Abstract
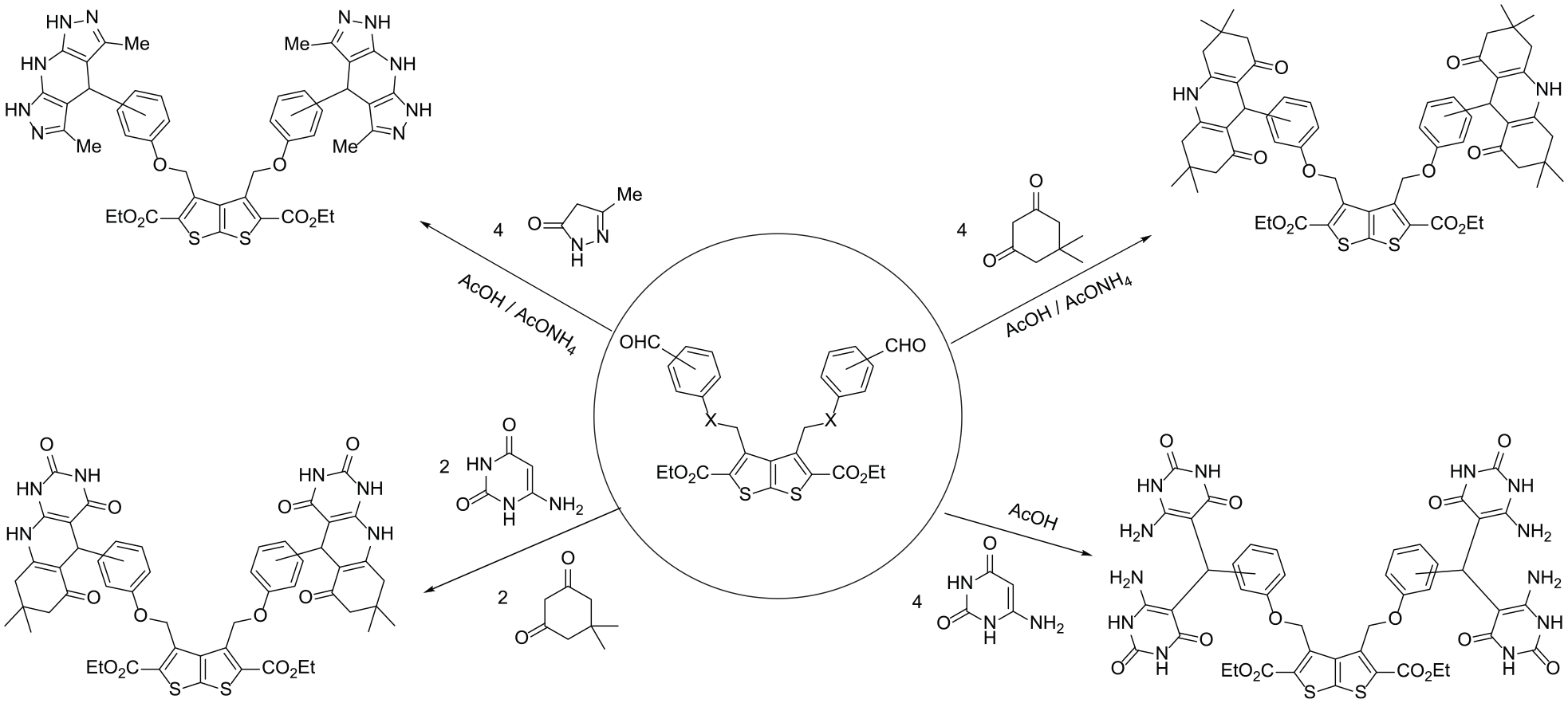
A novel series of bis(hexahydroacridine-1,8-diones), bis(tetrahydrodipyrazolo[3,4-b:4′,3′-e]pyridines), and bis(pyrimido[4,5-b]quinolines) incorporating a thieno[2,3-b]thiophene core via ether or ester linkages is prepared via a Hantzsch-like reaction.
