Abstract
The synthesis of C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives at the 4-OH via click reactions is accomplished, and a total of 16 novel sinomenine double N-heterocyclic derivatives are obtained in 74%–95% yields. The C-ring is first transformed into a 1,2-diketone structure under the action of hydrochloric acid, and then reacted with o-phenylenediamine to obtain a C-ring quinoxaline-substituted structure. The 4-OH of sinomenine reacts with chloropropyne to give an alkynyl sinomenine, and then reacts with sodium azide and various benzyl chlorides to give the target compounds. All the synthesized derivatives are characterized by Fourier-transform infrared spectrometry, high resolution mass spectrometry, 1H NMR, and 13C NMR spectroscopy.
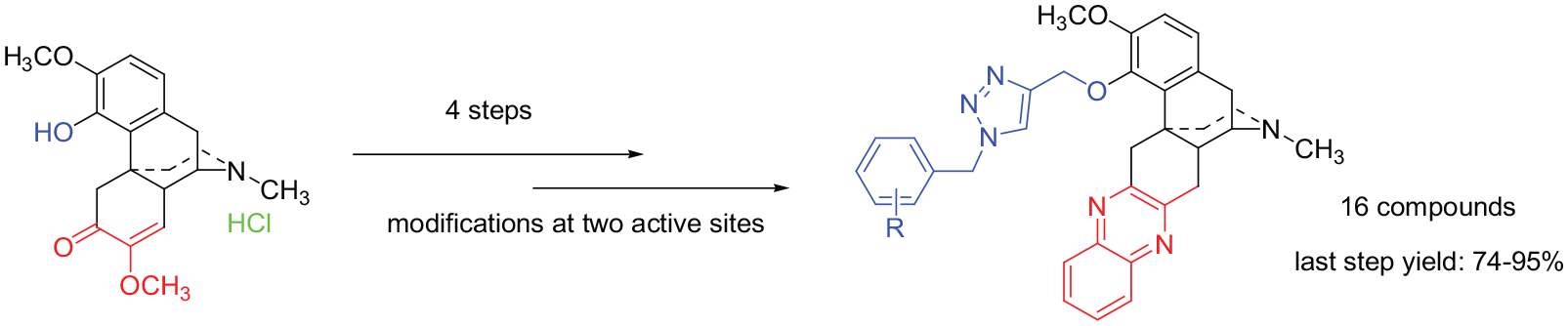
Sinomenine is mainly extracted from the roots and stems of the Sinomenium acutum1,2 and exists mostly in the form of sinomenine hydrochloride. It has been found to have unique effects in the treatment of rheumatoid arthritis with 85% rate total effective.3,4 It also exhibits pharmacological activities including antiarrhythmic, antineoplastic, analgesic and immunoregulatory effects.5–8 However, some side effects of sinomenine itself, such as slower duration of action and allergies,1,6,9–13 have limited the application of sinomenine in some diseases. Hence, structural modification research is inevitable as a preferred means to obtain derivatives with higher biological activity and lower side effects.
Structurally, sinomenine is similar to the morphine skeleton and consists of four fused rings. It contains a number of functional groups, such as aryl, methoxyl, phenolic hydroxy, carbonyl, double bond, and N-methyl. So it is very suitable for modification research. Previous structural modification often focused on only one site. Heterocyclic structures often show better biological activities. Therefore, modifying the heterocyclic components of sinomenine is an effective means to improve the biological activity of this natural product. Based on our previous research,14,15 herein we report a new process for double N-heterocyclic structure modifications on the A-ring and C-rings, respectively, in order to obtain potentially improved pharmacologically active derivatives.
