General procedure for the reaction of substituted arylalkynyl Grignard reagents with enol esters
The enol ester (0.1 mol, 2.0 equiv.) was dissolved in THF (40 mL) and added dropwise to a solution of the above prepared arylalkynyl Grignard reagent. After the mixture had been stirred under a nitrogen atmosphere overnight, the reaction was quenched with saturated NH4Cl solution. The organic products were extracted with ethyl acetate. The combined organic layers were washed with brine and dried over Na2SO4. The organic phase was concentrated under reduced pressure and the resulting residue was purified by silica gel column chromatography (eluent: petroleum ether/ethyl acetate = 15:1 to 6:1) to give the title compounds.
anti-3,5-Dimethyl-1,7-diphenylhepta-1,6-diyne-3,5-diol (
anti-4aa
). Light yellow oil, yield 39%; 1H NMR (500 MHz, CDCl3): δ = 7.32 (dd, J = 7.4, 1.6 Hz, 4H), 7.16–7.15 (m, 6H), 4.40 (s, 2H), 2.25 (s, 2H), 1.64 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 131.48, 128.55, 128.27, 122.01, 92.60, 85.08, 67.18, 53.62, 31.23; high resolution mass spectrometry (HRMS) (electrospray ionization (ESI)): m/z calcd for C21H20O2 ([M + Na]+): 327.1356; found: 327.1353.
syn-3,5-Dimethyl-1,7-diphenylhepta-1,6-diyne-3,5-diol (
syn-4aa
). Light yellow oil, yield 18%; 1H NMR (500 MHz, CDCl3): δ = 7.19–7.02 (m, 10H), 4.01 (s, 2H), 2.24 (d,J = 14.6 Hz, 1H), 2.13 (d,J = 14.6 Hz, 1H), 1.60 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 131.24, 127.60, 127.55, 122.27, 91.91, 84.60, 67.39, 52.37, 32.48; HRMS (ESI): m/z calcd for C21H20O2 ([M + Na]+): 327.1356; found: 327.1352.
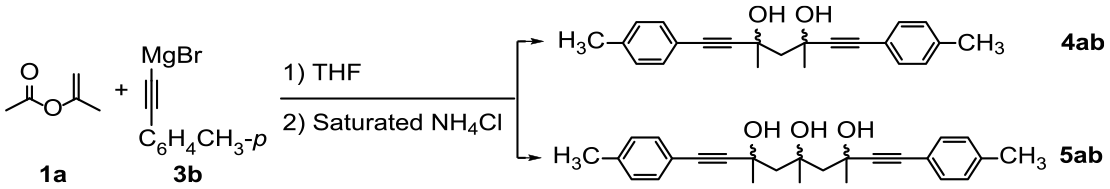
anti-3,5-Dimethyl-1,7-Di-p-tolylhepta-1,6-diyne-3,5-diol (
anti-4ab
). White solid, yield 49%, m.p. 90.1–90.4 °C; 1H NMR (500 MHz, CDCl3): δ = 7.33 (d, J = 7.9 Hz, 4H), 7.10 (d, J = 7.8 Hz, 4H), 4.49 (s, 2H), 2.34 (s, 8H), 1.74 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 138.60, 131.33, 128.97, 118.88, 91.90, 85.19, 67.10, 53.69, 31.18, 21.33; HRMS (ESI): m/z calcd for C23H24O2 ([M + K]+): 371.1408; found: 371.1410.
syn-3,5-Dimethyl-1,7-di-p-tolylhepta-1,6-diyne-3,5-diol (
syn-4ab
). White solid, yield 12%, m.p. 116.2–116.8 °C; 1H NMR (500 MHz, CDCl3): δ = 7.17 (d, J = 7.8 Hz, 4H), 6.96 (d, J = 7.7 Hz, 4H), 3.80 (s, 2H), 2.34 (d, J = 14.5 Hz, 1H), 2.30 (s, 6H), 2.21 (d, J = 14.5 Hz, 1H), 1.68 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 138.10, 131.53, 128.70, 119.52, 91.57, 85.15, 67.70, 53.10, 32.77, 21.40; HRMS (ESI): m/z calcd for C23H24O2 ([M + Na]+): 355.1669; found: 355.1667.
