Abstract
In this study, we have investigated the electrochromic properties including the change in absorption spectra and colour after oxidation and reduction reactions of tetraphenylporphyrin and its metal complexes in dichloromethane. The first oxidation potential is determined from CV measurements, and the reduction potential is estimated from comparison with literature values. Electrolytic reactions are carried out by applying the oxidation potential and reduction potential to each sample solution. The metals used are Ag(II), Cu(II), Fe(III), Mg(II), Mn(III), Ni(II) and Zn(II). Various colours can be expressed after the redox reactions by changing the central metal.

Introduction
Electrochromism is the phenomenon where the colour of materials changes by oxidation or reduction upon applying an electric potential.1–15 Inorganic materials are excellent in stability and repetition durability, but there is a disadvantage that the range of colour is limited. Tungsten oxide is a typical inorganic material. 16 On the contrary, organic compounds are often less stable; however, various colours are possible by fine-tuning the molecular design. Porphyrins, having an 18π electron system, can generate π-cations as well as π-anions by redox reactions. Although many pioneering studies on redox properties, sometimes including spectral changes of porphyrins, have been reported, 17 systematic research on the electrochromic properties including colour changes has not been reported except for a few studies using phthalocyanines.18–22 Here, we report the colour changes as well as spectroscopic changes of tetraphenylporphyrin (H2TPP) and metal complexes (MTPP) upon redox reactions. The metals used are Ag(II), Cu(II), Fe(III), Mg(II), Mn(III), Ni(II) and Zn(II) (Figure 1).
Structures of the MTPPs: M = Ag(II), Cu(II), Fe(III), Mg(II), Mn(III), Ni(II) and Zn(II).
Results and discussion
The colour photographs in the neutral states of the synthesized H2TPP and metal complexes are shown in Figures 2–9 (written as neutral). Six types, AgTPP, CuTPP, H2TPP, MgTPP (difficult to see because of the poor photograph), NiTPP and ZnTPP are reddish purple, FeTPPCl is orange and MnTPPCl is yellowish green. The ideal colours for electrochromic materials for displays are cyan, magenta and yellow (CMY) or red, green and blue (RGB).
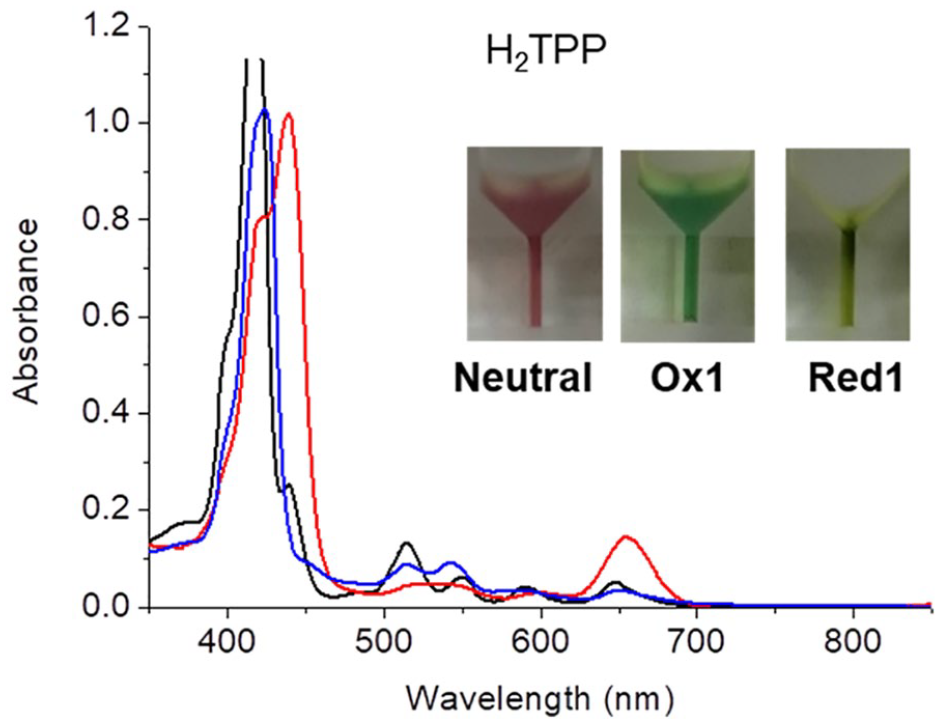
UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of H2TPP and their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.

UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of ZnTPP and their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.

UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of AgTPP and their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.

UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of CuTPP and their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.

UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of FeTPPCl and their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.

UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of MgTPP and their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.

UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of MnTPPCl, their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.