Introduction
Multicomponent reactions represent a facile and rapid access to varieties of heterocyclic compounds. In addition, they have the advantages of atom-economy and selectivity.1–20 Moreover, 1,4-dihydropyridines (1,4-DHPs) have gained particular interest as they have major therapeutic effects in the treatment of cardiovascular diseases as a class of calcium-channel modulators. Also, they exhibit pharmacological activities that include antitumor,21–23 antioxidant,24–27 and antivasodilator. 28 Furthermore, acridines have attracted considerable attention due to their reported bioactivities.29–43 In conjunction with these findings, and in continuation of our recent project aimed at the synthesis of novel bis(heterocycles),44–55 we report herein on the one-pot synthesis of novel bis(acridines), bis(dipyrazolo[3,4-b:4′,3′-e]pyridines), and bis(pyrimido[4,5-b]quinolines) incorporating thieno[2,3-b]thiophenes through ether or ester bridges.
Results and discussion
Bis(aldehydes) linked to a thieno[2,3-b]thiophene core via a phenoxymethyl linker (
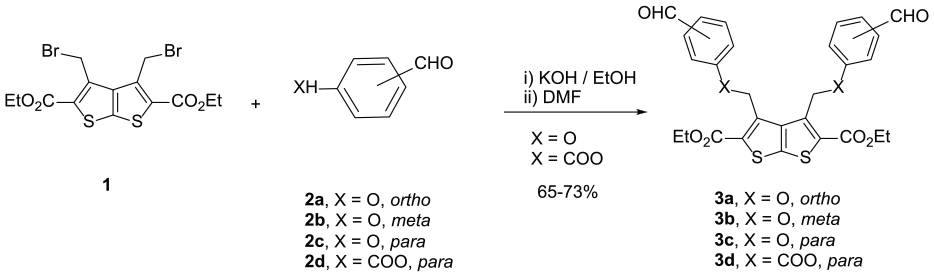
Synthesis of the bis(aldehydes)
The reactivity of bis(aldehydes) (

Synthesis of the bis(hexahydroacridine-1,8-diones)
In a similar manner, bis(tetrahydrodipyrazolo[3,4-b:4’,3’-e]pyridines) (

Synthesis of bis(3,5-dimethyl-1,4,7,8-tetrahydrodipyrazolo[3,4-b:4′,3′-e]pyridin-4-yl)phenoxy)alkanes
On the contrary, investigations to synthesize the bis(decahydropyrido[2,3-d:6,5-d′] dipyrimidinetetraones (

Synthesis of tetrabis(6-aminopyrimidine-2,4-diones)
In an extension to this work to include the unsymmetric Hantzsch reaction, we investigated the reactivity of bis(aldehydes) toward both 6-aminouracil and dimedone. Thus, the reaction of bis(aldehydes) (

Synthesis of bis(pyrimido[4,5-b]quinolines)
Generally, the reaction mechanism for the formation of compounds
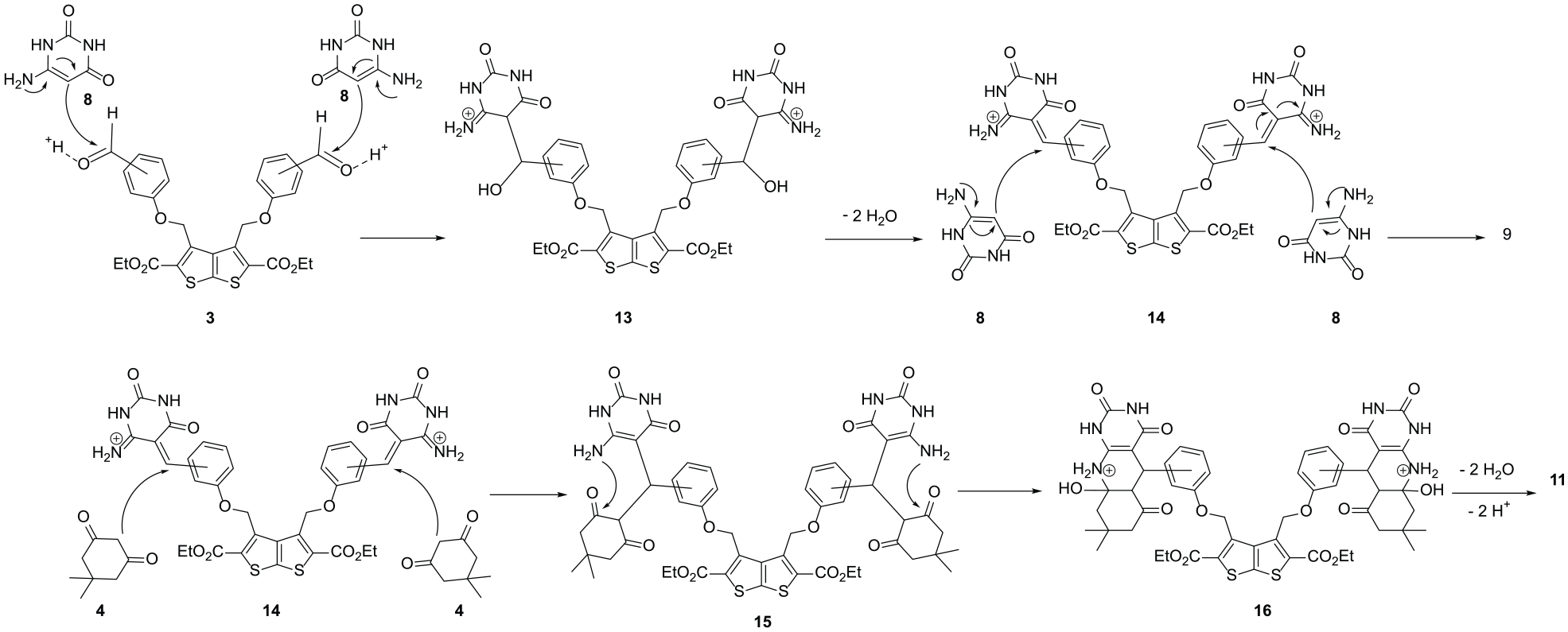
Suggested mechanism for the synthesis of tetrabis(6-aminopyrimidine-2,4-diones)
Conclusion
We have developed an efficient approach for the synthesis of novel series of bis(hexahydroacridine-1,8-diones), bis(tetrahydrodipyrazolo[3,4-b:4′,3′-e]pyridines), and bis(pyrimido[4,5-b]quinolines), which are linked to a thieno[2,3-b]thiophene core via ether or ester linkages. The target compounds were prepared under mild conditions and were obtained in good yields from inexpensive starting materials.
