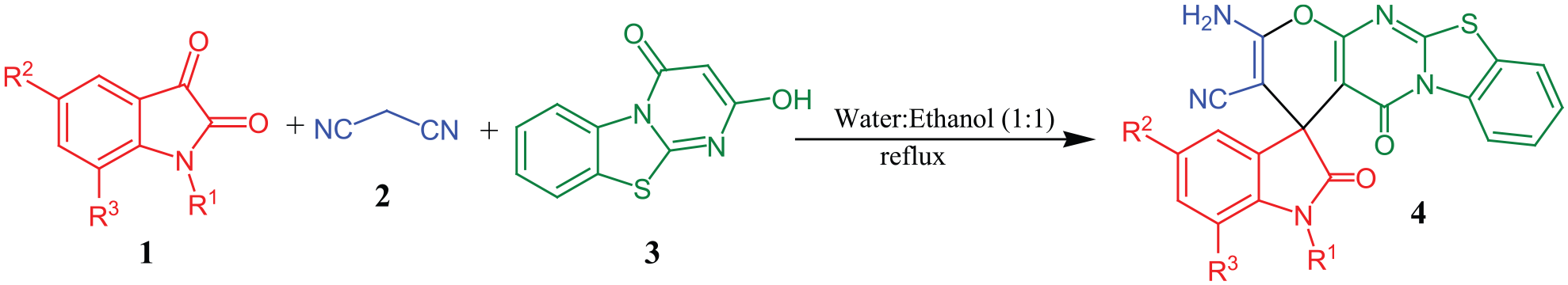
Abstract
The present report describes one-pot three-component condensation of isatins, malononitrile, and 2-hydroxy-4H-pyrimido[2,1-b][1,3]benzothiazol-4-one in water–ethanol mixture at reflux to develop an efficient one-pot protocol for the synthesis of novel spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole] derivatives. The significant features of this protocol are short reaction times, avoidance of toxic catalysts, and provision of excellent yields, no column chromatographic purification, and use of ethanol-water as an environmentally benign solvent. The molecular structure of

Introduction
As a major heterocycle, the spirooxindole system has been identified in a variety of pharmacological and natural products such as spirotryprostatin A, welwitindolinone A, horsfiline, elacomine, coerulescine, and alstonisine.1,2 Spirotryprostatin A is a novel inhibitor of microtubule assembly 3 extracted from the fermentation broth of Aspergillus fumigatus. This natural alkaloid, along with pteropodine, positively modulate muscarinic 5-HT2 and M2 receptors. 4 Furthermore, owing to their beneficial pharmacological and biological properties, including antioxidant, antifungal, 5 anticoagulant, 6 anti-tumor,7,8 anticancer, 9 anti-HIV,10,11 and anti-malarial 12 activities, spirooxindole derivatives play a significant role in synthetic medicinal chemistry. 13 Some additional biologically active spirocyclic oxindoles have also been described in a broader-context review.14–20 Spirooxindoles, especially those spiroannulated with heterocycles at the 3-position, have been reported to possess good biological activities. 21 For example, pyran-annulated oxindoles are widely spread in nature and exhibit various physiological activities. 22 The cytotoxic, 23 anticancer, 24 antimicrobial 25 (Figure 1), analgesic, 26 and herbicidal 27 activities of some synthetic spiroheterocyclic compounds containing both indole and pyran moieties have also been reported. Therefore, intensive efforts have been made to design and develop new approaches for the construction of novel synthetic spirooxindole-fused heterocycles.