UV/Vis absorption spectra of neutral (black), first oxidation (red), first reduction (blue) states of NiTPP and their photographs (inset) in a 0.1 M TBAP solution of dichloromethane.
The redox potential was determined by cyclic voltammetry (CV) to examine changes in colour and absorption spectra during the redox reaction. Dichloromethane, in which 0.1 M tetrabutylammonium perchlorate (TBAP) had been dissolved, was used as the solvent. The reference electrode was Ag/Ag+, and the working electrode and counter electrode were both platinum. In some cases, a clear CV peak could not be obtained in the reduction side using our measurement system. Therefore, the first reduction potential was estimated using the value of ΔE1/2 (Ox1 − Red1) obtained from the literature17,23–30 and the value of the observed first oxidation potential. The solvent and electrolyte used in this work are the same as in the literature. The reference electrode is Ag/Ag+ in this study, but SCE was used in the literature. In the case of H2TPP, both the first oxidation and the first reduction potentials could be obtained from CV measurements (see the Supplementary material), giving a value of ΔE1/2 (Ox1 − Red1) of 2.30 V. The value obtained from the literature 25 was 2.22 V, which is almost consistent with our measurements. Furthermore, when the first oxidation potentials obtained for other porphyrins were compared with the literature values, the difference, which was originated from the difference in the reference electrode, was found to be 0.3 ± 0.03 V. The results are summarized in Table 1.
The first oxidation and reduction potentials of the MTPPs.

