Abstract
Two novel mononuclear Zn(II) complexes, [ZnL1Cl2] (

Introduction
With three nitrogen coordination sites, terpyridines have attracted widespread attention because of their excellent complexing properties as N-donor ligands toward a wide range of transition and rare earth metal ions. 1 The terpyridine-based complexes play a prominent role in research owing to their interesting and diverse properties. They have been investigated for their potential applications in materials research, polymer science, nanotechnology, catalysis, medicine, and light-to-energy conversion.2–8
Among the various terpyridine ligands, a few coumarin-contained terpyridines and/or their complexes have been reported to exhibit prominent properties and are used as fluorescent chemosensors or molecular energy transfer switch.9–11 In recent years, we have been engaging in the synthesis of novel coumarin derivatives and investigation of their fluorescent properties.12–15 As a part of the foregoing research in our group, we present herein the concise and efficient synthesis of two novel coumarin-based terpyridine derivatives, 3-(4-(2,2′:6′,2″-tripyridyl-4′-)styryl)-7-diethylaminocoumarin (L1) and 3-(4-(2,2′:6′,2″-tripyridyl-4′-)phenylimine)-7-diethylaminocoumarin (L2). By using the above ligands, two corresponding Zn(II) complexes were obtained and characterized by infrared (IR) spectroscopy, elemental analysis, and structurally analyzed by X-ray single-crystal diffraction. Moreover, the absorption and emission spectra of the synthesized ligands and complexes in dichloromethane were measured.
Results and discussion
Synthesis of ligands L1 and L2
The synthesis of the two ligands L1 and L2 is outlined in
Scheme 1
. The 3-formylcoumarin

Synthesis routes of L1 and L2.
Crystal structures of complexes 1 and 2
The single-crystal structures of complexes

Molecular structure of complex
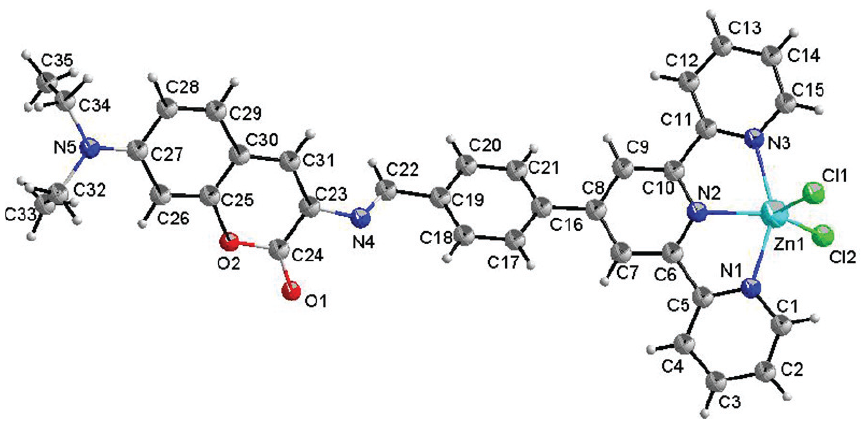
Molecular structure of complex
Crystal data and structure refinement for complexes

Selected bond lengths (Å) and bond angles (°) for complexes
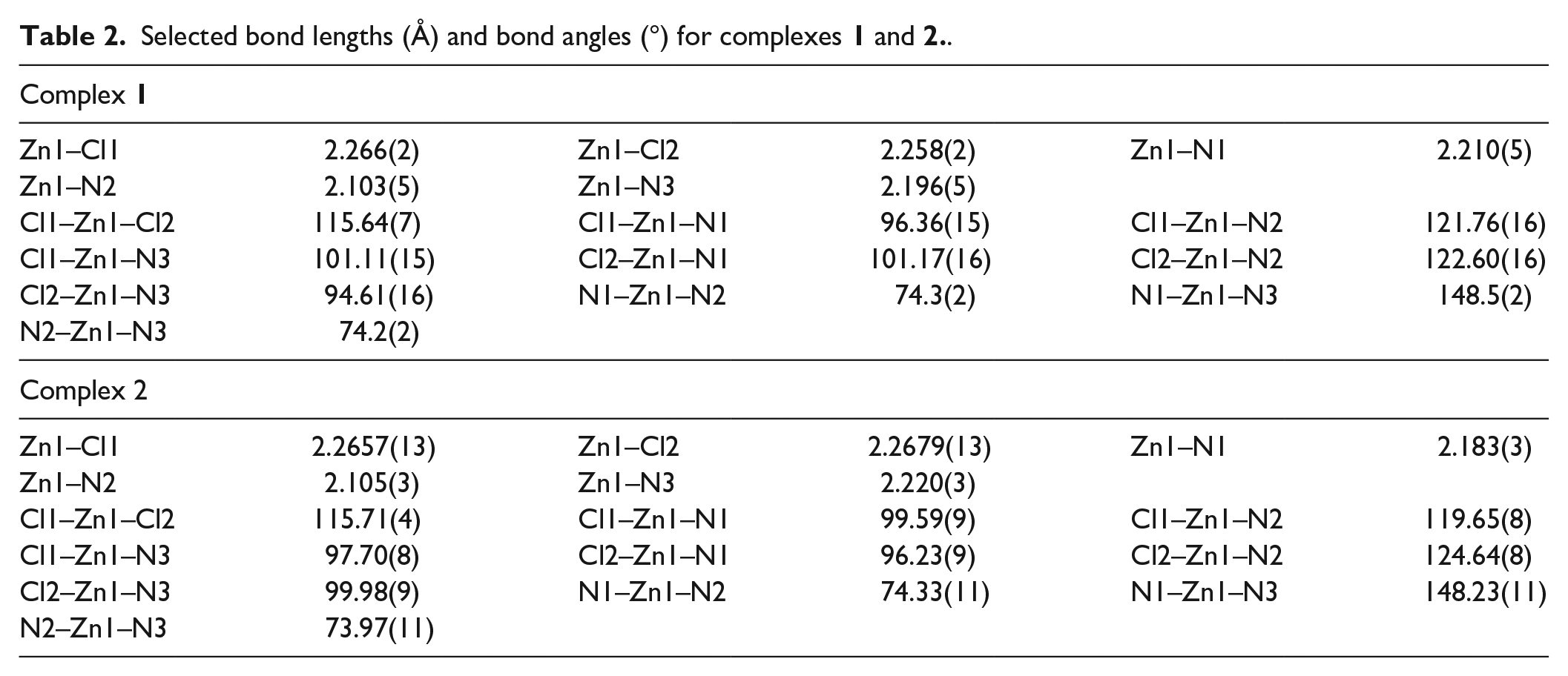
In complex
In complex
Absorption and fluorescence spectra of the ligands and the Zn(II) complexes
The UV–Vis absorption spectra of the free ligands (L1 and L2) and the corresponding complexes (

