homemade Teflon-lined stainless steel autoclave which was then filled with water up to 70% of its total volume (190 mL). The autoclave was then sealed and placed in an oven maintained at 160 °C for 12 h. After heating, the autoclave was left to cool to room temperature. The formed black precipitate was recovered by centrifugation and washed repeatedly with acetone and distilled water. The resulting powder was then dried under vacuum at 70 °C for 4 h.
Characterization
The phase purity and crystalline structure of the MoS2 nanospheres were studied by XRD using a Bruker D8 ADVANCE ECO diffractometer with Cu/Kα radiation (λ = 0.154 nm) operating at 35 kV and 35 mA. The Bragg’s angles between 4° and 80° were scanned at a rate of 0.04°/s.
The morphology of the MoS2 sample was observed by SEM/EDS on a Hirox SH-4000M. AFM observations were performed on a Nanosurf FlexAFM C3000, operating at room temperature, at 150.97 kHz and 800 mV, in phase contrast mode, using an NCLR cantilever. The AFM specimens were prepared by dispersing a small amount of MoS2 powder in ethanol and placing one drop of the dispersion on a lacey mica film and allowing the solvent to evaporate.
Fourier transform–infrared spectroscopy (FT-IR) was performed on a Jasco 410 spectrometer at a spectral resolution of 4 cm−1. MoS2 nanospheres powder (2 wt%) was thoroughly ground and mixed with KBr and pressed into disks.
Supplemental Material
supplementary_File – Supplemental material for Subcritical hydrothermal synthesisof amorphous molybdenum disulfide spheres
Supplemental material, supplementary_File for Subcritical hydrothermal synthesisof amorphous molybdenum disulfide spheres by Hanane Akram, Ouafae Achak and Tarik Chafik in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Research Institute for Solar Energy and New Energies, IRESEN, Morocco, under project Innowind13Nanolubricant. The authors also thank SALUB Company (Afriquia Holding, Morocco) for kindly supplying the 2H-MoS2.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