Results and Discussion
Scheme 1 demonstrates the synthetic method toward the double N-heterocyclic sinomenine derivatives in four steps, and 16 target compounds are synthesized in this work. 1,4-Quinoxaline is a six-membered benzo N-heterocyclic ring, and 1,2,3-triazole is a five-membered N-heterocyclic ring. Compounds containing these heterocyclic structures often exhibit good biological activity.16–18 The 1,2-diketone scaffold sinomenine
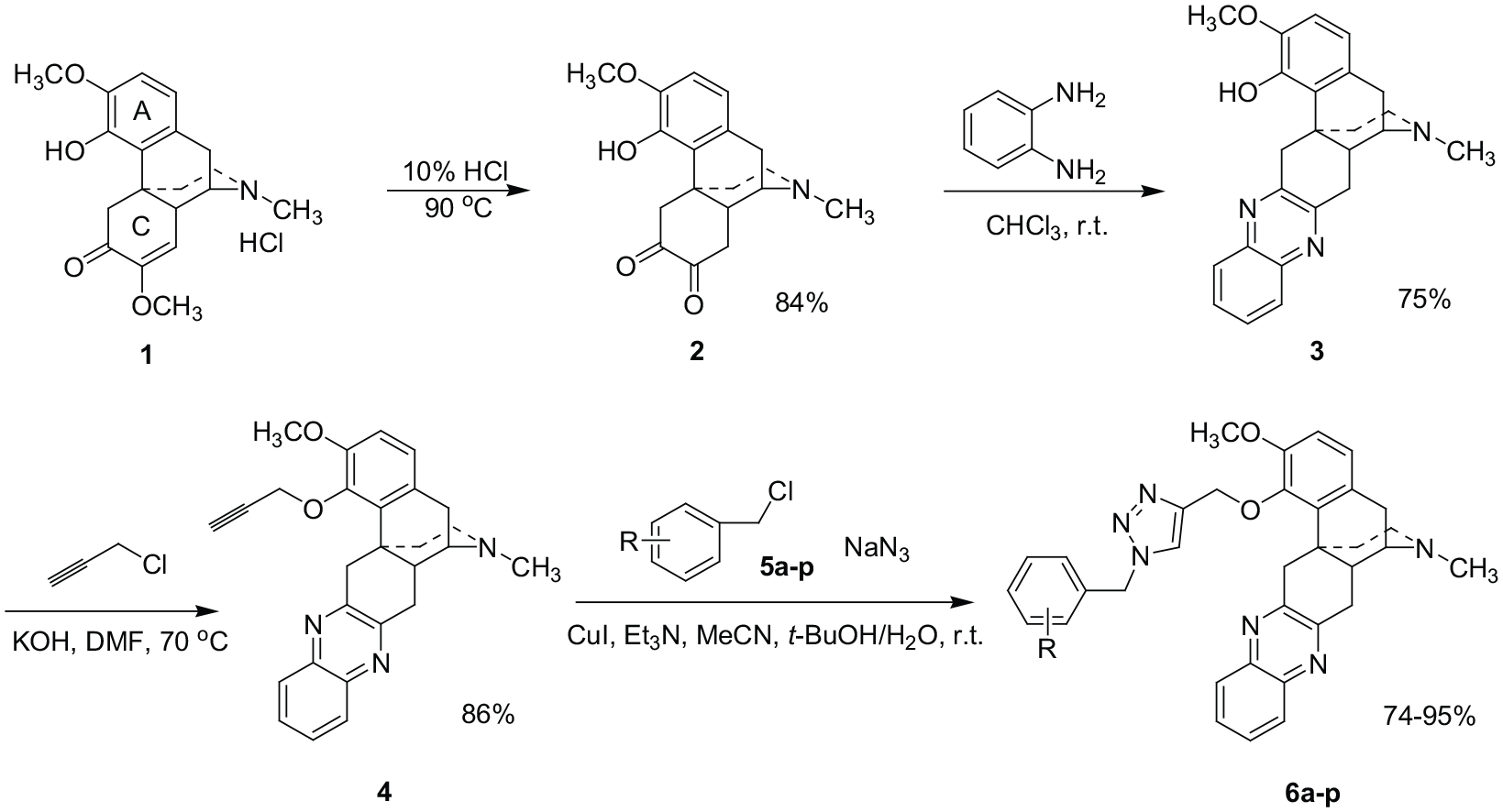
Synthesis of C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives.
Scheme 1 shows the functionalization of the C-ring with a quinoxaline and then A-ring modification with 1,2,3-triazole. We also tried to synthesize the target compounds using another method as shown in Scheme 2, which involves synthesizing the A-ring 1,2,3-triazole first and then the C-ring quinoxaline heterocycle. However, the yield was very low when reducing the C-ring to a 1,2-diketone structure with hydrochloric acid. Hence, the second method was not successful. This may be due to the steric hindrance of the previously synthesized 1,2,3-triazole ring which interferes with the formation of the 1,2-diketone.

An inefficient synthesis method of sinomenine 1,2,3-triazole derivatives.