Experimental
General
Melting points were determined in open glass capillaries with a Gallenkamp apparatus. The IR spectra were recorded as potassium bromide disks on Pye Unicam SP 3-300 and Shimadzu FTIR 8101 PC IR spectrophotometers. NMR spectra were recorded with a Varian Mercury VXR-300 NMR spectrometer at 300 MHz ( 1 H NMR) and at 75 MHz ( 13 C NMR). Mass spectra (EI) were obtained at 70 eV with a Shimadzu GCMQP 1000 EX spectrometer. Analytical thin-layer chromatography was performed using precoated silica gel 60778 plates (Fluka), and the spots were visualized with ultraviolet (UV) light at 254 nm.
General procedure for the synthesis of compounds 5a–c
A mixture of bis(aldehyde) (
Diethyl 3,4-Bis((2-(3,3,6,6-tetramethyl-1,8-dioxo-1,2,3,4,5,6,7,8,9,10-decahydroacridin-9yl)phenoxy)methyl)thieno[2,3-b]thiophene-2,5-dicarboxylate (5a )
Yield:(550 mg (53%), AcOH); Colorless crystals; m.p. >300 °C. IR (KBr): 3376 (NH), 1720 (CO), 1635 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 0.81 (s, 12H, 4 CH3), 0.89 (s, 12H, 4 CH3), 1.23 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 1.86–2.02 (m, 16H, 8 CH2), 4.30 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 4.87 (s, 2H, acridine-H9), 5.38 (s, 4H, 2 OCH2), 6.15–7.15 (m, 8H, ArH), 8.44 (s, 2H, 2 NH); MS (ESI): m/z 1039 [M + 1]+. Anal. calcd for C60H66N2O10S2: C, 69.34; H, 6.40; N, 2.70; found: C, 69.55; H, 6.57; N, 2.67.
Diethyl 3,4-Bis((3-(3,3,6,6-tetramethyl-1,8-dioxo-1,2,3,4,5,6,7,8,9,10-decahydroacridin-9yl)phenoxy)methyl)thieno[2,3-b]thiophene-2,5-dicarboxylate (5b )
Yield:(582 mg (56%), Dioxane); Colorless crystals; m.p. >300 °C. IR (KBr): 3201 (NH), 1712 (CO), 1635 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 0.78 (s, 12H, 4 CH3), 0.96 (s, 12H, 4 CH3), 1.23 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 1.78-2.42 (m, 16H, 8 CH2), 4.27 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 4.77 (s, 2H, acridine-H9), 5.45 (s, 4H, 2 OCH2), 6.33–6.89 (m, 8H, ArH), 8.11 (s, 2H, 2 NH); 13C NMR (100 MHz, DMSO-d6): δ 14.4, 26.8, 29.5, 32.5, 40.7, 47.4, 50.6, 62.1, 111.9, 115.4, 118.8, 121.2, 125.5, 128.8, 134.7, 137.2, 149.1, 149.7, 148.8, 149.9, 157.6, 161.9, 165.8, 194.9. MS (ESI): m/z 1039 [M + 1]+. Anal. calcd for C60H66N2O10S2: C, 69.34; H, 6.40; N, 2.70; found: C, 69.53; H, 6.14; N, 2.62.
