Abstract
Seven (E)-2-aroyl-4-(4-fluorobenzylidene)-5-oxopyrrolidines were synthesized via a tandem Ugi 4CC/SN cyclization sequence starting from a Baylis-Hillman-derived acid, primary amines, arylglyoxals, and isocyanides in one pot. In addition, the cytotoxicity of these compounds on the cervical cancer cell line HeLa was studied by MTT assay. The results showed that most of the compounds could inhibit the proliferation of HeLa cells significantly.
Keywords

Introduction
Heterocyclic compounds are widely used in the fields of pesticides, medicinal chemistry, and industry because of their useful properties. Fluorine-containing compounds have been widely used as pharmaceuticals due to their excellent lipophilicity, hydrophobicity, and biological activities. Therefore, the synthesis of heterocyclic compounds containing a fluorine atom is of interest to drug researchers.
Heterocycles containing a pyrrolidinone are of importance because they have many applications in pharmaceuticals, chemicals, and other fields. 1 They have significant antimuscarinic, 2 antiviral, 3 antiepileptic, 4 and anti-HIV activities. 5 Atigadda et al. 6 reported a substituted 2-pyrrolidinone that was a low nanomolar inhibitor of neuraminidase. Wu and Feldkamp 7 utilized a pyrrolidinone as a scaffold in a novel selective inhibitor of matrix metalloproteinases (MMPs).
The classic synthetic methodology for the construction of pyrrolidinones is focused primarily on the cyclization of a dialkyl itaconate with different primary amines. 8 In addition, pyrrolidinones have been prepared by other methods, such as rearrangements of lactams, 9 radical addition cyclization reactions, cycloaddition reactions, and transition-metal-catalyzed intramolecular allylic alkylation reaction. 10
Studies showed that the alkaloids Rhopaladins A–D (see Figure 1), which exist in marine cysts, have significant cytotoxicity to human tumor cells. 11 Rhopaladin B inhibited a cyclin-dependent kinase (CDK4) and tyrosine kinase C-erbB2 (IC50 was 12.5 and 7.4 μg mL−1, respectively),12,13 in particular. The data show that C-erbB2 expression is a late event in cervical carcinogenesis. 14 Cancer incidence and mortality are rapidly growing worldwide. There were about 570,000 cases and 310,000 deaths worldwide in 2018. The incidence of cervical cancer ranked fourth and was the fourth leading cause of cancer in women. 15
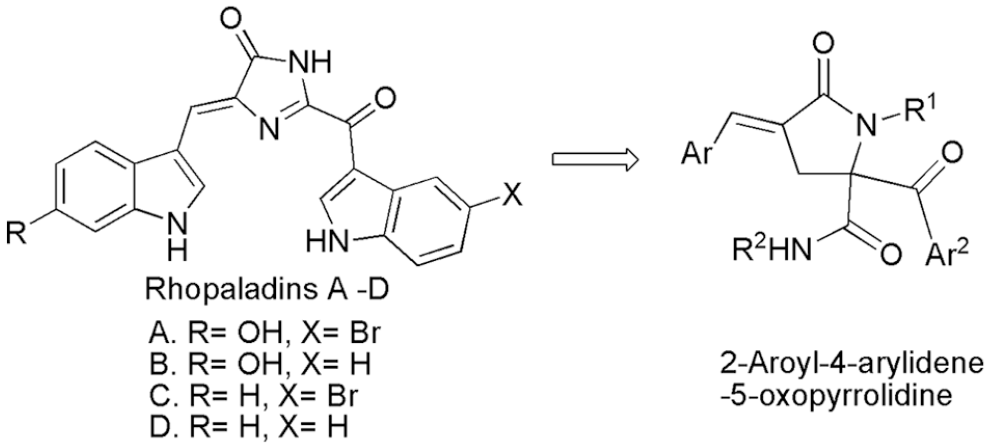
The strategy for the choice of 2-aroyl-4-arylidene-5-oxopyrrolidines as targets.
Results and discussion
We have previously synthesized 2-aroyl-4-arylidene-5-oxopyrrolidines via tandem Ugi and cyclization reactions (see Figure 1) 16 and found they are biological isochores of Rhopaladins because of the five-membered nitrogen heterocycles with arylidene and aroyl groups. In order to study the anti-cervical cancer activity of these compounds, we decided to synthesize fluorine-containing (E)-2-aroyl-4-(4-fluorobenzylidene)-5-oxopyrrolidines. Herein, we report the synthesis of seven (E)-2-aroyl-4-(4-fluorobenzylidene)-5-oxopyrrolidines via a tandem Ugi 4CC/SN cyclization sequence starting from a Baylis-Hillman-derived acid, primary amines, arylglyoxals, and isocyanides in one pot. In addition, the cytotoxicity of these compounds to HeLa cells was studied by the MTT assay.
The optimized conditions
16
for the fluorine-containing Baylis-Hillman-derived acid
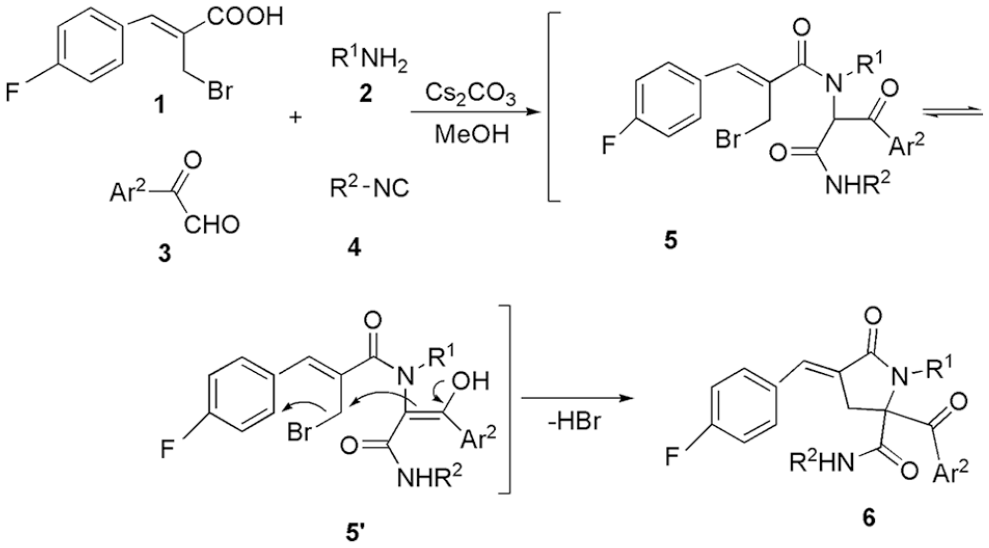
Synthesis of (E)-2-aroyl-4-(4-fluorobenzylidene)-5-oxopyrrolidines
Synthesis of compound

