Abstract
A three-step synthesis of 2-arylamino-5-formyl-pyrimidines is developed by condensation of the bis(hexafluorophosphate) Arnold salt with N-arylguanidines. This method conveniently provides the corresponding 2-arylaminopyrimidine derivatives in good yields.

Introduction
The Arnold salt is the only practical source of triformylmethane known to date, which was first isolated by Arnold as the perbromide salt

Some typical Arnold salts.

Examples of drugs with 2-arylaminopyrimidine moieties.
Since bis(hexafluorophosphate) Arnold salt
Results and discussion
Our synthesis of bis(hexafluorophosphate) salt

Our synthesis of
With the desired salt in hand, we attempted to prepare an array of N-arylguanidines. The direct coupling of aromatic amines with cyanamide under acidic conditions is a straightforward method to prepare N-arylguanidines. However, this protocol was not appealing since the resulting crude products were difficult to purify and the yields were fairly low with significant qualities of aromatic amines recovered. Thus, a two-step method was adopted. Aromatic amines
The structures and yields of compounds
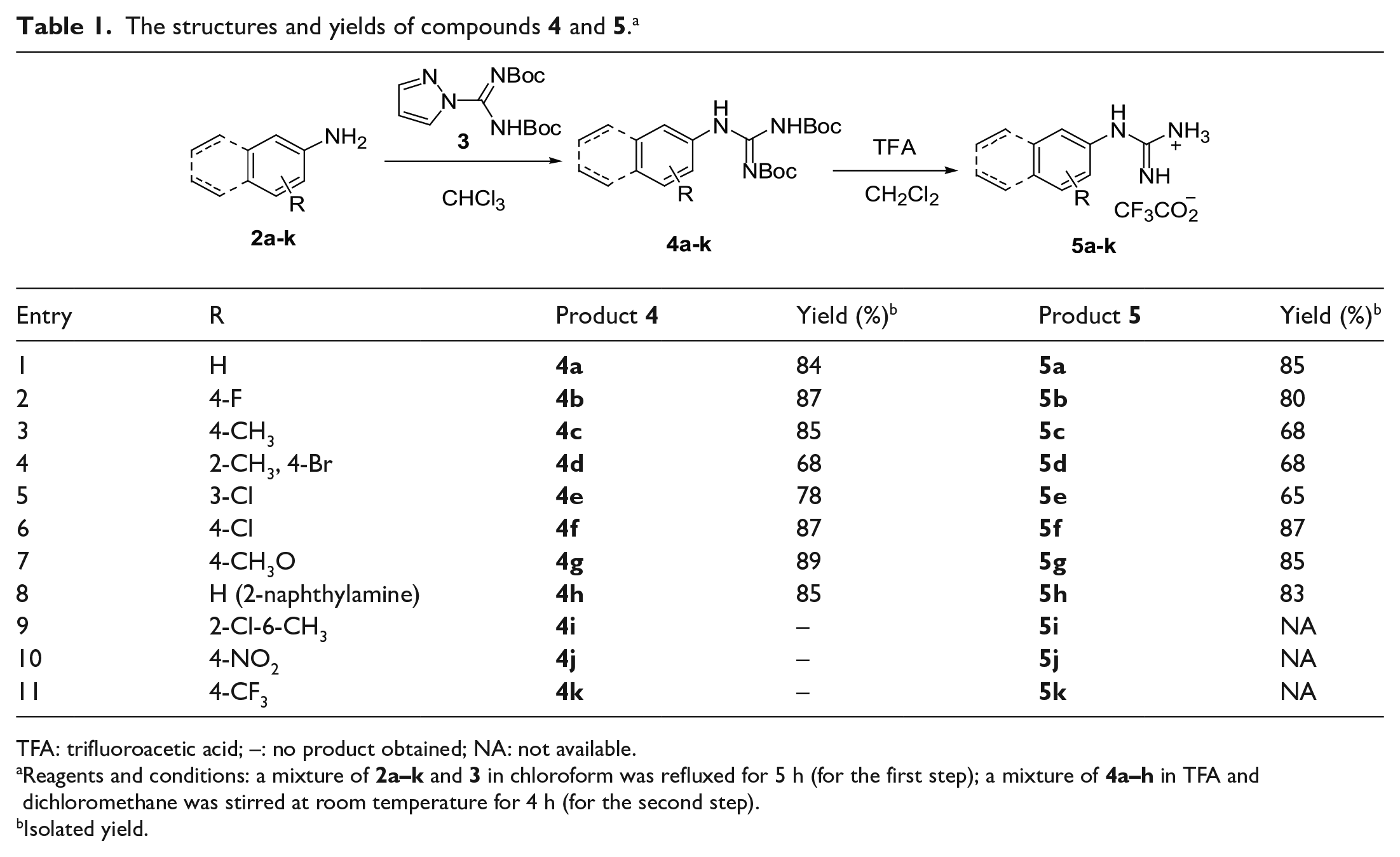
TFA: trifluoroacetic acid; –: no product obtained; NA: not available.
Reagents and conditions: a mixture of
Isolated yield.
Subsequently, the condensation of Arnold salt
Effect of bases and solvents on the condensation. a