Diethyl 3,4-Bis((4-(3,3,6,6-tetramethyl-1,8-dioxo-1,2,3,4,5,6,7,8,9,10-decahydroacridin-9yl)phenoxy)methyl)thieno[2,3-b]thiophene-2,5-dicarboxylate (5c )
Yield:(581 mg (56%), AcOH); Colorless crystals; m.p. >300 °C. IR (KBr): 3379 (NH), 1720 (CO), 1635 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 0.86 (s, 12H, 4 CH3), 1.00 (s, 12H, 4 CH3), 1.25 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 1.97–2.49 (m, 16H, 8 CH2), 4.27 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 4.75 (s, 2H, acridine-H9), 5.57 (s, 4H, 2 OCH2), 6.70 (d, J = 8.1 Hz, 4H, ArH), 7.02 (d, J = 8.1 Hz, 4H, ArH), 9.21 (s, 2H, 2 NH); 13C NMR (75 MHz, DMSO-d6): δ 13.7, 26.4, 28.8, 31.6, 31.9, 50.1, 61.5, 111.4, 113.9, 120.7, 120.8, 128.3, 134.2, 136.9, 140.0, 149.1, 155.5, 161.1, 194.3. MS (ESI): m/z 1038 [M + 1]+. Anal. calcd for C60H66N2O10S2: C, 69.34; H, 6.40; N, 2.70; found: C, 69.58; H, 6.17; N, 2.68.
General procedure for the synthesis of compounds 7a–c
A mixture of bis(aldehyde)
Diethyl 3,4-Bis((2-(3,5-dimethyl-1,4,7,8-tetrahydrodipyrazolo[3,4-b:4′,3′-e]pyridin-4yl)phenoxy)methyl)thieno[2,3-b] (7a )
Yield: (436 mg, 50%); Colorless crystals; m.p. 295–297 °C. IR (KBr): 3387 (NH), 3217 (NH), 1712 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 1.26 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 1.53 (s, 12H, 4 CH3), 4.30 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 4.73 (s, 2H, dihydrpyridine-H4), 4.95 (s, 4H, 2 OCH2), 6.25–6.71 (m, 8H, ArH), 7.42 (s, 2H, 2 NH), 11.25 (br, 4H, 2 NH). MS (ESI): m/z 871 [M + 1]+. Anal. calcd for C44H42N10O6S2: C, 60.68; H, 4.86; N, 16.08; found: C, 60.84; H, 4.98; N, 16.20.
Diethyl 3,4-Bis((4-(3,5-dimethyl-1,4,7,8-tetrahydrodipyrazolo[3,4-b:4′,3′-e]pyridin-4yl)phenoxy)methyl)thieno[2,3-b] (7b )
Yield: (505 mg, 58%); Orange crystals; m.p. 264–269 °C. IR (KBr): 3857 (NH), 3101 (NH), 1705 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 1.27 (t, J = 7.2 Hz, 6H, CH3CH2), 2.03 (s, 12H, CH3), 3.57 (s, 2H, NH), 4.32 (q, J = 7.2 Hz, 4H, CH3 CH2), 4.74 (s, 2H, dihydrpyridine-H4), 5.64 (s, 4H, OCH2), 6.77 (d, J = 8.7 Hz, 4H, ArH), 6.98 (d, J = 8.7 Hz, 4H, ArH), 11.32 (br, 4H, NH); MS (ESI): m/z 871 [M + 1]+. Anal. calcd for C44H42N10O6S2: C, 60.68; H, 4.86; N, 16.08; found: C, 60.90; H, 5.01; N, 16.23.