We have synthesized 16 sinomenine derivatives (see Figure 1) using the method shown in Scheme 1 by using benzyl chlorides with different substituents. A wide range of derivatives was obtained with the benzyl moiety possessing ortho, meta, or para substituents, either electron-withdrawing or electron-donating. The derivatives were obtained in 74%–95% yields. The results show that this method is reliable for the synthesis of other sinomenine derivatives.

The prepared C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives.
Conclusion
In summary, we have developed a practical approach to synthesize novel C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives in good to excellent yields via click reactions. The sinomenine derivatives possess both a five-membered N-heterocycle and a six-membered N-heterocycle, thus enriching the library of sinomenine derivatives. At the same time, we also studied different synthetic methods toward such derivatives. All the products were characterized by 1H NMR, 13C NMR, high resolution mass spectrometry (HRMS), and Fourier-transform infrared spectrometry (FTIR). These new sinomenine derivatives may be beneficial for future drug research and development.
Experimental
Sinomenine and all the reagents are commercially available (Macklin and China National Pharmaceutical Group Corporation) and used directly without further purification. Reactions were monitored by the thin-layer chromatography (TLC) with GF254 silica gel plates, and products were purified by the column chromatography over brand 300–400 mesh silica gel with CH2Cl2/CH3OH/NH3·H2O (200:10:1–400:10:1 v/v) as eluents. Melting points were measured with a Jinke SGWX-4B melting point apparatus and are uncorrected. All NMR spectra were recorded on a Bruker AV-III 400 MHz NMR spectrometer using CDCl3 as the solvent and tetramethylsilane (TMS) as an internal standard. FTIR spectra were recorded on a Nicolet 6700 FTIR spectrometer. HRMS measurements were conducted with an Agilent model G6220 mass spectrometer. The optical rotations of six representative target products were recorded on a Rudolph A25829-T polarimeter in CH3OH. HRMS, FT-IR, 1H NMR, and 13C NMR spectroscopy, please see Supplemental Material.
Synthesis of the 1,2-diketone scaffold of sinomenine (2 )
Sinomenine hydrochloride (
Synthesis of C-ring quinoxaline-substituted sinomenine (3 )
The 1,2-diketone scaffold sinomenine (
Synthesis of alkynyl sinomenine (4 )
The C-ring quinoxaline-substituted sinomenine (
Synthesis of C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6a–p ); general procedure
Benzyl chloride (
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6a )