DMF: dimethylformamide; –: no product obtained.
Reagents and conditions: a mixture of
Isolated yield.
Methanol:water = 3:1.
Synthesis of 2-arylamino-5-formyl-pyrimidines. a

Reagents and conditions: a mixture of
Isolated yield.
Conclusion
In conclusion, the condensation of N-arylguanidines with bis(hexafluorophosphate) Arnold salt
Experimental
Commercial reagents were used without further purification. Melting points were measured on a SGW X-4 (INESA) melting point apparatus and are uncorrected. 1H NMR spectra were recorded on Bruker DRX-500 (500 MHz) or DRX-600 (600 MHz) instrument. 13C NMR spectra were obtained on JNMEX400 (125 MHz) or JNMEX400 (150 MHz) instrument. Mass spectra (MS) were recorded on a Bruker MicrOTOF II mass spectrometer or a Waters High Resolution UPLCTOFMS spectrometer. Infrared (IR) spectra were obtained using attenuated total reflectance (ATR) on a Fourier transform infrared (FTIR) Bruker Tensor 27. DSC was determined on a Mettler Toledo TGA/DSC 3+.
General procedure for the synthesis of Arnold salt 1d
Synthesis of bis(hexafluorophosphate) Arnold salt (1d )
Trichlorophosphate (73 g, 0.48 mol) was added dropwise to DMF (63.3 g, 0.87 mol) at 0°C. Next, malonic acid (15 g, 0.14 mol) was added and the resulting mixture was stirred at 90 °C for 7 h. NH4PF6 (47 g, 0.29 mol) was dissolved in ice water and added to the reaction mixture. The Arnold salt precipitated and was triturated with isopropanol (120 mL) to give pure
General procedure for the synthesis of bis-Boc guanidines
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-4-benzene (4a )
N,N′-bis-Boc-1H-pyrazole-1-carboxamidine (
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-4-fluorobenzene (4b )
The general procedure was followed (3.22 mmol scale). The crude residue was purified by column chromatography (10% ethyl acetate in petroleum ether) to give
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-4-methylbenzene (4c )
The general procedure was followed (3.22 mmol scale). The crude residue was purified by column chromatography (10% ethyl acetate in petroleum ether) to give
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-2-methyl-4-bromobenzene (4d )
The general procedure was followed (3.22 mmol scale). The crude residue was purified by column chromatography (10% ethyl acetate in petroleum ether) to give
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-3-chlorobenzene (4e )
The general procedure was followed (3.22 mmol scale). The crude residue was purified by column chromatography (10% ethyl acetate in petroleum ether) to give
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-4-chlorobenzene (4f )
The general procedure was followed (3.22 mmol scale). The crude residue was purified by column chromatography (10% ethyl acetate in petroleum ether) to give
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-4-methoxybenzene (4g )
The general procedure was followed (3.22 mmol scale). The crude residue was purified by column chromatography (10% ethyl acetate in petroleum ether) to give
Synthesis of 1-[2,3-di(tert-butoxycarbonyl)guanidino]-naphthalene (4h )
The general procedure was followed (3.22 mmol scale). The crude residue was purified by column chromatography (10% ethyl acetate in petroleum ether) to give
General procedure for the synthesis of guanidines
Synthesis of 1-phenylguanidine trifluoroacetate (5a)
1-[2,3-Di(tert-butoxycarbonyl)guanidino]-4-benzene
Synthesis of 1-(4-fluorophenyl)guanidine trifluoroacetate (5b)
The general procedure was followed (2.83 mmol scale). The crude residue was triturated with 50% ethyl acetate in petroleum ether (3 mL) to give