(anti,anti)-3,5,7-Trimethyl-1,9-di-p-tolylnona-1,8-diyne-3,5,7-triol (
anti,anti-5ab
). White solid, yield 9% m.p. 102.1–102.5 °C; 1H NMR (500 MHz, CDCl3): δ = 7.28 (d, J = 7.7 Hz, 4H), 7.04 (d, J = 5 Hz, 4H), 4.56 (s, 1H), 4.24 (s, 2H), 2.70 (d, J = 14.9 Hz, 2H), 2.31 (s, 6H), 2.07 (d, J = 14.9 Hz, 2H), 1.65 (s, 6H), 1.55 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 138.36, 131.32, 128.97, 119.35, 92.96, 84.80, 75.28, 67.38, 51.87, 32.99, 31.31, 21.38; HRMS (ESI): m/z calcd for C26H30O3 ([M + Na]+): 413.2087; found: 413.2088.
anti-1,7-Bis(4-methoxyphenyl)-3,5-dimethylhepta-1,6-diyne-3,5-diol (
anti-4ac
). White solid, yield 44%, m.p. 114.6–115.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.36 (d, J = 8.7 Hz, 4H), 6.82 (d, J = 8.7 Hz, 4H), 4.27 (s, 2H), 3.79 (s, 6H), 2.30 (s, 2H), 1.69 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 159.83, 133.01, 113.95, 91.18, 85.37, 67.01, 55.24, 54.15, 31.34; HRMS (ESI): m/z calcd for C26H30O5 ([M + Na]+): 387.1567; found: 387.1569.
(anti,anti)-1,9-Bis(4-methoxyphenyl)-3,5,7-trimethylnona-1,8-diyne-3,5,7-triol (
anti,anti-5ac
). White solid, yield 6%, m.p. 106.8–109.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.33 (d, J = 8.7 Hz, 4H), 6.76 (d, J = 8.7 Hz, 4H), 4.56 (s, 1H), 4.07 (s, 2H), 3.77 (s, 6H), 2.71 (d, J = 14.9 Hz, 2H), 2.06 (d, J = 15.0 Hz, 2H), 1.65 (s, 6H), 1.54 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 159.55, 132.91, 114.48, 113.85, 92.26, 84.58, 75.35, 67.45, 55.19, 51.82, 33.01, 31.36; HRMS (ESI): m/z calcd for C26H30O5 ([M + Na]+): 445.1985; found: 445.1990.
anti-1,7-Bis(4-chlorophenyl)-3,5-dimethylhepta-1,6-diyne-3,5-diol (
anti-4ad
). White solid, yield 21%, m.p. 90.7–90.9 °C; 1H NMR (500 MHz, CDCl3): δ = 7.34 (d, J = 8.5 Hz, 4H), 7.28 (d, J = 8.5 Hz, 4H), 4.05 (s, 2H), 2.33 (s, 2H), 1.73 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 134.69, 132.71, 128.67, 120.51, 93.55, 83.88, 67.34, 53.21, 31.20; HRMS (ESI): m/z calcd for C21H18O2Cl2 ([M + Na]+): 395.0576; found: 395.0582.
syn-1,7-Bis(4-chlorophenyl)-3,5-dimethylhepta-1,6-diyne-3,5-diol (
syn-4ad
). White solid, yield 13%, m.p. 125.4–128.3 °C; 1H NMR (500 MHz, CDCl3): δ = 7.13 (d, J = 8.5 Hz, 4H), 7.09 (d, J = 8.5 Hz, 4H), 4.00 (s, 2H), 2.29 (d, J = 14.6 Hz, 1H), 2.19 (d, J = 14.6 Hz, 1H), 1.66 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 134.15, 132.70, 128.27, 121.16, 93.27, 84.00, 67.87, 52.55, 32.86; HRMS (ESI): m/z calcd for C21H18O2Cl2 ([M + K]+): 411.0315; found: 411.0316.
(syn,anti)-1,9-Bis(4-chlorophenyl)-3,5,7-trimethylnona-1,8-diyne-3,5,7-triol (
syn,anti-5ad
). White solid, yield 9%, m.p. 161.9–163.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.36–7.27 (m, 8H), 4.93 (d, J = 7.2 Hz, 2H), 4.52 (s, 1H), 2.04 (d, J = 14.9 Hz, 3H), 1.78 (s, 1H), 1.70 (s, 3H), 1.63 (d, J = 3.1 Hz, 6H); 13C NMR (125 MHz, CDCl3): δ = 134.32, 132.53, 128.63, 121.21, 94.62, 94.51, 83.68, 83.43, 75.91, 67.82, 67.32, 53.44, 49.59, 33.96, 33.45, 29.86; HRMS (ESI): m/z calcd for C24H24O3 ([M + Na]+): 453.0995; found: 453.1030.
anti-1,7-Bis(4-fluorophenyl)-3,5-dimethylhepta-1,6-diyne-3,5-diol (
anti-4ae
). Light yellow oil, yield 40%, 1H NMR (500 MHz, CDCl3): δ = 7.43 (dd, J = 8.7, 5.4 Hz, 4H), 7.03 (t, J = 8.7 Hz, 4H), 4.57 (s, 2H), 2.40 (s, 2H), 1.81 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 162.52 (d, JC-F = 247.9 Hz), 133.35 (d, JC-F = 8.5 Hz), 118.21 (d, JC-F = 3.5 Hz), 115.16 (d, JC-F = 21.8 Hz), 92.49, 83.68, 67.37, 53.10, 31.16; HRMS (ESI): m/z calcd for C21H18O2F2 ([M + Na]+): 363.1167; found: 363.1169.
