Abstract
Seven novel carbohydrate conjugates of new chalcone-3-O-glycosides were synthesized and characterized. Starting from the substituted 3′-hydroxyarylmethylacetophenone derivatives (chalcones) with α-acetobromoglucose in anhydrous acetone were synthesized 2,3,4,6-tetra-O-acetyl-3′-O-β-
Introduction
Chalcones, compounds bearing the 1,3-diaryl-2-propen-1-one system, play a vital role in chemical compounds associated with diverse pharmacological activities. 1 Due to their flexible structures, chalcones can bind effectively to many kinds of enzymes or receptors and exhibit a broad spectrum of biological activities such as anticancer, 2 anti-HIV, 3 anti-inflammatory,4,5 anti-invasive,6,7 and antibacterial properties. 8
Glycosylation of small biologically active molecules, either of natural or synthetic origin, has an effect on their solubility, and bioactivity. Notably, β-glucosylation improves the drug’s targeting to cells as well as their “solubility” in cell membranes.9,10 O-glycosides are a family of carbohydrates widely found in plants with carbon–oxygen bond formation employing activated sugar.11,12 Phenolic hydroxyl groups occur in the free state or in combined form as O-glycosides in plants where they play many interesting biological activities, such as antitumor activity and inhibitors of the metabolic process. 13
Much attention has been paid to chalcones as potential biological agents after the discovery of the natural chalcone-O-glycosides which demonstrated a broad spectrum of antibiotic, antifungal, and anti-HIV activity. 14 The isolation of some chalcone glycosides (see Scheme 1) from the aerial parts of Brassica rapa L. “hidabeni” has been reported. 15 However, to the best of the author’s knowledge, few reports have been dedicated to the synthesis and inhibitory activity of chalcone derivatives containing an O-glycoside skeleton.

Structures of isolated chalcone glycosides from Brassica rapa L. “hidabeni.”
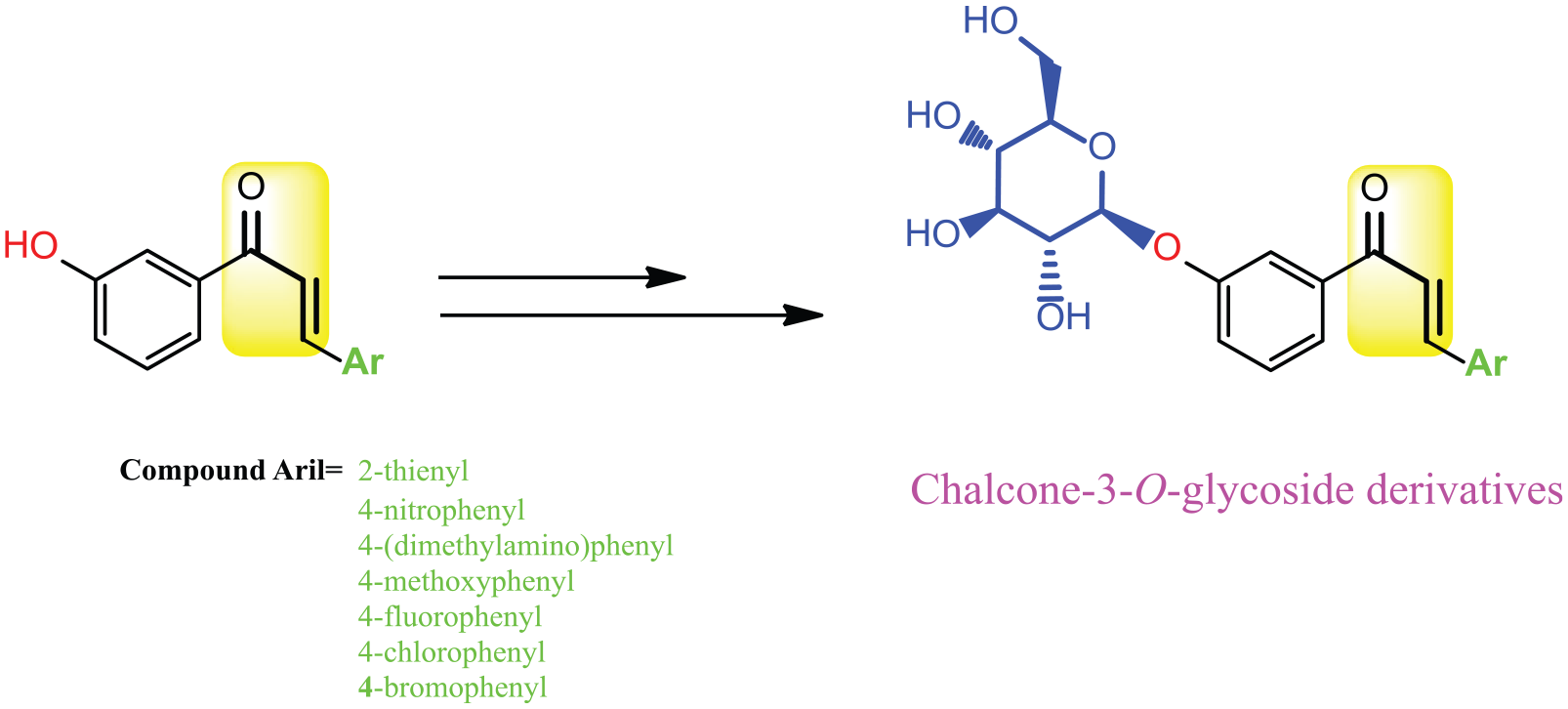
In this study, preliminary results in the organic synthesis of some new chalcone-3-O-glycosides will be reported. Scheme 2 shows the synthetic analysis of the target molecules. Target molecules in this study are chalcones with various sugar substitutions at 3-position.

