Abstract
Selective mono-aza-Michael additions of divinyl ketones with 3-(arylimino)indolin-2-ones in the presence of cesium carbonate are described. N-Enone-functionalized 3-(arylimino)indolin-2-ones were efficiently synthesized in satisfactory yield. The salient features of this protocol are high regioselectivity, high yield, and mild conditions.
Keywords

Introduction
The indolin-2-one framework bearing various functional groups at the N-position is a privileged heterocyclic motif that is found in many bioactive natural products and pharmaceutically active compounds.1,2 One important method for the N-functionalization of indolin-2-one is the aza-Michael addition. However, the 3-position is more active than the 1-position for indolin-2-one making the Michael addition to C-3 more favorable.3–5 Therefore, 3-substituted indolin-2-ones may be better candidates for aza-Michael additions. 3-(Arylimino)indolin-2-ones, synthesized by reactions of isatins with aromatic amines, 6 may act as good aza-Michael donors. For example, Imanzadeh et al. 7 reported the aza-Michael additions of 3-(arylimino)indolin-2-ones with fumaric esters; Zari 8 described the thiourea catalyzed asymmetric aza-Michael additions of 3-(arylimino)indolin-2-ones with unsaturated 1,4-diketoesters; Metsala 9 investigated these aza-Michael reactions by means of density functional theory (DFT) calculations; and Metsala 10 also performed conformational analyses across reaction paths for these aza-Michael reactions.