Isolated yields.
The structures of compounds
The MTT assay was used to test the cytotoxic activity of the target compounds
Toxicity of compound 6 on HeLa cells in vitro.

Conclusion
Seven (E)-2-aroyl-4-(4-fluorobenzylidene)-5-oxopyrrolidines have been synthesized via a tandem Ugi 4CC/SN cyclization starting from a Baylis-Hillman-derived acid, primary amines, arylglyoxals, and isocyanides in one pot. All are new compounds which have not been reported before. In the experimental procedure, the Ugi reaction intermediates do not need to be separated and purified, but directly used in the following reaction. The cytotoxicities of compounds
Experimental
Melting points were determined using a X-4 model apparatus and were uncorrected. MS were measured on a Finnigan Trace MS spectrometer. IR were recorded on a PE-983 infrared spectrometer as KBr pellets with absorption in cm−1. NMR were recorded in CDCl3 or [D6] DMSO on a Varian Mercury 600 or 400 spectrometer and resonances relative to TMS. Elementary analyses were taken on a Vario EL III elementary analysis instrument.
General procedure for the synthesis of 6a–g
A mixture of 2-(bromomethyl)-3-(4-fluorobenzyl)-2-propenoic acid
(E)-1-Benzyl-2-(4-chlorobenzoyl)-N-cyclohexyl-4-(4-fluorobenzylidene)-5-oxopyrrolidine-2-carboxamide (
(E)-1-Benzyl-2-(4-bromobenzoyl)-N-cyclohexyl-4-(4-fluorobenzylidene)-5-oxopyrrolidine-2-carboxamide (
(E)-1-Benzyl-N-cyclohexyl-4-(4-fluorobenzylidene)-2-(4-nitrobenzoyl)-5-oxopyr-rolidine-2-carboxamide (
(E)-1-Benzyl-N-tert-butyl-2-(4-chlorobenzoyl)-4-(4-fluorobenzylidene)-5-oxopyrrolidine-2-carboxamide (
(E)-1-Benzyl-2-(4-bromobenzoyl)-N-tert-butyl-4-(4-fluorobenzylidene)-5-oxopyrrolidine-2-Carboxamide (
(E)-1-Benzyl-N-tert-butyl-4-(4-fluorobenzylidene)-2-(4-nitrobenzoyl)-5-oxopyrrolidine-2-carboxamide (
(E)-2-(4-Chlorobenzoyl)-N-cyclohexyl-4-(4-fluorobenzylidene)-1-isopropyl-5-oxopyrrolidine-2-carboxamide (
MTT assay
Cell proliferation was assessed using the MTT method.
17
The evaluation of cell population growth is based on the capability of living cells to reduce the yellow product MTT to a blue product, formazan, by a reduction reaction occurring in the mitochondria. Cervical carcinoma cell line HeLa was cultured in Dulbecco’s Modified Eagle Medium (DMEM) which contained 10% fetal bovine serum (FBS, Gibco, USA). The cells were grown in an incubator with a humidified atmosphere of 5% CO2 at 37 °C. HeLa cells in logarithmic growth phase were digested with 0.25% Trypsin EDTA solution. The cells were incubated for 24 h in 96-well plates at a density of 5 × 103 cells/well. The cells were treated with varying concentrations of compound
Supplemental Material
Supporting_Information – Supplemental material for Synthesis and cytotoxic effects on HeLa cervical cancer cells of (E)-2-aroyl-4-(4-fluorobenzylidene)-5-oxopyrrolidine
Supplemental material, Supporting_Information for Synthesis and cytotoxic effects on HeLa cervical cancer cells of (E)-2-aroyl-4-(4-fluorobenzylidene)-5-oxopyrrolidine by Hongmei Wang, XiuLian Zhu, Shouheng Deng, Qinhua Chen and Xiaohua Zeng in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (81872509), the Hubei Provincial Technology Innovation Project (2017ACA176), the Open Project of Hubei Key Laboratory of Wudang Local Chinese Medicine Research (Hubei University of Medicine) (Grant No. WDCM2018001), the Shiyan Municipal Science and Technology Bureau Science and technology project (Grant Nos 18K79 and 18Y01), and the scientific research innovation team of Hubei University of Medicine (Grant Nos 2014QDJZR10 and 2014CXZ01).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