Brown powder; yield: 74%; m.p.: 87–89 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.22 (1H, s), 7.84 (1H, m), 7.73 (1H, m), 7.58 (2H, m), 7.45-7.38 (5H, m), 6.90 (1H, d, J = 8.4 Hz), 6.68 (1H, d, J = 8.4 Hz), 5.65 (2H, dd, J = 31.6, 14.8 Hz), 5.18 (2H, dd, J = 17.6, 11.2 Hz), 4.86 (1H, d, J = 17.2 Hz), 3.67 (3H, s), 3.23-2.93 (6H, m), 2.58 (2H, m), 2.48 (3H, s), 2.21-2.14 (1H, m), 2.00-1.91 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.5, 153.0, 151.2, 146.6, 145.7, 141.3, 141.2, 134.9, 130.7, 130.4, 129.1, 128.8, 128.7, 128.6, 128.5, 128.2, 128.1, 123.7, 123.6, 111.1, 65.6, 56.4, 55.6, 54.3, 46.7, 44.9, 42.9, 42.8, 39.5, 36.5, 33.5, 23.6; IR (υmax, cm−1): 2909, 1602, 1575, 1480, 1435, 1334, 1278, 1213, 1150, 1053, 763; HRMS: m/z calcd for C34H35N6O2 [M+H]+: 559.2821; found: 559.2817.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6b )
Brown powder; yield: 90%; m.p.: 85–86 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.27 (1H, s), 7.85 (1H, m), 7.80 (1H, m), 7.58 (2H, m), 7.47 (1H, m), 7.35-7.28 (3H, m), 6.84 (1H, d, J = 8.4 Hz), 6.67 (1H, d, J = 8.4 Hz), 5.78 (2H, s), 5.20 (2H, dd, J = 15.6, 11.6 Hz), 4.87 (1H, d, J = 17.2 Hz), 3.69 (3H, s), 3.22-2.90 (6H, m), 2.52 (2H, m), 2.46 (3H, s), 2.17-2.10 (1H, m), 2.01-1.89 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.1, 146.6, 145.6, 141.3, 141.2, 133.5, 132.8, 130.7, 130.6, 130.4, 130.1, 129.9, 128.8, 128.6, 128.5, 128.1, 127.6, 124.0, 123.7, 111.0, 65.6, 56.3, 55.5, 51.5, 46.6, 44.9, 42.9, 42.8, 39.5, 36.5, 33.5, 23.6; IR (υmax, cm−1): 2906, 1595, 1573, 1474, 1433, 1333, 1271, 1208, 1152, 1048, 755; HRMS: m/z calcd for C34H34N6O2Cl [M+H]+: 593.2432; found: 593.2433.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6c )
Brown powder; yield: 90%; m.p.: 98–100 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.27 (1H, s), 7.85 (1H, m), 7.76 (1H, m), 7.59 (2H, m), 7.38-7.32 (3H, m), 7.26 (1H, s), 6.85 (1H, d, J = 8.4 Hz), 6.68 (1H, d, J = 8.4 Hz), 5.63 (2H, q, J = 15.2 Hz), 5.19 (2H, dd, J = 21.2, 11.6 Hz), 4.86 (1H, d, J = 17.6 Hz), 3.68 (3H, s), 3.23-2.91 (6H, m), 2.56 (2H, m), 2.47 (3H, s), 2.20-2.13 (1H, m), 2.00-1.88 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.1, 146.6, 145.6, 141.3, 141.2, 133.5, 132.8, 130.7, 130.6, 130.4, 130.1, 129.9, 128.8, 128.6, 128.5, 128.1, 127.6, 124.0, 123.7, 111.0, 65.6, 56.3, 55.5, 51.5, 46.6, 44.9, 42.9, 42.8, 39.5, 36.5, 33.5, 23.6; IR (υmax, cm−1): 2907, 1602, 1575, 1480, 1435, 1334, 1275, 1209, 1150, 1049, 763; HRMS: m/z calcd for C34H34N6O2Cl [M+H]+: 593.2432; found: 593.2429.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6d )
Brown powder; yield: 91%; m.p.: 103–104 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.19 (1H, s), 7.83 (2H, m), 7.58 (2H, m), 7.43 (2H, m), 7.32-7.28 (1H, m), 6.82 (1H, d, J = 8.4 Hz), 6.66 (1H, d, J = 8.4 Hz), 5.96 (2H, dd, J = 17.6, 14.4 Hz), 5.17 (2H, dd, J = 15.2, 11.6 Hz), 4.87 (1H, d, J = 17.2 Hz), 3.67 (3H, s), 3.21-2.89 (6H, m), 2.52 (2H, m), 2.45 (3H, s), 2.14-2.07 (1H, m), 1.99-1.83 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.7, 153.1, 151.0, 146.7, 145.1, 141.2, 136.9, 131.0, 130.8, 130.7, 130.4, 128.9, 128.8, 128.6, 128.5, 128.0, 123.6, 123.5, 111.0, 65.6, 56.2, 55.5, 49.1, 46.6, 44.8, 43.0, 42.9, 39.5, 36.5, 33.6, 23.5; IR (υmax, cm−1): 2906, 1581, 1566, 1477, 1435, 1335, 1277, 1209, 1151, 1049, 760; HRMS: m/z calcd for C34H33N6O2Cl2 [M+H]+: 627.2042; found: 627.2038.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6e )