Divinyl ketones (e.g. 1,4-dien-3-ones) have been used as Michael acceptors in recent years to synthesize spiro-, cyano-, and other important compounds.11–19 However, divinyl ketones as Michael acceptors have problems of regioselective control and can undergo 1,2-addition and two kinds of 1,4-addition reactions.
Herein, we report the regioselective mono-aza-Michael additions of divinyl ketones with 3-(arylimino)indolin-2-ones under mild, transition metal-free and acid-free conditions to synthesize N-enone-functionalized 3-(arylimino)indolin-2-ones bearing both C=C and C=N functional groups.
Results and discussion
In previous work, the reactions of 3-(arylimino)indolin-2-ones with enones had been reported, which could give the aza-Michael addition products in satisfactory yields.
20
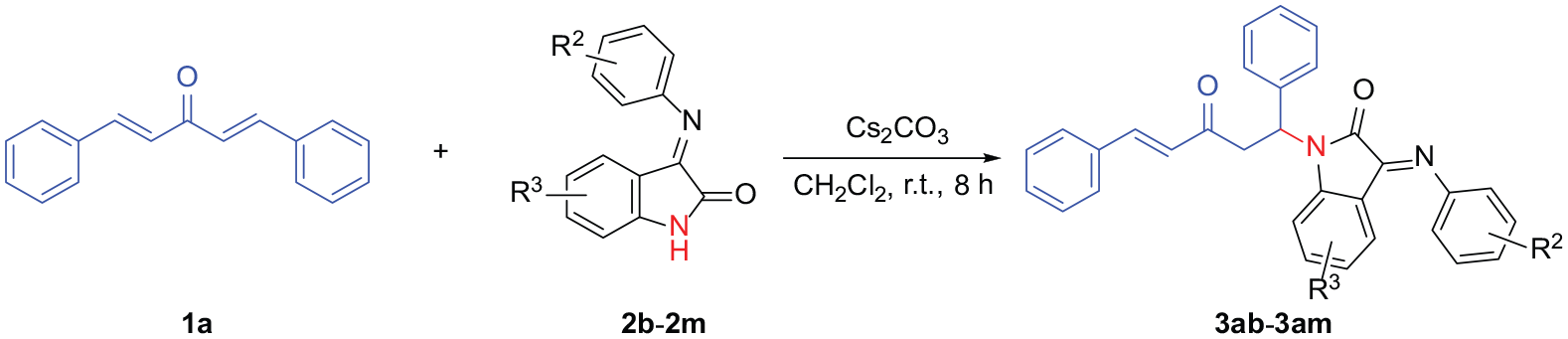
In order to compare enones and divinyl ketones, the aza-Michael additions of 3-(arylimino)indolin-2-ones with divinyl ketones were investigated. Initially, (1E,4E)-1,5-diphenylpenta-1,4-dien-3-one (
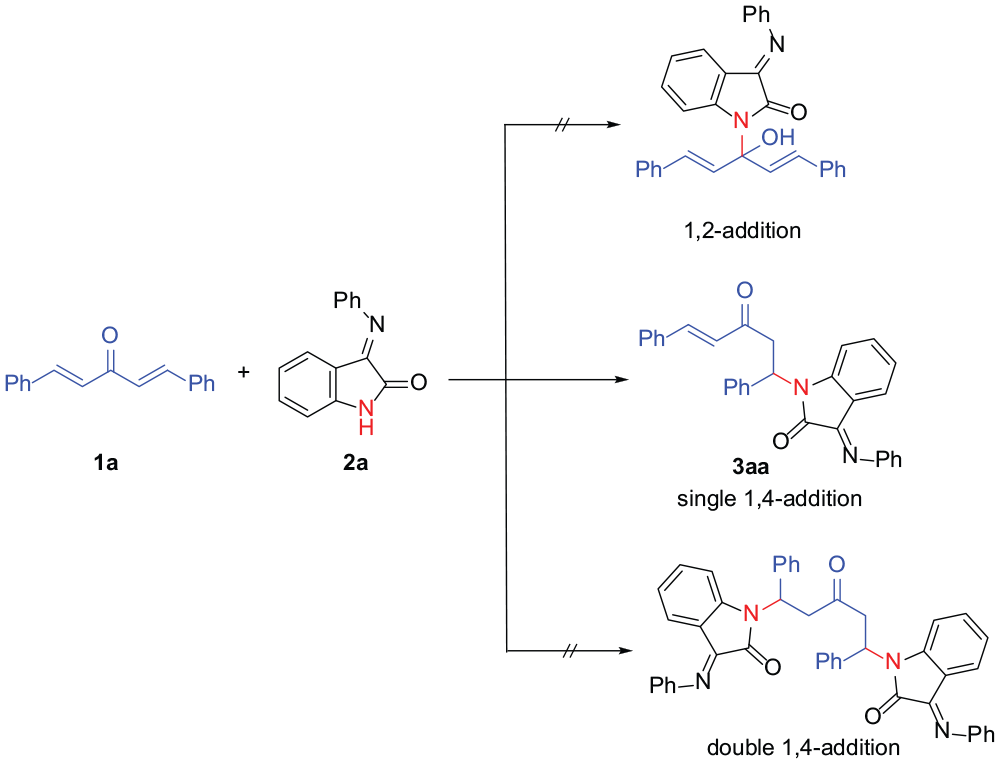
Regioselective addition of
In order to optimize further the reaction conditions for the model reaction, other bases including inorganic bases, such as NaOH, K2CO3, Na2CO3, CsF, and Cs2CO3, and organic bases, such as Et3N, 1,4-diazabicyclo[2.2.2]octane (DABCO), 4-dimethylaminopyridine (DMAP), and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), were examined for the reaction (Table 1, entries 2−5 and Supporting information, Table S1). Of these, Cs2CO3 as a base gave the best yield of the desired product
Optimization of the reaction conditions. a

Reaction conditions:
Isolated yields.
Reaction condition: 50 °C.