syn-1,7-Bis(4-fluorophenyl)-3,5-dimethylhepta-1,6-diyne-3,5-diol (
syn-4ae
). White solid, yield 20%, m.p. 95.9–97.8 °C; 1H NMR (500 MHz, CDCl3): δ = 7.21 (dd, J = 8.7, 5.4 Hz, 4H), 6.82 (t, J = 8.7 Hz, 4H), 4.01 (s, 2H), 2.30 (d, J = 14.6 Hz, 1H), 2.19 (d, J = 14.6 Hz, 1H), 1.66 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 163.25 (d, JC-F = 249.48 Hz), 133.40 (d, JC-F = 8.82 Hz), 118.76 (d, JC-F = 3.78 Hz), 115.16 (d, JC-F = 22.68 Hz), 91.97, 83.92, 77.25, 52.53, 32.88; HRMS (ESI): m/z calcd for C21H18O2F2 ([M − H]−): 339.1202; found: 339.1213.
(syn,anti)-1,9-Bis(4-fluorophenyl)-3,5,7-trimethylnona-1,8-diyne-3,5,7-triol (
syn,anti-5ae
). White solid, yield 5%, m.p. 157.2–159.1 °C; 1H NMR (500 MHz, CDCl3): δ = 7.38 (dt, J = 19.5, 6.7 Hz, 4H), 6.98 (t, J = 7.0 Hz, 4H), 4.98 (s, 2H), 4.60 (s, 1H), 3.11 (d, J = 14.8 Hz, 1H), 2.05–1.83 (m, 4H), 1.71 (s, 3H), 1.62 (d, J = 5.3 Hz, 6H); 13C NMR (125 MHz, CDCl3): δ = 162.48 (d, JC-F = 248.4 Hz), 133.24 (d, JC-F = 3.6 Hz), 133.17 (d, JC-F = 3.6 Hz), 118.82 (t, JC-F = 3.9 Hz), 115.65 (d, JC-F = 4.7 Hz), 115.48 (d, JC-F = 4.6 Hz), 93.38, 93.26, 83.75, 83.50, 75.89, 67.85, 67.32, 53.54, 49.71, 34.04, 33.55, 29.89; HRMS (ESI): m/z calcd for C24H24F2O3 ([M + Na]+): 421.1586; found: 421.1583.
anti-3-Methyl-1,7-diphenylhepta-1,6-diyne-3,5-diol (
anti-4ba
). White solid, yield 34%, m.p. 95.5–95.8 °C; 1H NMR (500 MHz, CDCl3): δ = 7.45 (t, J = 6.7 Hz, 4H), 7.31 (s, 6H), 5.38 (d, J = 10.7 Hz, 1H), 4.44 (d, J = 10.7 Hz, 1H), 3.55 (d, J = 19.7 Hz, 1H), 2.36–2.29 (m, 1H), 2.18 (d, J = 14.4 Hz, 1H), 1.65 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 131.68, 131.64, 128.47, 128.36, 128.24, 128.22, 122.41, 122.30, 91.18, 89.16, 85.07, 84.52, 68.58, 61.88, 48.74, 30.77; HRMS (ESI): m/z calcd for C20H15O2 ([M + K]+): 329.0938; found: 329.0942.
syn-3-Methyl-1,7-diphenylhepta-1,6-diyne-3,5-diol (
syn-4ba
). White solid, yield 22%, m.p. 91.8–92.3 °C; 1H NMR (500 MHz, CDCl3): δ = 7.43 (d, J = 7.0 Hz, 4H), 7.24–7.20 (m, 6H), 5.08–5.04 (m, 1H), 3.46 (s, 1H), 3.33 (s, 1H), 2.49 (q, J = 8.3 Hz, 1H), 2.31 (dd, J = 14.4, 4.6 Hz, 1H), 1.71 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 131.64, 131.63, 128.45, 128.40, 128.19, 122.24, 122.22, 92.03, 89.40, 85.64, 84.14, 67.26, 60.24, 49.24, 29.82; HRMS (ESI): m/z calcd for C20H15O2 ([M + K]+): 329.0938; found: 329.0932.
anti-3-Methyl-1,7-di-p-tolylhepta-1,6-diyne-3,5-diol (
anti-4bb
). White solid, yield 42%, m.p. 93.3–93.8 °C; 1H NMR (500 MHz, CDCl3): δ = 7.34 (t, J = 7.6 Hz, 4H), 7.11 (d, J = 7.6 Hz, 4H), 5.37 (d, J = 10.6 Hz, 1H), 4.39 (s, 1H), 3.47 (s, 1H), 2.34 (s, 6H), 2.29 (d, J = 10.9 Hz, 1H), 2.17 (d, J = 14.2 Hz, 1H), 1.64 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 138.56, 138.41, 131.57, 131.53, 128.94, 128.98, 119.36, 119.24, 90.52, 88.53, 85.16, 84.60, 68.58, 61.92, 48.84, 30.79, 21.38; HRMS (ESI): m/z calcd for C22H21O2 ([M + Na]+): 341.1512; found: 341.1513.