Brown powder; yield: 90%; m.p.: 108–110 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.29 (1H, s), 7.86 (1H, m), 7.76 (1H, m), 7.62-7.50 (4H, m), 7.33-7.28 (2H, m), 6.85 (1H, d, J = 8.4 Hz), 6.68 (1H, d, J = 8.4 Hz), 5.63 (2H, q, J = 15.2 Hz), 5.21 (2H, dd, J = 21.6, 11.6 Hz), 4.85 (1H, d, J = 17.6 Hz), 3.68 (3H, s), 3.23-2.90 (6H, m), 2.54 (2H, m), 2.47 (3H, s), 2.19-2.12 (1H, m), 2.00-1.89 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.1, 146.5, 146.0, 141.3, 141.1, 137.2, 131.8, 131.0, 130.8, 130.7, 130.6, 128.8, 128.7, 128.4, 128.1, 126.7, 123.8, 123.7, 123.1, 111.0, 65.6, 56.2, 55.6, 53.5, 46.6, 44.9, 43.0, 42.9, 39.7, 36.5, 33.5, 23.6; IR (υmax, cm−1): 2904, 1597, 1572, 1477, 1432, 1334, 1275, 1210, 1151, 1048, 760; HRMS: m/z calcd for C34H34N6O2Br [M+H]+: 637.1927; found: 637.1930.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6f )
Brown powder; yield: 92%; m.p.: 102–104 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.27 (1H, s), 7.90-7.85 (2H, m), 7.60 (2H, m), 7.42-7.35 (1H, m), 7.01 (2H, m), 6.83 (1H, d, J = 8.4 Hz), 6.67 (1H, d, J = 8.4 Hz), 5.74 (2H, dd, J = 17.6, 14.4 Hz), 5.17 (2H, dd, J = 18.4, 11.6 Hz), 4.87 (1H, d, J = 17.6 Hz), 3.68 (3H, s), 3.22-2.90 (6H, m), 2.52 (2H, m), 2.46 (3H, s), 2.16-2.09 (1H, m), 1.99-1.86 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 162.8, 162.7, 160.3, 160.2, 153.7, 153.1, 151.1, 146.7, 145.6, 141.3, 131.5, 131.4, 131.2, 130.8, 130.7, 128.8, 128.6, 128.1, 123.7, 123.6, 112.0, 111.9, 111.8, 111.7, 111.3, 111.1, 111.0, 110.9, 65.6, 56.3, 55.5, 46.6, 44.9, 43.0, 42.9, 41.5, 41.4, 41.3, 39.6, 36.5, 33.6, 23.6; IR (υmax, cm−1): 2906, 1625, 1595, 1473, 1434, 1334, 1274, 1231, 1153, 1047, 763; HRMS: m/z calcd for C34H33N6O2F2 [M+H]+: 595.2633; found: 595.2630. (13C data assignment for this compound, please see Supplemental Material.)
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6g )
Brown powder; yield: 95%; m.p.: 89–91 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.29 (1H, s), 7.86 (2H, m), 7.60 (2H, m), 7.24-7.11 (3H, m), 6.84 (1H, d, J = 8.4 Hz), 6.68 (1H, d, J = 8.4 Hz), 5.73 (2H, d, J = 1.2 Hz), 5.19 (2H, dd, J = 26.4, 11.6 Hz), 4.86 (1H, d, J = 17.2 Hz), 3.68 (3H, s), 3.23-2.90 (6H, m), 2.53 (2H, m), 2.46 (3H, s), 2.17-2.10 (1H, m), 2.00-1.87 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.8, 151.7, 151.0, 149.9, 149.8, 149.3, 149.2, 147.5, 147.3, 146.5, 145.9, 141.3, 141.2, 130.8, 130.7, 128.8, 128.7, 128.5, 128.1, 125.2, 125.2, 125.2, 125.2, 124.9, 124.9, 124.8, 124.8, 124.6, 124.5, 123.8, 123.7, 118.0, 117.8, 111.0, 65.5, 56.2, 55.5, 47.2, 47.2, 47.2, 46.6, 45.0, 43.0, 42.9, 39.7, 36.5, 33.5, 23.5; IR (υmax, cm−1): 2904, 1629, 1600, 1476, 1438, 1335, 1278, 1215, 1150, 1052, 763; HRMS: m/z calcd for C34H33N6O2F2 [M+H]+: 595.2633; found: 595.2628.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6h )