Reaction condition:
With the optimized reaction conditions in hand, the generality of this reaction was examined for the reactions of various divinyl ketones with 3-(arylimino)indolin-2-ones in CH2Cl2 at room temperature using Cs2CO3 as the base. The reactions were first tested using various divinyl ketones (
The reactions of various divinyl ketones with
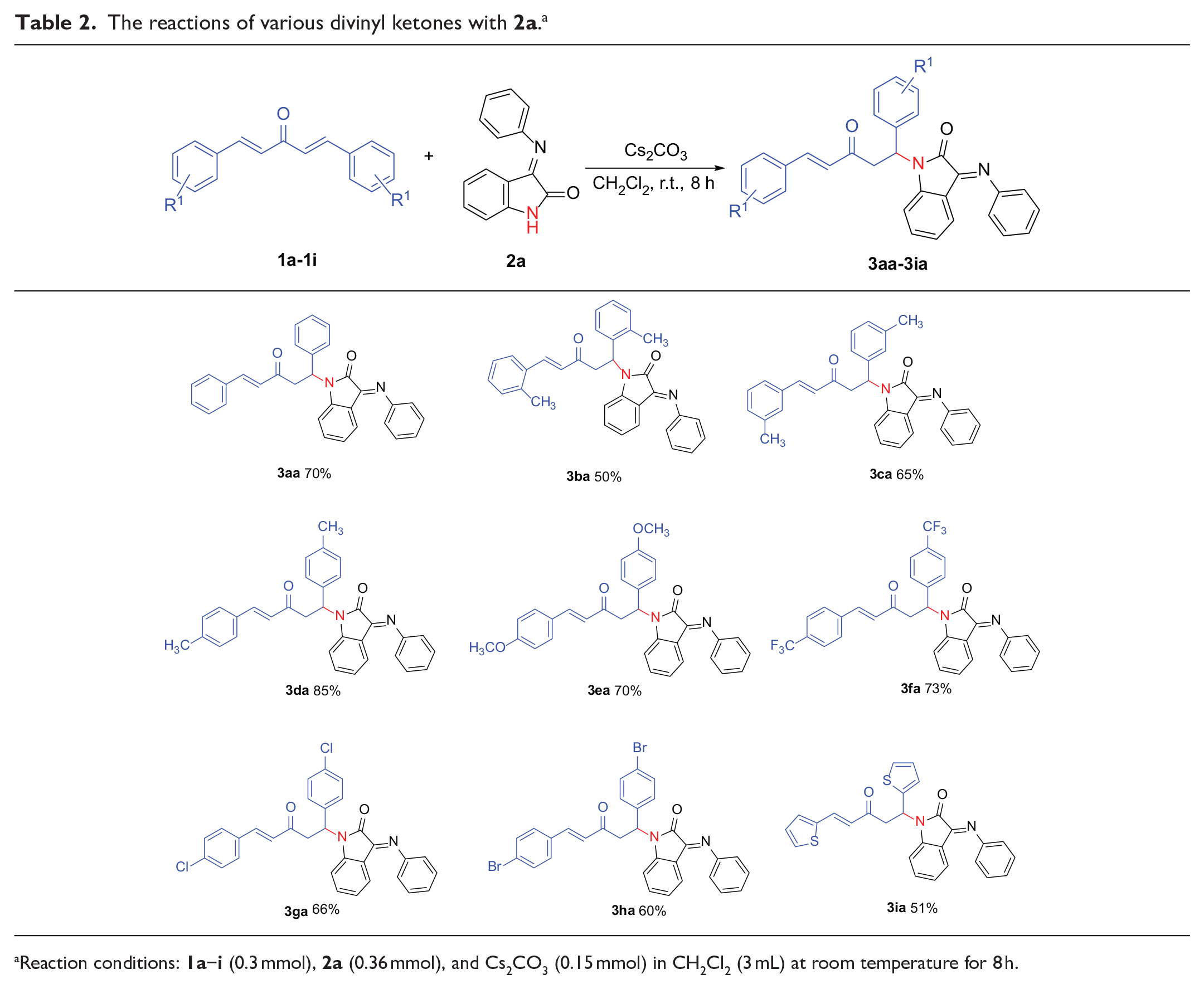
Reaction conditions:
The scope of the reactions of
The reactions of
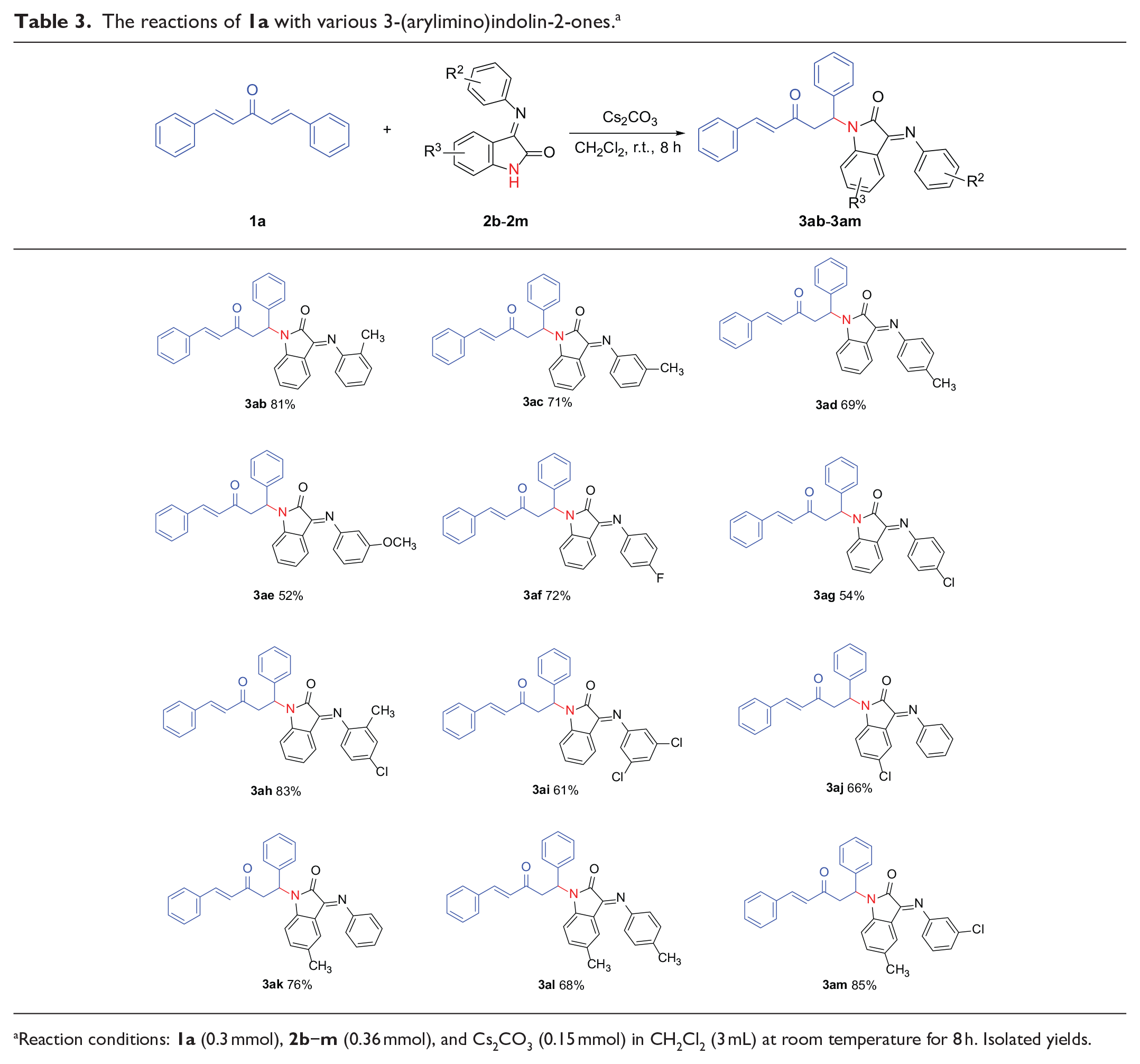
Reaction conditions:
The reaction of

Gram-scale synthesis of
A plausible mechanism is proposed for the reaction of

The proposed mechanism for formation of product
Conclusion
In conclusion, an efficient method for mono-aza-Michael additions of symmetric divinyl ketones with 3-(arylimino)indolin-2-ones using cesium carbonate as the base has been developed. This protocol has advantages of high regioselectivity, high yields, and mild conditions. This will be a useful alternative for the synthesis of N-enone-functionalized indolin-2-ones. Owing to the compatibility of multiple functional groups, such as C=C, C=O, C=N, and aryl rings, with the reaction, the resulting compounds will have many applications for the synthesis of other useful fine chemicals.
Experimental
General information
1H NMR and 13C NMR spectra were obtained with Mercury-400BB and Mercury-600BB instruments using CDCl3 as solvent and Me4Si as the internal standard. High-resolution mass spectra (HRMS) (ESI) were obtained with a Bruker Daltonics APEX II 47e and Orbitrap Elite mass spectrometer. Divinyl ketones21,22 and 3-(arylimino)indolin-2-ones6,23 were synthesized according to literature procedures.