syn-3-Methyl-1,7-di-p-tolylhepta-1,6-diyne-3,5-diol (
syn-4bb
). White solid, yield 24%, m.p. 116.2–116.7 °C; 1H NMR (500 MHz, CDCl3): δ = 7.32 (d, J = 7.0 Hz, 4H), 7.08 (d, J = 7.6 Hz, 4H), 5.10 (s, 1H), 3.43 (s, 1H), 3.29 (s, 1H), 2.46 (dd, J = 14.2, 8.4 Hz, 1H), 2.33 (s, 6H), 2.28 (dd, J = 14.3, 4.1 Hz, 1H), 1.69 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 138.58, 138.51, 131.57, 131.54, 128.95, 119.16, 119.15, 91.35, 88.74, 85.80, 84.27, 67.23, 60.29, 49.42, 29.80, 21.39; HRMS (ESI): m/z calcd for C22H21O2 ([M + Na]+): 341.1512; found: 341.1514.
(anti,anti)-3-Methyl-1,9-di-p-tolylnona-1,8-diyne-3,5,7-triol (
anti,anti-5bb
). White solid, yield 5%, m.p. 123.7–124.5 °C; 1H NMR (500 MHz, CDCl3): δ = 7.28–7.22 (m, 4H), 6.98 (t, J = 8.6 Hz, 4H), 5.03 (t, J = 9.6 Hz, 1H), 4.92 (d, J = 4.2 Hz, 1H), 4.49 (s, 1H), 4.24 (s, 1H), 3.83 (s, 1H), 2.31 (d, J = 5.4 Hz, 6H), 2.08–2.03 (m, 1H), 1.96 (t, J = 14.0 Hz 1H), 1.89–1.85 (m, 1H), 1.78 (d, J = 14.1 Hz, 1H), 1.60 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 138.33, 138.17, 131.59, 128.92, 119.39, 119.29, 91.09, 88.47, 85.58, 84.26, 69.21, 68.99, 61.54, 47.95, 42.77, 31.36, 21.41; HRMS (ESI): m/z calcd for C24H26O3 ([M + Na]+): 385.1774; found: 385.1773.
(syn,syn)-3-Methyl-1,9-di-p-tolylnona-1,8-diyne-3,5,7-triol (
syn,syn-5bb
). White solid, yield 3%, m.p. 106.1–112.4 °C; 1H NMR (500 MHz, CDCl3): δ = 7.32–7.28 (m, 4H), 7.08–7.02 (m, 4H), 4.95–4.90 (m, 1H), 4.70 (d, J = 13.2 Hz, 1H), 4.25–4.08 (m, 2H), 3.84 (s, 1H), 2.32 (d, J = 3.6 Hz, 6H), 2.20–1.88 (m, 4H), 1.67–1.59 (m, 3H); 13C NMR (125 MHz, CDCl3): δ = 138.45, 130.30, 131.53, 128.91, 119.47, 119.11, 92.15, 88.89, 85.10, 83.80, 67.71, 66.77, 60.88, 48.36, 43.24, 29.66, 21.35; HRMS (ESI): m/z calcd for C24H26O3 ([M + Na]+): 385.1774; found: 385.1773.
anti-1,7-Bis(4-methoxyphenyl)-3-methylhepta-1,6-diyne-3,5-diol (
anti-4bc
). White solid, yield 35%, m.p. 82.7–83.0 °C; 1H NMR (500 MHz, CDCl3): δ = 7.36 (t, J = 7.5 Hz, 4H), 6.81 (d, J = 8.1 Hz, 4H), 5.35 (d, J = 10.6 Hz, 1H), 4.40 (s, 1H), 3.78 (s, 6H), 3.55 (s, 1H), 2.31–1.98 (m, 2H), 1.62 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 159.63, 159.54, 133.11, 133.70, 114.54, 114.40, 113.86, 113.82, 89.85, 87.90, 84.92, 84.35, 68.58, 61.93, 55.19, 48.89, 30.84; HRMS (ESI): m/z calcd for C20H15O2 ([M + Na]+): 373.1410; found: 373.1407.
syn-1,7-Bis(4-methoxyphenyl)-3-methylhepta-1,6-diyne-3,5-diol (
syn-4bc
). White solid, yield 17%, m.p. 105.5–109.0 °C; 1H NMR (500 MHz, CDCl3): δ = 7.33 (d, J = 6.7 Hz, 4H), 6.78 (d, J = 8.1 Hz, 4H), 5.09 (s, 1H), 3.77 (s, 6H), 3.59 (s, 1H), 3.47 (s, 1H), 2.44 (dd, J = 14.3, 8.3 Hz, 1H), 2.26 (dd, J = 14.3, 4.6 Hz, 1H), 1.66 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 159.60, 159.55, 133.11, 133.07, 114.34, 114.31, 113.79, 90.73, 88.13, 85.52, 83.96, 67.21, 60.25, 55.16, 49.43, 29.83; HRMS (ESI): m/z calcd for C24H27O4 ([M + Na]+): 373.1410; found: 373.1415.