Selected bioactive molecules containing pyran-annulated oxindoles.
Fused-pyrimidine-containing heterocyclic skeletons are an important biologically active core unit in heterocyclic compounds and possess remarkable biological properties.28–30 Among them, pyrimidobenzothiazole scaffolds have received considerable attention because of the synthetic challenges associated with their complex molecular architecture and their interesting biological properties, such as antihistaminic, 31 antibacterial,32–34 anticancer, 35 antiviral, 36 and antifungal activities. 37 Recent studies focusing on the synthesis of novel benzo[4,5]thiazolo[1,2-a]pyrimidine-3-carboxylate derivatives have found these compounds to show cytotoxic activity against human breast adenocarcinoma MDA-MB-231 and MCF-7 tumor cell lines. 38 Moreover, pyranopyrimidine contains a diverse array of natural products, as well as pharmaceutically significant compounds, with a wide range of biological activities.39,40 For example, pyrano[2,3-d]pyrimidine are known to exhibit antitumor,41,42 hepatoprotective, 43 antibronchitic, 44 and pronounced antitubercular and antimicrobial activities. 45
Based on the discussed facts and the significant biological properties of spirooxindoles, pyrano[2,3-d]pyrimidine, and pyrimidobenzothiazole, we assume that the synthesis of new substituted polyheterocyclic scaffolds containing spirooxindole, pyrano[2,3-d]pyrimidine, and pyrimidobenzothiazole may result in the finding of novel drug candidates. Hence, this paper describes an easy, highly efficient, environment-friendly, catalyst-free, one-pot procedure for the synthesis of novel spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole] derivatives via the reaction of isatin
A review of the literature showed that in the past few years, several pathways have been developed for the synthesis of spirooxindole from isatin, malononitrile, and various types of activated methylene group in one-pot multicomponent synthesis.48,49 To the best of our knowledge, there is no report using 2-hydroxy-4H-pyrimido[2,1-b][1,3]benzothiazol-4-one
Results and discussion
We first investigated the optimum conditions for the three-component reaction of isatin
Optimization of reaction conditions.

RT: room temperature.
In order to investigate the effects of solvent, we used a number of polar and non-polar reaction media to perform the test reactions. No conversion was detected in non-polar solvents, for example, toluene and xylene, even after long reaction times (Table 1, entries 4–5). On the other hand, moderate to good yields were obtained (60%–85%) in polar solvents like DMF, DMSO, THF, acetonitrile, and ethanol (Table 1, entries 6–10). Due to its low cost, availability, and environment-friendliness, H2O was used as the reaction medium. However, no further improvement was observed in the yield due to the solubility problem of reactants (Table 1, entry 11). Subsequently, we decided to perform the one-pot reaction in a sequential manner using water-ethanol. As discussed earlier, ethanol–water (1:1) mixture was the best among other solvents in this study (Table 1, entry 12). Finally, examining the ethanol–water ratio failed to further improve the yield (Table 1, entries 13–16). We also used an acid (p-toluene sulfonic acid) and a base (diisopropylethylamine) as catalysts in this reaction, but the catalyst-free approach in H2O/EtOH gave the best result.
Subsequently, a variety of isatins were reacted with malononitrile and 2-hydroxy-4H-pyrimido[2,1-b][1,3]benzothiazol-4-one. All reactions completed within 2 hours and resulted the formation of expected novel spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole] derivatives (4a -m, Table 2) in high to excellent yields (85% -95%).
Reaction of isatin

