Abstract
The reaction mechanism of HNCO with HO2 radicals is investigated by means of the B3PW91/6-311+G(d,p) method to determine a more reasonable pathway. Four possible entrance patterns are designed; however, only three situations are finally confirmed. When HNCO and HO2 are close to each other, they first form an intermediate

Introduction
Isocyanic acid, HNCO, is an important species for life and synthetic chemistry, since it is the simplest molecule with four elements including H, C, N, and O. As early as 1830, HNCO has been synthesized. 1 Up to now, it has been identified in various media. 2 Besides the abundance, its special importance arises from the role it plays in urban air pollution and toxicity. HNCO is a major emission from cars with gasoline and diesel engines. 3 In addition, HNCO is also a constituent of cigarette smoke, 4 which not only has negative effects on the atmosphere but also poses health risks.
Numerous theoretical and experimental studies have been performed on the reactions of HNCO. From 1840 to 1850, geometrical information on HNCO was descended in various reports,5–8 with studies have focused on its decomposition. Certainly, HNCO can react with radicals and atoms in the atmosphere, such as H, 9 O3, 10 and CH2CH. 11 However, its reaction with HO2 has never been reported. The HO2 radical is an important species in atmospheric chemistry. The subsequent reaction pathway of HO2 would affect the composition of the troposphere. Therefore, reactions involving HO2 have been extensively studied with HX (X = F, Cl, Br, I), 12 H2CO, 13 XCHO (X = F, Cl), 14 and HCNO. 15 However, to our best knowledge, the reaction of HO2 and HNCO has never been reported. Since they are abundant and important species in the atmosphere, they would also be expected to react with radicals and atoms in the atmosphere, and this aspect deserves to be elucidated. The HO2 species is theoretically studied by density functional theory (DFT). Our ultimate goal is to find the major product, which would be helpful to determine its effect in the air.
Computational details
All the quantum chemical calculations were carried out using the Gaussian 09 program. 16 The molecular geometries of the title compounds for the HNCO + HO2 reaction were fully optimized by Becke’s three-parameter exact exchange functional combined with the Perdew and Wang (B3PW91)17,18 and 6-311+G(d,p) basis sets. 19 At the same level, the vibrational frequency was calculated to confirm the nature of the stationary point and to make the zero-point energy correction. The minimum is confirmed to be all positive frequencies and the transition state has only one imaginary frequency. Starting from the transition states, the minimum energy path (MEP) was constructed from the intrinsic reaction coordinate (IRC) to ensure that two desired minima were connected with the transition state. Finally, the energy of some of the stationary points were corrected by Correlation-Coupled Cluster Theory with single and double excitations with perturbative inclusion of the triple excitation (CCSD(T)) method20,21 with the 6-311+G(d,p) 22 basis set on the basis of the optimized geometries.
Results and discussion
Six products, P1(H2NO + CO2), P2(H2O + NO + CO), P3(NO + CO2 + H2), P4(CO2 + HNOH), P5(NCOO + H2O), and P6(NCO + HOOH); 17 intermediates,


Optimized structures of the intermediates at the B3PW91/6-311+G(d,p) level.

Optimized structures of the transition states at the B3PW91/6-311+G(d,p) level.
The schematic potential energy surface of reaction HNCO + HO2 calculated at the B3PW91/6-311+G(d,p) level is shown in Figure 4, in which

Potential energy surface of the HNCO + HO2 reaction calculated at the B3PW91/6-311+G(d,p) level.
The relative energies of the reactants and products (in kcal/mol).