anti-1,7-Bis(4-chlorophenyl)-3-methylhepta-1,6-diyne-3,5-diol (
anti-4bd
). White solid, yield 38%, m.p. 120.2–121.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.33 (t, J = 8.5 Hz, 4H), 7.25 (d, J = 6.8 Hz, 4H), 5.30 (d, J = 11.0 Hz, 1H), 4.17 (s, 1H), 3.27 (s, 1H), 2.27 (q, J = 3.2 Hz, 1H), 2.14 (d, J = 14.35 Hz, 1H), 1.61 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 134.68, 134.53, 132.88, 128.62, 120.84, 120.70, 92.05, 89.91, 84.13, 83.54, 68.59, 61.90, 48.60, 30.80; HRMS (ESI): m/z calcd for C20H16O2Cl2 ([M + Na]+): 381.0420; found: 381.0423.
syn-1,7-Bis(4-chlorophenyl)-3-methylhepta-1,6-diyne-3,5-diol (
syn-4bd
). White solid, yield 27%, m.p. 136.4–136.9 °C; 1H NMR (500 MHz, CDCl3): δ = 7.27 (d, J = 8.2 Hz, 4H), 7.20 (d, J = 7.8 Hz, 4H), 5.02 (t, J = 4.3 Hz, 1H), 3.07 (s, 1H), 2.97 (s, 1H), 2.39 (q, J = 8.3 Hz, 1H), 2.22 (dd, J = 14.50, 4.4 Hz, 1H), 1.20 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 134.63, 132.89, 128.63, 120.71, 92.91, 90.21, 84.64, 83.21, 67.34, 60.32, 49.04, 29.95; HRMS (ESI): m/z calcd for C20H16O2Cl2 ([M + Na]+) 381.0420; found: 381.0426.
(anti,anti)-1,9-Bis(4-chlorophenyl)-3-methylnona-1,8-diyne-3,5,7-triol (
anti,anti-5bd
). White solid, yield 10%, m.p. 143.3–143.7 °C; 1H NMR (500 MHz, CDCl3): δ = 7.27–7.22 (m, 4H), 7.16 (t, J = 8.6 Hz, 4H), 5.01 (t, J = 9.5 Hz, 1H), 4.93 (q, J = 5.1 Hz, 1H), 4.17 (s, 1H), 4.05 (s, 1H), 3.55 (d, J = 5.6 Hz, 1H), 2.10–2.05 (m, 1H), 1.98 (dd, J = 14.3, 10.6 Hz, 1H), 1.87 (ddd, J = 14.3, 5.6, 2.4 Hz, 1H), 1.79 (d, J = 14.1 Hz, 1H), 1.61 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 134.56, 134.41, 132.83, 128.59, 128.54, 120.91, 120.77, 92.78, 90.09, 84.46, 83.15, 69.31, 69.06, 61.65, 47.92, 42.56, 31.26; HRMS (ESI): m/z calcd for C22H20O3Cl2 ([M + Na]+): 425.0682; found: 425.0686.
(syn,anti)-1,9-Bis(4-chlorophenyl)-3-methylnona-1,8-diyne-3,5,7-triol (
syn,anti-5bd
). White solid, yield 7%, m.p. 114.4–115.7 °C: 1H NMR (500 MHz, CDCl3): δ = 7.34–7.27 (m, 4H), 7.26–7.21 (m, 4H), 4.94–4.90 (m, 1H), 4.68 (q, J = 7.0 Hz, 1H), 4.33 (d, J = 165.5 Hz, 1H), 3.99–3.89 (m, 1H), 3.51–3.44 (m, 1H), 2.21–1.79 (m, 5H), 1.64 (d, J = 42.7 Hz, 3H); 13C NMR (125 MHz, CDCl3): δ = 134.59, 132.85, 128.62, 120.95, 120.66, 120.59, 93.60, 92.78, 90.37, 90.05, 84.09, 82.98, 82.73, 70.53, 69.09, 67.83, 66.91, 62.36, 61.10, 48.12, 44.12, 42.85, 31.21, 29.63; HRMS (ESI): m/z calcd for C22H20O3Cl2 ([M + Na]+): 425.0682; found: 425.0685.