Isolated yield.
The structures of compounds
The structure of compound
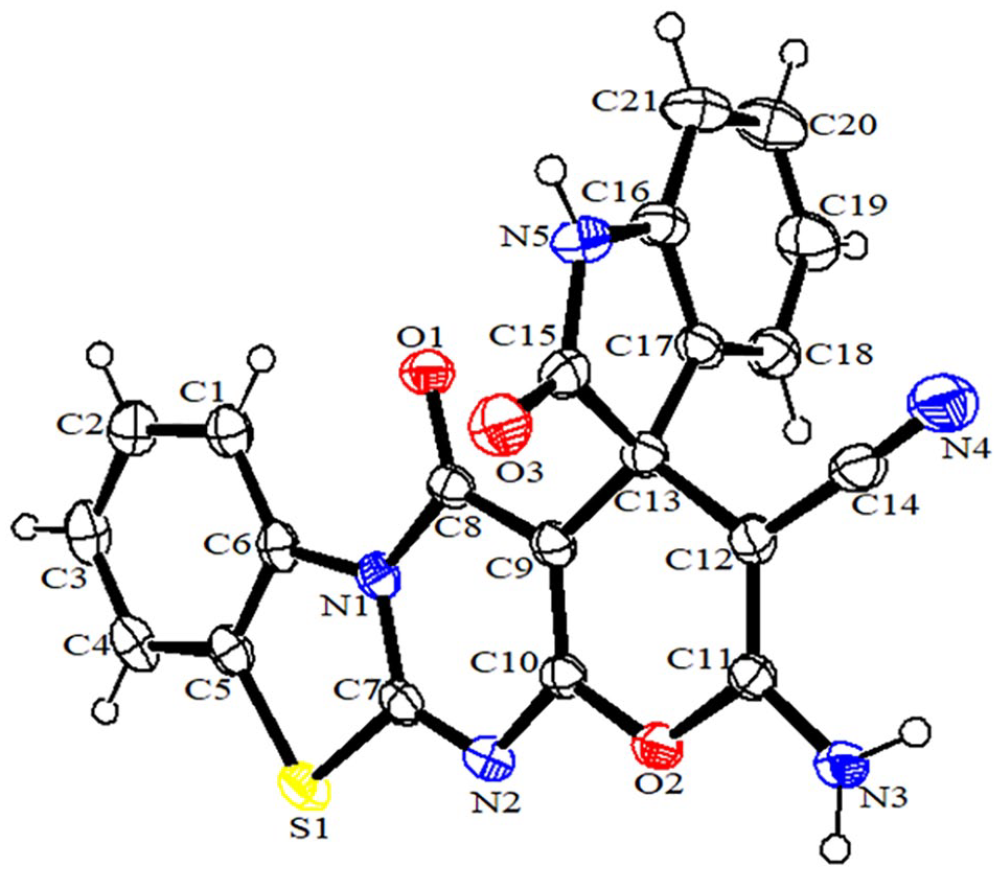
ORTEP diagram of compound
A plausible mechanism is presented in Scheme 1. As seen, during a typical cascade reaction, the first step was a fast Knoevenagel condensation in which isatin

A plausible mechanism.
Overall, this paper described a modified environment-friendly one-pot catalyst-free protocol for the synthesis of novel spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole] derivatives from a three-component reaction between isatin, malononitrile, and 2-hydroxy-4H-pyrimido[2,1-b][1,3]benzothiazol-4-one. We put forward a method in which the target products could be isolated by a simple filtration method due to a difference in the solubility of product and reactant materials. The obtained products were crystallized from MeOH/DMF (4:1) to afford the pure products. This prevented multiple extraction steps and separation by chromatography. Considering the availability of the starting materials, the green and one-pot procedure, easy workup, and high to excellent yields of the products, the products synthesized in the present study may find useful applications in synthetic organic and medicinal chemistry.
Melting points were recorded on an Electrothermal-type 9100 melting point apparatus and are uncorrected. The IR spectra were obtained on an Avatar 370 FT-IR Thermo-Nicolet spectrometer. 1H and 13C NMR spectra were run on BRUKER DRX-300 AVANCE spectrometer at 300 for 1H NMR, and 75 MHz for 13C NMR DMSO-d6 was used as solvent. The mass spectra were scanned on a Varian Mat CH-7 at 70 eV. Elemental analysis was performed on a Thermo Finnigan Flash EA microanalyser. X-ray crystal structure data were collected on a Bruker D8 VENTURE PHOTON 100 CMOS diffractometer with graphite monochromated Cu Kα radiation at 296(2) K.
General procedure for the synthesis of 4
A mixture of isatin
2′-Amino-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-1-methyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-1-ethyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-1-benzyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-1-(4-chlorobenzyl)-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-5-bromo-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-5-methoxy-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-5-chloro-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-5-chloro-1-methyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-5-chloro-1-ethyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-5-methyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-1,5-dimethyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
2′-Amino-7-methyl-2,5′-dioxo-1,2-dihydro-5′H-spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole]-3′-carbonitrile (
Supplemental Material
Supplementary_data – Supplemental material for One-pot, catalyst-free synthesis of novel spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole] derivatives
Supplemental material, Supplementary_data for One-pot, catalyst-free synthesis of novel spiro[indole-3,4′-pyrano[2′,3′:4,5]pyrimido[2,1-b][1,3]benzothiazole] derivatives by Mahdieh Zangouei and Abbas Ali Esmaeili in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Research Council of Ferdowsi University of Mashhad is acknowledged for financial support (Grant No. 3/32089).
Supplemental Material
Supplemental material for this article is available online. Experimental procedures and characterization of synthesized products are accessible in supplemental information. Copies of the NMR, IR, and mass spectra are available.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.