The reduction potentials, Red1, in this work were obtained by calculation. See text for details.
First, changes in the colour and absorption spectra of the solution after the redox reactions of H2TPP were examined. As is well-known, porphyrins have an absorption maximum called the Soret band near 400 nm and have four (free base) or two (metal complexes) Q-bands in the 500–700 nm region. H2TPP is reddish purple in the neutral state, and its photograph and absorption spectra are shown in Figure 2. Those of the first oxidized H2TPP solution by electrolytic reaction with applying a potential of 0.85 V, which is 0.10 V higher than the first oxidation potential (see the experimental section for details), are also shown in Figure 2. The reddish-purple colour in the neutral state changed to green after the first oxidation. In the absorption spectra, the Soret band shifted from 418 to 440 nm with a shoulder at around 422 nm, and a relatively strong absorption was confirmed at 650 nm. After the first reduction with applying a potential of −1.57 V, which is 0.10 V lower than the first reduction potential, it can be seen that the Soret band as seen in Figure 2 was slightly red-shifted and became weak compared with the neutral state. The colour became brighter green or yellow.
Changes in the colour and absorption spectra after redox reactions of the TPP metal complexes were observed using the same operations as for H2TPP. First, the colour and absorption spectra of ZnTPP after the first oxidation were measured (Figure 3). In the absorption spectra after the first oxidation with applying a potential of 0.60 V, the Soret band shifted from 420 to 442 nm, and the two Q-bands also shifted to longer wavelength. The colour was brownish/dark green. The Soret band after the first reduction with applying a potential of −1.77 V split into two bands at 418 and 429 nm, and the colour became yellowish green.
According to the literature, in the cases of Ag(II), 28 Fe(III) 24 and Mn(III), 23 it appears that the first oxidation and the first reduction are involved in the redox of the central metal, not the porphyrin ring. Figure 4 shows the absorption spectra after the first oxidation of AgTPP involving an Ag(II)/Ag(III) transition 28 with applying a potential of 0.38 V. A slight red-shift and broadening compared with the neutral state were observed, and the intensity decreased. The colour after oxidation changed from reddish purple to light brown. In the absorption spectra after the first reduction corresponding to Ag(II)/Ag(I) 28 with applying a potential of −1.42 V, a further decrease in absorption intensity was observed, and the colour was also light brown.
The Soret band after the first oxidation of CuTPP with applying a potential of 0.83 V was also split into two bands at 415 and 440 nm with a relatively large absorption at around 655 nm, and the colour was brown as shown in Figure 5. In the absorption spectra after the first reduction with applying a potential of −1.69 V, splitting of the Soret band was also observed, but the absorption became weaker and the colour was light brown.
In the case of FeTPPCl, a detailed discussion of the first oxidation and the first reduction potentials from a study on the counter ion and solvent effects has been made, 24 and it was mentioned that they might be due to the oxidation of the porphyrin ring and the Fe(III)/Fe(II) transition, respectively. In the absorption spectra of FeTPPCl after the first oxidation with applying a potential of 0.94 V as represented in Figure 6, although the shape was slightly changed as compared with the neutral state, the colour did not change largely. After the first reduction with applying a potential of −0.69 V, although the absorption intensity of the Soret band decreased, the appearance of the colour did not change significantly.
MgTPP showed no significant change in absorption spectra and colour even when an oxidation potential of 0.44 V was applied. Furthermore, when a reduction potential of −1.91 V was applied, bleaching occurred and the colour turned pale yellow (Figure 7).
In MnTPPCl as well as FeTPPCl, it was considered that the first oxidation potential and the first reduction potential might be due to the oxidation of the porphyrin ring and the Mn(III)/Mn(II) transition, respectively, from the studies on the counter ion and solvent effects. 23 The absorption spectra and colour of MnTPPCl after the first oxidation with applying a potential of 0.96 V did not change much compared to the neutral state (see Figure 8). After the first reduction with applying a potential of −0.67 V, the Soret band absorption weakened and the colour changed to yellowish green.
The behaviour of NiTPP was similar to that of MgTPP. Even when an oxidation potential of 0.85 V was applied, the absorption spectra and colour did not change significantly, but after applying a reduction potential of −1.65 V, bleaching occurred and the colour became paler (Figure 9).
Conclusion
In summary, after the redox reactions of the TPP metal complexes in dichloromethane, it was found that various colours can be expressed by changing the central metal. Not only the redox of the porphyrin ring, but also the redox reaction of the central metals, as seen in silver, iron and manganese, caused a colour change due to a change in the electronic environment of the porphyrin ring. By changing the molecular structure and central metal, these porphyrins might be applied to electrochromic materials that can output CMY or RGB from a single compound. We are currently investigating TPP with other metal ions and phthalocyanines. Furthermore, not only the first oxidation and the first reduction, but also the colour changes in the second oxidation and the second reduction are investigated. Although the reversibility of the redox reaction can be seen in CV measurements that only observe near the electrode surface, almost no reversibility of the electrochromism was observed in the electrolytic reactions probably due to low stability of the radical species. It is necessary to consider the stabilization of the radical species, for example, by introducing picket fences or by complete fluorination.
Experimental
The UV-Vis absorption spectra were measured using a JASCO Corporation V-630 spectrometer. MALDI-TOF mass measurements were performed with a Shimadzu AXIMA-CFR spectrometer using dithranol as a matrix. CV measurements and electrolytic reactions were measured using a BAS ALS600C electrochemical analyzer with a three-electrode system. Dichloromethane, in which 0.1 M TBAP was dissolved, was used as the solvent. The solution was purged with nitrogen before measurements. The reference electrode was Ag/Ag+, and the working electrode and counter electrode were both platinum. A platinum mesh electrode was used as a working electrode in the electrolytic reaction for observing changes in colour and absorption in the redox reaction. In the electrolysis experiment, the applied potentials were the first oxidation potential (+0.10 V) in the oxidation reaction and the first reduction potential (−0.10 V) in the reduction reaction, and the reaction was performed until no current flowed. AgTPP, 31 CuTPP, 32 FeTPPCl, 32 H2TPP, 33 MgTPP,34,35 MnTPPCl,23,32 NiTPP 32 and ZnTPP 36 were prepared according to the literature. The synthesized compounds were identified by MALDI-TOF mass measurements. In the mass measurements of FeTPPCl and MnTPPCl, these were detected as molecular ion peaks without Cl. AgTPP; found m/z = 719.72 [M + H]+, calcd for C44H28N4Ag: 719.14. CuTPP; found m/z = 675.99 [M + H]+, calcd for C44H28N4Cu: 675.16. FeTPPCl; found m/z = 668.93 [M + H]+, calcd for C44H28N4Fe: 668.17. H2TPP; found m/z = 615.84 [M + H]+, calcd for C44H30N4: 614.25. MgTPP; found m/z = 637.01 [M + H]+, calcd for C44H28N4Mg: 636.22. MnTPPCl; found m/z = 668.01 [M + H]+, calcd for C44H30N4Mn: 667.17. NiTPP; found m/z = 671.27 [M + H]+, calcd for C44H28N4Ni: 670.17. ZnTPP; found m/z = 676.99 [M + H]+, calcd for C44H28N4Zn: 676.16.
Supplemental Material
SI – Supplemental material for Changes in the absorption spectra and colour of tetraphenylporphyrins after redox reactions
Supplemental material, SI for Changes in the absorption spectra and colour of tetraphenylporphyrins after redox reactions by Kazuya Ogawa, Yuria Umetsu and Kenji Kamimura in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Grant-in-Aids for Scientific Research (C) (grant no. 18K05258) from Ministry of Education, Culture, Sports, Science and Technology, Japan (Monbu Kagakusho). MALDI-TOF measurements were carried out by the Shimadzu AXIMA-CFR, Instrumental Analysis Centre at Yokohama National University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