(syn,syn)-1,9-Bis(4-chlorophenyl)-3-methylnona-1,8-diyne-3,5,7-triol (
syn,syn-5bd
). White solid, yield 4%, m.p. 147.1–147.4 °C: 1H NMR (500 MHz, CDCl3): δ = 7.34 (d, J = 8.3 Hz, 4H), 7.30–7.23 (m, 4H), 4.91 (dt, J = 9.0, 3.6 Hz, 1H), 4.44 (t, J = 9.6 Hz, 1H), 3.95 (s, 1H), 3.53 (d, J = 3.3 Hz, 1H), 3.42 (s, 1H), 2.18 (ddt, J = 14.0, 9.5, 4.6 Hz, 2H), 2.01–1.89 (m, 2H), 1.68 (s, 3H); 13C NMR (125 MHz, dimethyl sulfoxide (DMSO)): δ = 132.80, 132.77, 128.63, 128.57, 121.38, 96.00, 93.67, 82.14, 80.91, 66.37, 65.53, 59.18, 50.87, 46.59, 30.56; HRMS (ESI): m/z calcd for C22H20O3Cl2 ([M + Na]+): 425.0682; found: 425.0654.
anti-1,7-Bis(4-fluorophenyl)-3-methylhepta-1,6-diyne-3,5-diol (
anti-4be
). White solid, yield 29%, m.p. 99.6–100.3 °C; 1H NMR (500 MHz, CDCl3): δ = 7.40 (q, J = 7.1 Hz, 4H), 6.98 (t, J = 8.1 Hz, 4H), 5.33 (d, J = 10.7 Hz, 1H), 4.44 (s, 1H), 3.66 (d, J = 3.2 Hz, 1H), 2.34–2.11 (m, 2H), 1.63 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 162.57 (d, JC-F = 248.4 Hz), 162.50 (d, JC-F = 248.3 Hz), 133.60 (d, JC-F = 2.7 Hz), 133.53 (d, JC-F = 2.7 Hz), 118.42 (d, JC-F = 3.5 Hz), 118.32 (d, JC-F = 3.5 Hz), 115.63 (d, JC-F = 6.2 Hz), 115.46 (d, JC-F = 6.1 Hz), 90.77, 88.76, 84.06, 83.53, 68.59, 61.80, 48.62, 30.76; HRMS (ESI): m/z calcd for C20H15O2 ([M − H]−): 325.1046; found: 325.1050.
syn-1,7-Bis(4-fluorophenyl)-3-methylhepta-1,6-diyne-3,5-diol (
syn-4be
). White solid, yield 20%, m.p. 104.4–104.7 °C; 1H NMR (500 MHz, CDCl3): δ = 7.37 (t, J = 7.7 Hz, 4H), 6.95 (t, J = 8.4 Hz, 4H), 5.08 (t, J = 4.0 Hz, 1H), 3.41 (d, J = 42.4 Hz, 2H), 2.47–2.43 (m, 1H), 2.27 (dd, J = 14.4, 4.3 Hz, 1H), 1.68 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 162.57 (d, JC-F = 248.6 Hz), 162.54 (d, JC-F = 248.3 Hz), 133.59 (d, JC-F = 2.7 Hz), 133.52 (d, JC-F = 2.8 Hz), 118.31 (d, JC-F = 1.6 Hz), 118.29 (d, JC-F = 1.4 Hz), 115.60 (d, JC-F = 2.2 Hz), 115.43 (d, JC-F = 1.8 Hz), 91.72, 89.03, 84.62, 83.15, 67.30, 60.23, 49.08, 29.83; HRMS (ESI): m/z calcd for C20H16O2F2 ([M + Na]+): 349.1011; found: 349.1016.
(anti,anti)-1,9-Bis(4-fluorophenyl)-3-methylnona-1,8-diyne-3,5,7-triol (
anti,anti-5be
). White solid, yield 5%, m.p. 114.6–115.1 °C; 1H NMR (500 MHz, CDCl3): δ = 7.32–7.31 (m, 4H), 6.87 (t, J = 8.6 Hz, 4H), 5.00 (t, J = 9.5 Hz, 1H), 4.92 (d, J = 4.0 Hz, 1H), 4.46 (s, 1H), 4.23 (s, 1H), 3.83 (d, J = 5.3 Hz, 1H), 2.09–2.03 (m, 1H), 1.98–1.94 (m, 2H), 1.87 (dd, J = 14.1Hz, 3.4 Hz, 1H), 1.60 (s, 3H); 13C NMR (125 MHz, CDCl3): δ = 163.46 (d, JC-F = 248.4 Hz), 162.42 (d, JC-F = 248.2 Hz), 133.54 (d, JC-F = 8.3 Hz), 118.54 (d, JC-F = 3.5 Hz), 118.42 (d, JC-F = 3.5 Hz), 115.55 (d, JC-F = 6.7 Hz), 115.37 (d, JC-F = 7.0 Hz), 91.52, 88.89, 84.43, 69.25, 68.99, 61.48, 47.92, 42.70, 31.26; HRMS (ESI): m/z calcd for C22H20O3F2 ([M − H]−): 369.1308; found: 393.11306.