Brown powder; yield: 85%; m.p.: 87–88 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.26 (1H, s), 7.86 (1H, m), 7.75 (1H, m), 7.59 (2H, m), 7.24-7.18 (2H, m), 7.13 (1H, m), 6.85 (1H, d, J = 8.4 Hz), 6.68 (1H, d, J = 8.4 Hz), 5.61 (2H, dd, J = 29.2, 15.2 Hz), 5.20 (2H, dd, J = 27.6, 11.6 Hz), 4.85 (1H, d, J = 17.6 Hz), 3.68 (3H, s), 3.23-2.89 (6H, m), 2.54 (2H, m), 2.46 (3H, s), 2.18-2.11 (1H, m), 2.00-1.88 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.2, 151.8, 151.8, 151.7, 151.6, 151.0, 149.3, 149.3, 149.2, 149.1, 146.5, 146.0, 141.3, 141.1, 132.0, 132.0, 132.0, 131.9, 130.8, 130.7, 128.9, 128.7, 128.3, 128.1, 124.3, 124.3, 124.3, 124.2, 123.8, 123.6, 118.0, 117.9, 117.3, 117.2, 111.0, 65.6, 56.2, 55.5, 53.0, 46.5, 44.9, 42.9, 42.8, 39.8, 36.5, 33.5, 23.5; IR (υmax, cm−1): 2907, 1600, 1587, 1480, 1435, 1334, 1264, 1213, 1153, 1048, 763; HRMS: m/z calcd for C34H33N6O2F2 [M+H]+: 595.2633; found: 595.2631.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6i )
Brown powder; yield: 86%; m.p.: 90–91 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.24 (1H, s), 7.85 (1H, m), 7.75 (1H, m), 7.61-7.54 (2H, m), 7.35-7.31 (1H, m), 6.97 (1H, m), 6.92 (2H, m), 6.84 (1H, d, J = 8.4 Hz), 6.67 (1H, d, J = 8.4 Hz), 5.61 (2H, dd, J = 32, 14.8 Hz), 5.18 (2H, dd, J = 16.4, 12 Hz), 4.86 (1H, d, J = 17.6 Hz), 3.80 (3H, s), 3.68 (3H, s), 3.22-2.91 (6H, m), 2.55 (2H, m), 2.47 (3H, s), 2.19-2.12 (1H, m), 2.00-1.90 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 160.1, 153.6, 153.1, 151.1, 146.6, 145.7, 141.3, 141.2, 136.4, 130.8, 130.6, 130.2, 128.8, 128.6, 128.5, 128.1, 123.7, 120.3, 114.2, 113.7, 111.1, 65.6, 56.3, 55.5, 55.3, 54.2, 46.6, 44.9, 42.9, 42.8, 39.6, 36.5, 33.5, 23.6; IR (υmax, cm−1): 2903, 1613, 1571, 1477, 1518, 1477, 1437, 1335, 1274, 1210, 1152, 758; HRMS: m/z calcd for C35H37N6O3 [M+H]+: 589.2927; found: 589.2924.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6j )
Brown powder; yield: 85%; m.p.: 92–93 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.19 (1H, s), 7.84 (1H, m), 7.72 (1H, m), 7.61-7.53 (2H, m), 7.35-7.33 (2H, m), 6.94 (2H, d, J = 8 Hz), 6.83 (1H, d, J = 8.4 Hz), 6.67 (1H, d, J = 8.4 Hz), 5.57 (2H, dd, J = 35.2, 14.8 Hz), 5.17 (2H, dd, J = 17.2, 11.6 Hz), 4.86 (1H, d, J = 17.2 Hz), 3.81 (3H, s), 3.67 (3H, s), 3.21-2.88 (6H, m), 2.52 (2H, m), 2.45 (3H, s), 2.17-2.10 (1H, m), 1.99-1.85 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 159.9, 153.6, 153.1, 151.1, 146.6, 145.5, 141.2, 141.1, 130.8, 130.6, 129.7, 128.8, 128.6, 128.5, 128.0, 126.9, 123.7, 123.5, 114.4, 111.1, 65.6, 56.2, 55.5, 55.3, 53.7, 46.6, 44.9, 42.9, 42.8, 39.6, 36.5, 33.5, 23.5 ppm; IR (υmax, cm−1): 2904, 1616, 1587, 1477, 1513, 1477, 1437, 1335, 1277, 1248, 1175, 1156, 1052, 1034, 764; HRMS: m/z calcd for C35H37N6O3 [M+H]+: 589.2927; found: 589.2929.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6k )