Synthesis of 1-(p-tolyl)guanidine trifluoroacetate (5c )
The general procedure was followed (2.83 mmol scale). The crude residue was triturated with 50% ethyl acetate in petroleum ether (3 mL) to give
Synthesis of 1-(4-bromo-2-methylphenyl)guanidine trifluoroacetate (5d)
The general procedure was followed (2.83 mmol scale). The crude residue was triturated with 50% ethyl acetate in petroleum ether (3 mL) to give
Synthesis of 1-(3-chlorophenyl)guanidine trifluoroacetate (5e)
The general procedure was followed (2.83 mmol scale). The crude residue was triturated with 50% ethyl acetate in petroleum ether (3 mL) to give
Synthesis of 1-(4-chlorophenyl)guanidine trifluoroacetate (5f)
The general procedure was followed (2.83 mmol scale). The crude residue was triturated with 50% ethyl acetate in petroleum ether (3 mL) to give
Synthesis of 1-(4-methoxyphenyl)guanidine trifluoroacetate (5g )
The general procedure was followed (2.83 mmol scale). The crude residue was triturated with 50% ethyl acetate in petroleum ether (3 mL) to give
Synthesis of 1-(naphthalen-2-yl)guanidine trifluoroacetate (5h )
The general procedure was followed (2.83 mmol scale). The crude residue was triturated with 50% ethyl acetate in petroleum ether (3 mL) to give
General procedure for the synthesis of N-arylpyrimidines
Synthesis of 2-(phenylamino)pyrimidine-5-carbaldehyde (6a )
1-Phenylguanidine trifluoroacetate (
Synthesis of 2-[(4-fluorophenyl)amino]pyrimidine-5-carbaldehyde (6b )
The general procedure was followed (3.7 mmol scale). The crude residue was purified by column chromatography (50% ethyl acetate in petroleum ether) to give
Synthesis of 2-(p-tolylamino)pyrimidine-5-carbaldehyde (6c )
The general procedure was followed (3.7 mmol scale). The crude residue was purified by column chromatography (50% ethyl acetate in petroleum ether) to give
Synthesis of 2-[(4-bromo-2-methylphenyl)amino]pyrimidine-5-carbaldehyde (6d )
The general procedure was followed (3.7 mmol scale). The crude residue was purified by column chromatography (50% ethyl acetate in petroleum ether) to give
Synthesis of 2-[(3-chlorophenyl)amino]pyrimidine-5-carbaldehyde (6e )
The general procedure was followed (3.7 mmol scale). The crude residue was purified by column chromatography (50% ethyl acetate in petroleum ether) to give
Synthesis of 2-[(4-chlorophenyl)amino]pyrimidine-5-carbaldehyde (6f )
The general procedure was followed (3.7 mmol scale). The crude residue was purified by column chromatography (50% ethyl acetate in petroleum ether) to give
Synthesis of 2-[(4-methoxyphenyl)amino]pyrimidine-5-carbaldehyde (6g )
The general procedure was followed (3.7 mmol scale). The crude residue was purified by column chromatography (50% ethyl acetate in petroleum ether) to give
Synthesis of 2-(naphthalen-2-ylamino)pyrimidine-5-carbaldehyde (6h )
The general procedure was followed (3.7 mmol scale). The crude residue was purified by column chromatography (50% ethyl acetate in petroleum ether) to give
Supplemental Material
Supplementary_Information2020.02.14b – Supplemental material for Synthesis of 2-arylamino-5-formyl-pyrimidines from the bis(hexafluorophosphate) Arnold salt
Supplemental material, Supplementary_Information2020.02.14b for Synthesis of 2-arylamino-5-formyl-pyrimidines from the bis(hexafluorophosphate) Arnold salt by Qiuyu Lu, Wei He, Wen Sun, Ye Feng, Li Zhan and Yu Luo in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the Laboratory of Organic Functional Molecules and the Sino-French Institute of ECNU for their support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