The general procedure for the mono-aza-Michael additions of divinyl ketones with 3-(arylimino)indolin-2-ones: synthesis of 3aa −3ia and 3ab −3am
A mixture of divinyl ketone (0.3 mmol), 3-(arylimino)indolin-2-one (0.36 mmol), and cesium carbonate (0.15 mmol) in methylene chloride (3 mL) was stirred under air at room temperature for 8 h. Then, the resulting mixture was extracted with ethyl acetate (2 × 5 mL). The combined organic layers were dried over anhydrous magnesium sulfate and concentrated under reduced pressure. The residues were isolated by column chromatography using petroleum ether and ethyl acetate (v/v 5:1) as eluent to give pure product. Analytical data for the products are given below.
(E)-1-((E)-3-Oxo-1,5-diphenylpent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-di-o-tolylpent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-di-m-tolylpent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-di-p-tolylpent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-1,5-Bis(4-methoxyphenyl)-3-oxopent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-bis(4-(trifluoromethyl)phenyl)pent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-1,5-Bis(4-chlorophenyl)-3-oxopent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-1,5-Bis(4-bromophenyl)-3-oxopent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-di(thiophen-2-yl)pent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-diphenylpent-4-en-1-yl)-3-(o-tolylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-diphenylpent-4-en-1-yl)-3-(m-tolylimino)indolin-2-one (
(E)-1-((E)-3-Oxo-1,5-diphenylpent-4-en-1-yl)-3-(p-tolylimino)indolin-2-one (
(E)-3-((3-Methoxyphenyl)imino)-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)indolin-2-one (
(E)-3-((4-Fluorophenyl)imino)-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)indolin-2-one (
Orange solid (102 mg, 72%). m.p. 76 °C−78 °C. 1H NMR (600 MHz, CDCl3) δ 7.64 (d, J = 16.2 Hz, 1H), 7.56 (d, J = 7.4 Hz, 2H), 7.53 (dd, J = 7.3, 2.3 Hz, 2H), 7.41–7.35 (m, 5H), 7.32 (d, J = 7.8 Hz, 2H), 7.10 (t, J = 8.6 Hz, 2H), 7.05 (d, J = 8.1 Hz, 1H), 6.94 (dd, J = 8.8, 4.8 Hz, 2H), 6.79 (d, J = 16.2 Hz, 1H), 6.77–6.72 (m, 1H), 6.68 (d, J = 7.7 Hz, 1H), 5.80 (dd, J = 9.1, 5.2 Hz, 1H), 4.48 (dd, J = 17.5, 9.1 Hz, 1H), 3.53 (dd, J = 17.5, 5.2 Hz, 1H). 13C NMR (150 MHz, CDCl3) δ 196.8, 163.5, 160.5 (d, J = 244.4 Hz), 154.6, 147.7, 146.2 (d, J = 2.9 Hz), 143.9, 138.6, 134.2, 134.2, 130.7, 129.0, 128.9, 128.5, 128.2, 127.2, 125.8 (d, J = 25.3 Hz), 122.5, 119.5 (d, J = 8.0 Hz), 116.3, 116.2, 115.7, 110.6, 53.4, 43.0. HRMS (ESI): Calcd for C31H23FN2O2 [M+H]+ 475.1816, found 475.1816.
(E)-3-((4-Chlorophenyl)imino)-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)indolin-2-one (
(E)-3-((4-Chloro-2-methylphenyl)imino)-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)indolin-2-one (
(E)-3-((3,5-Dichlorophenyl)imino)-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)indolin-2-one (
(E)-5-Chloro-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-5-Methyl-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)-3-(phenylimino)indolin-2-one (
(E)-5-Methyl-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)-3-(p-tolylimino)indolin-2-one (
(E)-3-((3-Chlorophenyl)imino)-5-methyl-1-((E)-3-oxo-1,5-diphenylpent-4-en-1-yl)indolin-2-one (
Supplemental Material
Supporting_information – Supplemental material for Regioselective mono-aza-Michael additions of divinyl ketones with 3-(arylimino)indolin-2-ones: Synthesis of N-enone-functionalized 3-(arylimino)indolin-2-ones
Supplemental material, Supporting_information for Regioselective mono-aza-Michael additions of divinyl ketones with 3-(arylimino)indolin-2-ones: Synthesis of N-enone-functionalized 3-(arylimino)indolin-2-ones by Xiao Chen and Zheng Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the National Natural Science Foundation of China (21462038) for the financial support of this work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.