General route for the synthesis of chalcone-3-O-glycosides (
Results and discussion
First, substituted chalcones starting from 3-hydroxyacetophenone and different aldehydes were synthesized at room temperature according to the Claisen–Schmidt condensation method, which is a single-step process and a practical method for the synthesis of substituted chalcone derivatives (Scheme 2). 16
In the second step, O-glucosylation reactions were performed by condensing α-acetobromoglucose (ACBG) and 3-hyroxychalcones (
The synthesis of chalcone-3-O-glycosides was achieved in this second step. The structures of all the compounds were confirmed by nuclear magnetic resonance (NMR), liquid chromatography–mass spectrometry (LC/MS–MS), and Fourier-transform infrared spectroscopy (FTIR; attenuated total reflection (ATR)), spectroscopy. The results were consistent with the predicted structures for all the new chalcone-3-O-glycosides, as described in section “Experiment”.
The β-configuration of the glycosidic bond was confirmed from 1H NMR and 13C NMR. In the 1H NMR spectrum of
In the proton-decoupled 13C NMR, the anomeric carbon was located at δ ~101.00, which is consistent with the formation of the β-glucoside
The structure of 3-((2E)-3-(2-thienyl)prop-2-enol-yl)phenyl hexopyranoside (
In the 1H NMR spectra of the synthesized chalcone-3-O-glycoside derivatives (
In the 13C NMR spectra of the compound
The mass spectra of compounds
Conclusion
The synthesis of seven new chalcone-3-O-glycosides, which can be used to make more glycosylated chalcone, is reported in this study. The structures of the synthesized chalcone and chalcone-3-O-glycoside have been confirmed by NMR, LC/MS–MS, and FTIR (ATR) spectroscopy. Chalcones are important α,β-unsaturated ketones and constitute a class of naturally occurring substances, which are very attractive bioactive starting materials for the synthesis of substituted chalcone-O-glycosides.
Experiment
1
H and 13C NMR spectra were recorded on a 400 (100)-MHz Bruker Avance spectrometer in methanol-d4; δ is given in ppm relative to Me4Si (tetramethylsilane (TMS)) as an internal standard. The following abbreviations are used: s = singlet, d = doublet, dd = doublet doublet, t = triplet, m =multiplet, bs =broad singlet, bd = broad doublet. The high-resolution accurate masses were determined using Micromass Quattro LC–MS/MS. The IR spectra were recorded on a Perkin Elmer 1600 Fourier transform infrared (FTIR-ATR) spectrophotometer. Compounds
Synthesis of chalcones (3a–g ): general procedure
Compounds
Synthesis of 2,3,4,6-tetra-O-acetyl-3′-O-β-glucopyranosyloxybenzlideneacetophenones (4a–g ): general procedure
The 2,3,4,6-tetra-O-acetyl-α-
The general method for 3′-O-β-glucopyranosyloxybenzlideneacetophenones 5a–g
A freshly prepared solution of MeONa–MeOH (3 mL, 0.05 M) was added to a solution of 2,3,4,6-tetra-O-acetyl-3′-O-β-glucopyranosyloxybenzlideneacetophenones
3-[(2E)-3-(2-thienyl)prop-2-enol-yl]phenyl hexopyranoside (
3-[(2E)-3-(4-nitrophenyl)prop-2-enol-yl]phenyl hexopyranoside (
3-[(2E)-3-(4-dimethylaminophenyl)prop-2-enol-yl]phenyl hexopyranoside (
3-[(2E)-3-(4-methoxyphenyl)prop-2-enol-yl]phenyl hexopyranoside (
3-[(2E)-3-(4-fluorophenyl)prop-2-enol-yl]phenyl hexopyranoside (
3-[(2E)-3-(4-chlorophenyl)prop-2-enol-yl]phenyl hexopyranoside (
3-[(2E)-3-(4-bromophenyl)prop-2-enol-yl]phenyl hexopyranoside (
Supplemental Material
Supplementary_File – Supplemental material for New chalcone-3-O-glycoside derivatives: Synthesis and characterization
Supplemental material, Supplementary_File for New chalcone-3-O-glycoside derivatives: Synthesis and characterization by Gonca Çelik in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author acknowledges the financial support from The Scientific and Technological Research Council of Turkey (TUBITAK) with Project 116R016.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