Brown powder; yield: 89%; m.p.: 99–100 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 7.96 (1H, s), 7.84 (1H, m), 7.63-7.52 (3H, m), 7.24 (1H, m), 7.15 (2H, d, J = 7.6 Hz), 6.82 (1H, d, J = 8.4 Hz), 6.64 (1H, d, J = 8.4 Hz), 5.70 (2H, q, J = 14.8 Hz), 5.12 (2H, dd, J = 28, 11.6 Hz), 4.84 (1H, d, J = 17.2 Hz), 3.64 (3H, s), 3.20-2.89 (6H, m), 2.52 (2H, m), 2.45 (3H, s), 2.43 (6H, s), 2.15-2.08 (1H, m), 1.99-1.82 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.1, 146.7, 145.0, 141.2, 138.2, 130.8, 130.7, 130.7, 129.1, 129.0, 128.7, 128.6, 128.5, 128.0, 123.6, 123.1, 111.0, 65.6, 56.2, 55.5, 48.5, 46.6, 44.7, 43.0, 42.9, 39.6, 36.5, 33.6, 23.6, 19.9; IR (υmax, cm−1): 2904, 1595, 1573, 1477, 1435, 1334, 1278, 1212, 1153, 1048, 764; HRMS: m/z calcd for C36H39N6O2 [M+H]+: 587.3134; found: 587.3133.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6l )
Brown powder; yield: 85%; m.p.: 110–112 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.30 (1H, s), 7.86 (1H, m), 7.71 (3H, m), 7.63-7.55 (2H, m), 7.46 (2H, m), 6.86 (1H, d, J = 8.4 Hz), 6.68 (1H, d, J = 8.4 Hz), 5.73 (2H, dd, J = 26.8, 15.6 Hz), 5.20 (2H, dd, J = 35.6, 11.6 Hz), 4.84 (1H, d, J = 17.2 Hz), 3.69 (3H, s), 3.23-2.89 (6H, m), 2.54 (2H, m), 2.46 (3H, s), 2.19-2.12 (1H, m), 2.01-1.88 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.2, 151.1, 146.5, 146.2, 141.3, 141.1, 140.2, 132.8, 130.9, 130.8, 128.8, 128.6, 128.5, 128.2, 128.2, 123.8, 118.2, 112.6, 111.1, 65.6, 56.2, 55.5, 53.4, 46.6, 45.0, 43.0, 42.9, 39.8, 36.6, 33.6, 23.6; IR (υmax, cm−1): 2904, 2231, 1610, 1572, 1480, 1434, 1334, 1277, 1207, 1153, 1049, 763; HRMS: m/z calcd for C35H34N7O2 [M+H]+: 584.2774; found: 584.2772.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6m )
Brown powder; yield: 95%; m.p.: 86–88 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.10 (1H, s), 7.84 (1H, m), 7.68 (1H, m), 7.60-7.52 (2H, m), 7.34-7.28 (4H, m), 6.83 (1H, d, J = 8.4 Hz), 6.66 (1H, d, J = 8.4 Hz), 5.65 (2H, dd, J = 35.2, 14.8 Hz), 5.16 (2H, dd, J = 13.6, 11.6 Hz), 4.86 (1H, d, J = 17.6 Hz), 3.66 (3H, s), 3.21-2.88 (6H, m), 2.51 (2H, m), 2.45 (3H, s), 2.36 (3H, s), 2.16-2.09 (1H, m), 2.01-1.84 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.0, 146.6, 145.3, 141.2, 141.1, 137.0, 132.8, 131.0, 130.8, 130.7, 129.5, 129.0, 128.8, 128.6, 128.5, 128.0, 126.6, 123.7, 123.6, 111.1, 65.6, 56.2, 55.5, 52.4, 46.6, 44.8, 42.9, 42.8, 39.6, 36.5, 33.5, 23.5, 19.1; IR (υmax, cm−1): 2907, 1601, 1572, 1477, 1435, 1334, 1277, 1215, 1153, 1048, 760; HRMS: m/z calcd for C35H37N6O2 [M+H]+: 573.2978; found: 573.2984.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6n )