Diethyl 3,4-Bis(((4-(3,5-dimethyl-4,7a-dihydrodipyrazolo[3,4-b:4′,3′-e]pyridin-4yl)benzoyl)oxy)methyl)thieno[2,3-b] (7c )
Yield: (572 mg, 62%); Colorless crystals; m.p. 248–251°C. IR (KBr): 3866 (NH), 3110 (NH), 1604 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 1.29 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 2.05 (s, 12H, 4 CH3), 3.45 (br, 6H, NH), 4.32 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 4.88 (s, 2H, dihydrpyridine-H4), 5.87 (s, 4H, 2 OCH2), 7.22 (d, J = 6.1 Hz, 4H, ArH), 7.78 (d, J = 6.1 Hz, 4H, ArH); 13C NMR (75 MHz, DMSO-d6): δ 10.8, 14.4, 21.5, 33.4, 40.8, 58.0, 62.1, 126.8, 128.4, 129.3, 135.3, 135.9, 145.4, 145.8, 150.0, 161.5, 165.7, 172.5. MS (ESI): m/z 922 [M + 1]+. Anal. calcd for C46H42N10O8S2: C, 59.60; H, 4.57; N, 15.11; found: C, 59.79; H, 4.28; N, 15.12.
General procedure for the synthesis of compounds 9a–c
A mixture of bis(aldehyde) (
Diethyl 3,4-Bis((3-(bis(6-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-5yl)methyl)phenoxy)methyl)thieno[2,3-b]thiophene-2,5-dicarboxylate (9a )
Yield: (717 mg, 70%); Colorless crystals; m.p. >300 °C. IR (KBr): 3356 (NH2), 3170 (NH), 1712 (CO), 1627 (CO), 1604 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 1.28 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 4.31 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 5.25 (s, 2H, 2 CH), 5.55 (s, 4H, 2 OCH2), 6.17–7.02 (m, 16H, ArH, and 4 NH2), 10.08 (brs, 4H, 4 NH), 10.46 (s, 4H, 4 NH). MS (ESI): m/z 1024 [M + 1]+. Anal. calcd for C44H40N12O14S2: C, 51.56; H, 3.93; N, 16.40; found: C, 51.69; H, 4.08; N, 16.56.
Diethyl3,4-Bis((4-(bis(6-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-5yl)methyl)phenoxy)methyl)thieno[2,3-b]thiophene-2,5-dicarboxylate (9b )
Yield: (768 mg, 75% yield); Yellow crystals; m.p. >300°C. IR (KBr): 3410 (NH2), 3170 (NH), 1712 (CO), 1627 (CO), 1504 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 1.24 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 4.30 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 5.22 (s, 2H, 2 CH), 5.65 (s, 4H, 2 OCH2), 6.68 (br, 8H, 4 NH2), 6.70–6.93 (m, 8H, ArH), 10.25 (br, 4H, 4 NH), 10.39 (s, 4H, 4 NH); MS (ESI): m/z 1024 [M + 1]+. Anal. calcd for C44H40N12O14S2: C, 51.56; H, 3.93; N, 16.40; found: C, 51.72; H, 4.09; N, 16.55.
Diethyl 3,4-Bis(((4-(bis(6-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-5yl)methyl)benzoyl)oxy)methyl)thieno[2,3-b]thiophene-2,5-dicarboxylate (9c )
Yield: (864 mg, 80% yield); Colorless crystals; m.p. >300°C. IR (KBr): 3410 (NH2), 3194 (NH), 1720 (CO), 1627 (CO), 1527 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 1.30 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 4.34 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 5.33 (s, 2H, 2 CH), 5.89 (s, 4H, 2 OCH2), 6.68 (br, 8H, 4 NH2), 7.18 (d, J = 6.1 Hz, 4H, ArH), 7.77 (d, J = 6.1 Hz, 4H, ArH), 10.33 (br, 4H, 4 NH), 10.53 (s, 4H, 4 NH); 13C NMR (75 MHz, DMSO-d6): δ 14.4, 33.3, 56.5, 62.1, 74.6, 126.4, 127.5, 129.2, 135.3, 135.9, 146.8, 150.2, 151.4, 155.6, 161.5, 164.8, 165.7, 172.5. MS (ESI): m/z 1080 [M + 1]+. Anal. calcd for C46H40N12O16S2: C, 51.11; H, 3.73; N, 15.55; found: C, 51.25; H, 3.68; N, 15.70.