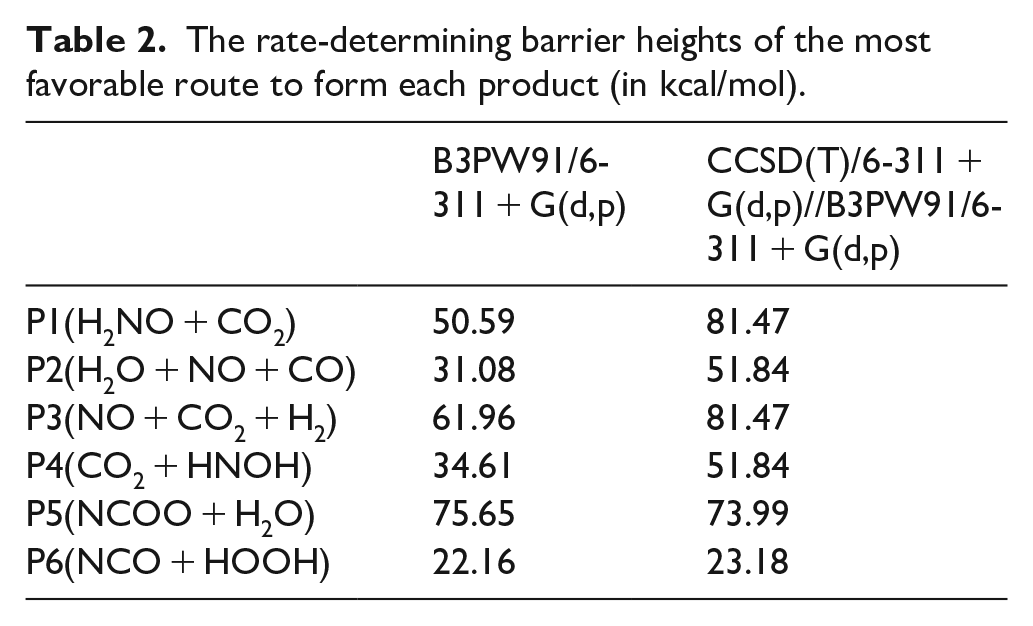
The rate-determining barrier heights of the most favorable route to form each product (in kcal/mol).
Initial process
When HNCO and HO2 are close to each other, there are several possible attaching positions. The direct hydrogen abstraction route is omitted in this article, since this pathway is simple enough and the product information is obvious. Besides the direct hydrogen abstraction route, four attaching models have been designed by us, that is, the terminal O of HO2 attaching to the C of HNCO, the middle O of HO2 attaching to the C of HNCO, and the terminal O and the middle O of HO2 attaching to the N of HNCO, respectively. Unfortunately, the pathway involving the middle O of HO2 attaching to the N of HNCO is not successfully confirmed. For other situations, there is a common property, that is, they skip a transition state to complete the attaching process. Subsequently, they would produce other intermediates and final products.
Formation of P1(H2NO + CO2)
Seventeen reaction channels are confirmed to form

The reaction routes to produce
Formation of P2 (H2O + NO + CO)
There are five routes to form
Formation of P3(NO + CO2 + H2), P4(CO2 + HNOH), and P5(NCOO + H2O)
The reaction routes to form
Formation of P6(NCO + HOOH)
As shown in Figure 6,