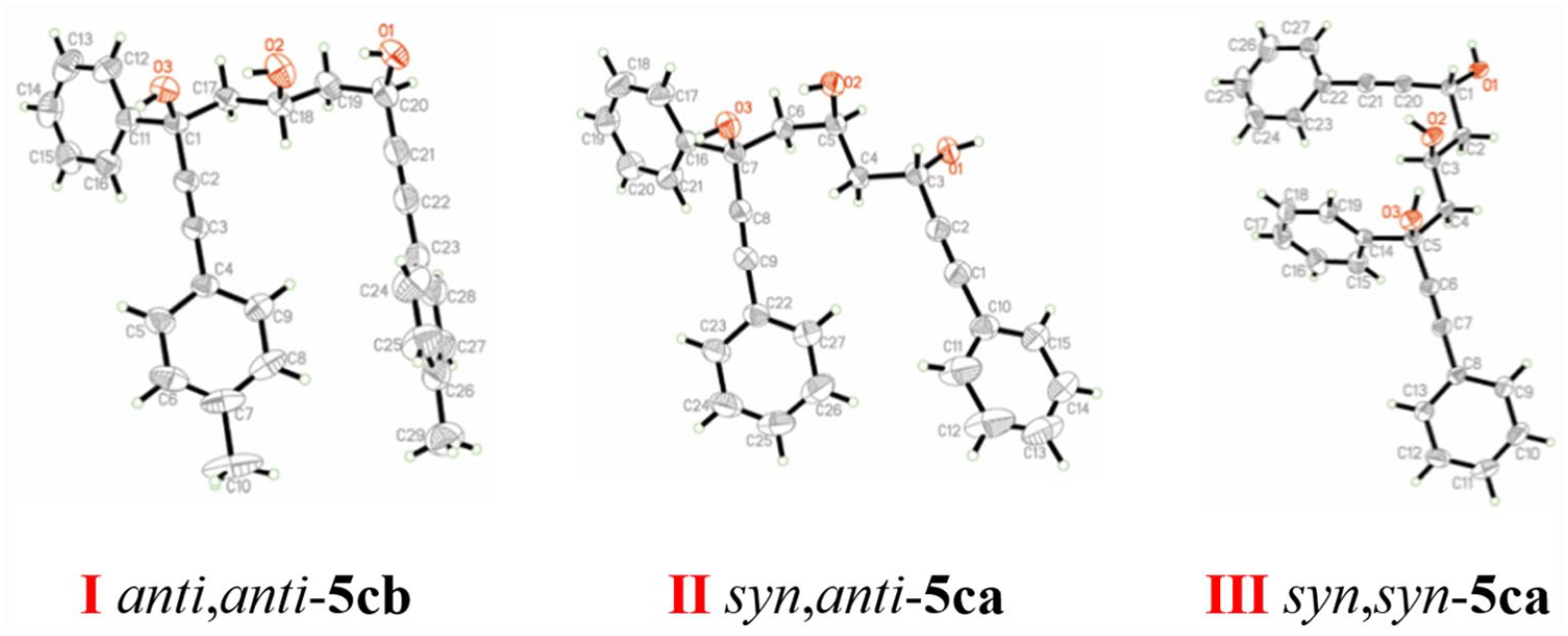
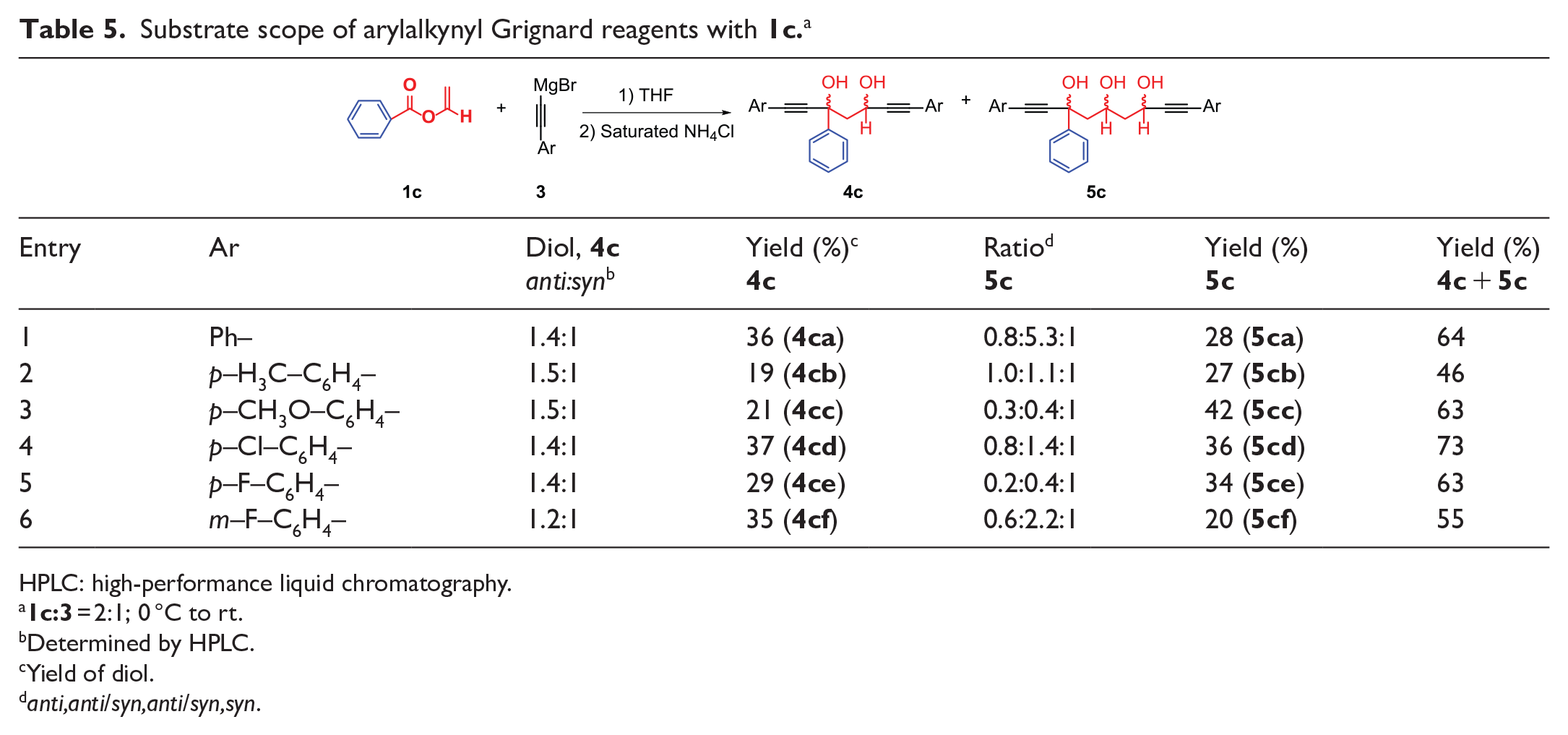
anti-1,3,7-Triphenylhepta-1,6-diyne-3,5-diol (
anti-4ca
). White solid, yield 21%, m.p. 82.1–87.9 °C; 1H NMR (500 MHz, CDCl3): δ = 7.66 (d, J = 7.3 Hz, 2H), 7.44–7.43 (m, 2H), 7.33–7.16 (m, 11H), 5.43 (d, J = 10.8 Hz, 1H), 4.33 (s, 1H), 3.27 (d, J = 3.3 Hz, 1H), 2.46–2.41 (m, 1H), 2.23 (dd, J = 14.5, 1.9 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 144.29, 131.80, 131.64, 128.69, 128.50, 128.36, 128.32, 128.24, 127.92, 125.24, 122.21, 122.18, 89.79, 88.82, 87.24, 85.24, 73.73, 62.09, 51.28; HRMS (ESI): m/z calcd for C25H20O2 ([M + Na]+): 375.1356; found: 375.1355.
syn-1,3,7-Triphenylhepta-1,6-diyne-3,5-diol (
syn-4ca