Brown powder; yield: 95%; m.p.: 82–84 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.24 (1H, s), 7.85 (1H, m), 7.73 (1H, m), 7.61-7.53 (2H, m), 7.32-7.29 (1H, m), 7.19 (3H, m), 6.84 (1H, d, J = 8.4 Hz), 6.67 (1H, d, J = 8.4 Hz), 5.61 (2H, dd, J = 33.6, 14.8 Hz), 5.18 (2H, dd, J = 16, 11.2 Hz), 4.87 (1H, d, J = 17.2 Hz), 3.67 (3H, s), 3.22-2.91 (6H, m), 2.54 (2H, m), 2.47 (3H, s), 2.36 (3H, s), 2.19-2.12 (1H, m), 2.00-1.90 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.2, 146.7, 145.7, 141.3, 141.2, 138.9, 134.9, 130.8, 130.7, 129.4, 129.0, 128.9, 128.8, 128.6, 128.5, 128.1, 125.3, 123.7, 123.6, 111.2, 65.7, 56.3, 55.6, 54.3, 46.6, 44.9, 43.0, 42.8, 39.6, 36.6, 33.6, 23.6, 21.4; IR (υmax, cm−1): 2904, 1611, 1572, 1476, 1437, 1334, 1277, 1210, 1151, 1050, 762; HRMS: m/z calcd for C35H37N6O2 [M+H]+: 573.2978; found: 573.2971.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6o )
Brown powder; yield: 89%; m.p.: 87–88 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.20 (1H, s), 7.85 (1H, m), 7.73 (1H, m), 7.61-7.53 (2H, m), 7.29 (2H, m), 7.22 (2H, m), 6.83 (1H, d, J = 8.4 Hz), 6.67 (1H, d, J = 8.4 Hz), 5.60 (2H, dd, J = 32.8, 14.8 Hz), 5.17 (2H, dd, J = 16.8, 11.6 Hz), 4.86 (1H, d, J = 16.8 Hz), 3.67 (3H, s), 3.21-2.89 (6H, m), 2.52 (2H, m), 2.46 (3H, s), 2.37 (3H, s), 2.17-2.10 (1H, m), 1.99-1.89 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.1, 151.1, 146.7, 145.6, 141.3, 141.2, 138.6, 131.9, 130.8, 130.6, 129.8, 128.8, 128.6, 128.5, 128.2, 128.1, 123.7, 123.5, 111.1, 65.6, 56.3, 55.5, 54.1, 46.6, 44.9, 42.9, 42.8, 39.7, 36.5, 33.6, 23.6, 21.2; IR (υmax, cm−1): 2906, 1601, 1572, 1480, 1435, 1335, 1277, 1210, 1151, 1050, 763; HRMS: m/z calcd for C35H37N6O2 [M+H]+: 573.2978; found: 573.2983.
C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives (6p )
Brown powder; yield: 80%; m.p.: 97–99 °C; 1H NMR (400 MHz; CDCl3; TMS): δ 8.29 (1H, s), 7.85 (1H, m), 7.69 (3H, m), 7.61-7.50 (2H, m), 7.49 (2H, m), 6.85 (1H, d, J = 8.4 Hz), 6.68 (1H, d, J = 8.4 Hz), 5.72 (2H, dd, J = 33.2, 15.2 Hz), 5.20 (2H, dd, J = 22.8, 11.6 Hz), 4.85 (1H, d, J = 17.2 Hz), 3.68 (3H, s), 3.22-2.90 (6H, m), 2.54 (2H, m), 2.46 (3H, s), 2.19-2.12 (1H, m), 2.01-1.91 (2H, m); 13C NMR (100 MHz; CDCl3; TMS): δ 153.6, 153.2, 151.1, 146.5, 146.2, 141.3, 141.2, 139.0, 130.8, 130.8, 128.9, 128.6, 128.3, 128.2, 126.2, 126.1, 126.1, 126.1, 123.8, 123.7, 111.0, 65.6, 56.2, 55.5, 53.6, 46.6, 45.0, 43.1, 42.9, 39.8, 36.6, 33.6, 23.6; IR (υmax, cm−1): 2904, 1622, 1573, 1479, 1439, 1421, 1323, 1277, 1210, 1123, 1066, 763; HRMS: m/z calcd for C35H34N6O3F3 [M+H]+: 643.2644; found: 643.2647.
Supplemental Material
3.Supplementary_information-2 – Supplemental material for Design and synthesis of C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives via click reactions
Supplemental material, 3.Supplementary_information-2 for Design and synthesis of C-ring quinoxaline-substituted sinomenine 1,2,3-triazole derivatives via click reactions by Tong Lu, Ling Dong, Hongmei Pan, Xuedan Wu, Xia Chen, Chengwen Gu, Naili Tao, Ao Wang, Kehua Zhang and Jie Jin in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Anhui University Natural Science Research Project (KJ2019A0770 and KJ2019JD19) and the Science and Technology Innovation Foundation for College Students (C19063, C19263, C19546, and C19559).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