Absorption spectra of L1 and complex

Absorption spectra of L2 and complex
As shown in Figure 4, the ligand L2 shows three absorption peaks at 251, 288, and 442 nm. Compared with the absorption peaks of the free ligand, the absorptions at 251 and 288 nm are blue-shifted to 241 and 286 nm, while the absorption at 442 nm is red-shifted to 451 nm in the absorption spectrum of complex
The fluorescence emission spectra of the dilute dichloromethane solutions of the L1, complex

Emission spectra of L1 and complex

Emission spectra of L2 and complex
Conclusion
In summary, two novel coumarin-based terpyridine ligands and their corresponding mononuclear Zn(II) complexes have been synthesized and characterized by spectroscopic methods and single-crystal X-ray diffraction. In the two complexes, Zn2+ is five coordinated with three nitrogen atoms from the terpyridine skeleton and two chloride atoms, resulting in the Zn atom being located at the distorted trigonal bipyramidal center. The fluorescence emissions of the complexes are significantly red shifted from those of the corresponding ligands mainly due to the coordination of the metal with the ligands. Applicability of these coumarin-based terpyridine ligands and their Zn(II) complexes is yet to be established.
Experimental
Materials and general methods
All chemicals and solvents were analytical reagent grade, commercially available and used without further purification. Elemental analyses (C, H and N) were performed on a GmbH VariuoEL V3.00 automatic elemental analyzer. 1H NMR and 13C NMR spectra were recorded using a Bruker Avance DRX-400 MHz or a Bruker Avance III 500 MHz spectrometer. Melting points were determined by a microscope apparatus and are uncorrected. IR spectra were recorded on a Digilab FTS-3000 FTIR spectrophotometer. UV–Vis absorption and fluorescence spectra were recorded on a Hitachi U-3900H spectrometer and on a Hitachi F7000 FL spectrophotometer, respectively. X-ray single-crystal diffraction measurements were made on a Bruker X8 APEX diffractometer working with graphite monochromated Mo Kα radiation.
Synthesis of L1
A mixture of compound
Synthesis of L2
Compound
Synthesis of complex 1
Zinc(II) chloride (4.1 mg, 0.03 mmol) was dissolved in anhydrous ethanol (2 mL), L1 (16.5 mg, 0.03 mmol) was dissolved in dichloromethane (6 mL). The two portions were mixed in a 25-mL Teflon-lined stainless steel autoclave. The autoclave was heated to 130 °C in an oven and kept there for 3 days and then allowed to cool. When the autoclave reached room temperature, yellow block crystals were obtained and used to collect the single-crystal X-ray data. Yield 41% based on ZnCl2. IR (KBr, cm−1): 1706, 1637, 1594, 1356, 1185, 1121, 799, 768, 525, 451. Anal. calcd for C36H30Cl2N4O2Zn: C, 62.95; H, 4.40; N, 8.16; found: C, 62.90; H, 4.46; N, 8.13%.
Synthesis of complex 2
Zinc(II) chloride (5.4 mg, 0.04 mmol) was dissolved in anhydrous ethanol (2 mL), L2 (22.0 mg, 0.04 mmol) was dissolved in trichloromethane (6 mL). The two portions were mixed in a 25-mL Teflon-lined stainless steel autoclave. The autoclave was heated to 130 °C in an oven and kept there for 3 days and then allowed to cool. When the autoclave reached room temperature, dark brown block crystals were obtained and used to collect the single-crystal X-ray data. Yield 30% based on ZnCl2. IR (KBr, cm−1): 1708, 1632, 1603, 1413, 1258, 1125, 796, 744, 518, 462. Anal. calcd for C35H29Cl2N5O2Zn: C, 61.11; H, 4.25; N, 10.18; found: C, 61.15; H, 4.28; N, 10.13%.
Crystal structure determination
X-ray single-crystal diffraction data of complexes
Supplemental Material
supplemental_material – Supplemental material for Synthesis and characterization of novel coumarin-based terpyridine ligands and their Zn(II) complexes
Supplemental material, supplemental_material for Synthesis and characterization of novel coumarin-based terpyridine ligands and their Zn(II) complexes by Xiaolong Wang, Xian Yue and Long Deng in Journal of Chemical Research
Footnotes
Acknowledgements
We thank the Instrument Analysis Center of Lanzhou Jiaotong University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material is available online for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