The reaction routes to produce
Favorable routes
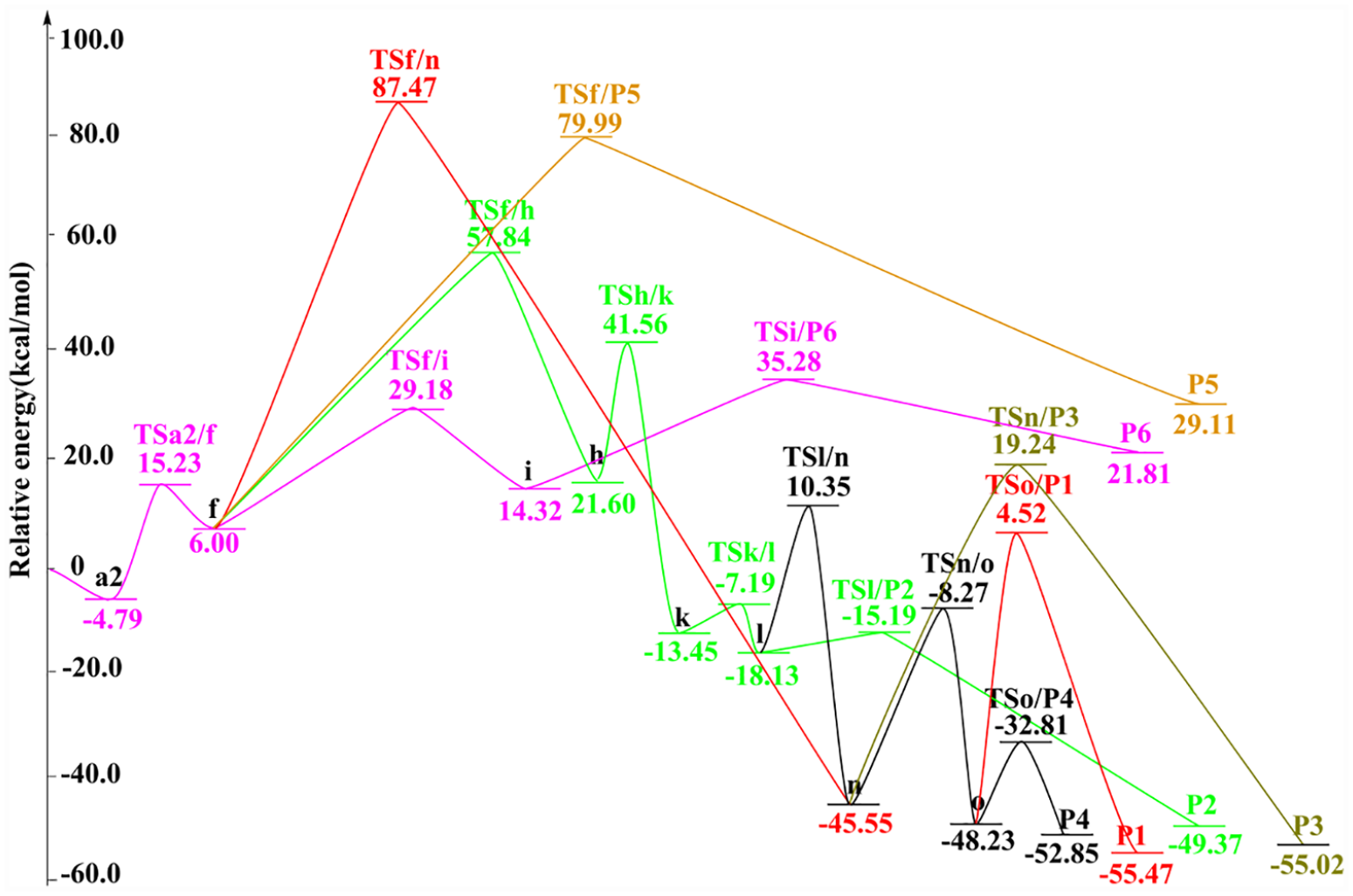
Potential energy surface of the HNCO + HO2 reaction at the CCSD(T)/6-311+G(d,p)//B3PW91/6-311+G(d,p) level.
Conclusion
In this work, the reaction mechanism of HNCO and HO2 is theoretically explored at the B3PW91/6-311+G(d,p) level. As a result, 46 pathways, 17 intermediates, 30 transitions, and 6 products are determined. It is a process without barrier height when the terminal O of HO2 attaches to the C of HNCO to form
Supplemental Material
Supporting_Information – Supplemental material for Theoretical studies of reaction mechanisms for potential reactions of HNCO with HO2 radicals
Supplemental material, Supporting_Information for Theoretical studies of reaction mechanisms for potential reactions of HNCO with HO2 radicals by Beibei An, Fuhua Huang, Ke Chen, Jiamin Jiang, Jianbin Xu and Li Wang in Journal of Chemical Research
Footnotes
Acknowledgements
We thank the National Supercomputing Center in Shenzhen (Shenzhen Cloud Computing Center) and the National Supercomputing Center in Changsha (Changsha Cloud Computing Center) for providing computational resources and software.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21476061, 21503069, and 21676071). The Key Scientific Research Plan Projects of Henan Province, China (18A150024) and the Innovation and Entrepreneurship Support Program for College Students of Minsheng College, Henan University (MSCXCY2018006).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