General procedure for the synthesis of compounds 11a–c
A mixture of bis(aldehyde) (
Diethyl 3,4-Bis((3-(8,8-dimethyl-2,4,6-trioxo-1,2,3,4,5,6,7,8,9,10-decahydropyrimido[4,5-b]quinolin-5-yl)phenoxy)methyl)thieno[2,3-b] (11a )
Yield: (740 mg, 73% yield); Colorless crystals; m.p. >300 °C. IR (KBr): 3394 (NH), 3201 (NH), 3059 (NH), 1720 (CO), 1666 (CO), 1527 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 0.86 (s, 6H, 2 CH3), 1.01 (s, 6H, 2 CH3), 1.26 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 2.02-2.45 (m, 8H, 4 CH2), 4.31 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 4.71 (s, 2H, 2 CH), 5.44–5.58 (m, 4H, 2 OCH2), 6.41–6.88 (m, 8H, ArH), 8.90 (brs, 2H, 2 NH), 10.71 (s, 2H, 2 NH), 11.88 (brs, 2H, 2 NH). MS (ESI): m/z 1014 [M + 1]+. Anal. calcd for C52H50N6O12S2: C, 61.53; H, 4.96; N, 8.28; found: C, 61.70; H, 5.15; N, 8.45.
Diethyl 3,4-Bis((4-(8,8-dimethyl-2,4,6-trioxo-1,2,3,4,5,6,7,8,9,10-decahydropyrimido[4,5-b]quinolin-5-yl)phenoxy)methyl)thieno[2,3-b] (11b )
Yield: (659 mg, 65%); Yellow crystals; m.p. 284–287°C. IR (KBr): 3309 (NH), 3209 (NH), 3055 (NH), 1712 (CO), 1666 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 0.88 (s, 6H, 2 CH3), 1.01 (s, 6H, 2 CH3), 1.28 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 2.02-2.45 (m, 8H, 4 CH2), 4.31 (q, J = 7.2 Hz, 4H, 2 CH3 CH2), 4.71 (s, 2H, 2 CH), 5.55 (m, 4H, 2 OCH2), 6.73 (d, J = 6.1 Hz, 4H, ArH), 7.06 (d, J = 6.1 Hz, 4H, ArH), 8.89 (br, 2H, 2 NH), 10.65 (s, 2H, 2 NH), 11.85 (br, 2H, 2 NH). MS (ESI): m/z 1014 [M + 1]+. Anal. calcd for C52H50N6O12S2: C, 61.53; H, 4.96; N, 8.28; found: C, 61.73; H, 5.18; N, 8.44.
Diethyl 3,4-Bis(((4-(8,8-dimethyl-2,4,6-trioxo-1,2,3,4,5,6,7,8,9,10-decahydropyrimido[4,5-b]quinolin-5-yl)benzoyl)oxy)methyl)thieno[2,3-b] (11c )
Yield: (728 mg, 68%); Colorless crystals; m.p. >300 °C. IR (KBr): 3394 (NH), 3289 (NH), 3240 (NH), 1720 (CO), 1666 (CO) cm−1; 1H NMR (300 MHz, DMSO-d6): δ 0.85 (s, 6H, 2 CH3), 1.00 (s, 6H, 2 CH3), 1.28 (t, J = 7.2 Hz, 6H, 2 CH3CH2), 1.97–2.51 (m, 8H, 4 CH2), 4.33 (q, 4H, 2 CH3 CH2, J = 7.2 Hz), 4.80 (s, 2H, 2 CH), 5.85 (s, 4H, 2 OCH2), 7.30 (d, J = 6.1 Hz, 4H, ArH), 7.75 (d, J = 6.1 Hz, 4H, ArH), 8.55 (br, 2H, 2 NH), 10.60 (br, 2H, 2 NH), 11.95 (br, 2H, 2 NH). MS (ESI): m/z 1071 [M + 1]+. Anal. calcd for C54H50N6O14S2: C, 60.55; H, 4.71; N, 7.85; found: C, 60.73; H, 4.91; N, 7.66.
Supplemental Material
SI – Supplemental material for Hantzsch-like synthesis of novel bis(hexahydroacridine-1,8-diones), bis(tetrahydrodipyrazolo[3,4-b:4′,3′-e]pyridines), and bis(pyrimido[4,5-b]quinolines) incorporating thieno[2,3-b]thiophenes
Supplemental material, SI for Hantzsch-like synthesis of novel bis(hexahydroacridine-1,8-diones), bis(tetrahydrodipyrazolo[3,4-b:4′,3′-e]pyridines), and bis(pyrimido[4,5-b]quinolines) incorporating thieno[2,3-b]thiophenes by Elshimaa M Eid, Huwaida M E Hassaneen, Ahmed H M Elwahy and Ismail A Abdelhamid in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