). White solid, yield 15%, m.p. 89.3–91.4 °C; 1H NMR (500 MHz, CDCl3): δ = 7.56 (d, J = 7.6 Hz, 2H), 7.32–7.11 (m, 13H), 4.62–4.58 (m, 1H), 3.81 (s, 1H), 3.03 (s, 1H), 2.66–2.61 (m, 1H), 2.40 (dd, J = 14.6, 3.3 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 143.34, 131.73, 131.65, 128.62, 128.45, 128.43, 128.23, 128.18, 127.85, 125.33, 122.20, 122.10, 91.29, 88.92, 85.82, 85.38, 72.42, 60.30, 50.97; HRMS (ESI): m/z calcd for C20H15O2 ([M + Na]+): 375.1356; found: 375.1357.
(syn,syn)-1,3,9-Triphenylnona-1,8-diyne-3,5,7-triol (
syn,syn-5ca
). White solid, yield 4%, m.p. 134.4–135.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.63 (d, J = 7.6 Hz, 2H), 7.43–7.19 (m, 13H), 4.75–4.72 (m, 1H), 4.05–4.01 (m, 2H), 3.76 (s, 1H), 3.24 (d, J = 3.2 Hz, 1H), 2.45–2.41 (m, 1H), 2.18–2.03 (m, 2H), 1.82 (d, J = 14.3 Hz, 1H), 1.65 (s, 1H); 13C NMR (125 MHz, CDCl3): δ = 144.00, 131.72, 131.67, 128.67, 128.41, 128.31, 128.21, 127.74, 125.32, 122.40, 122.15, 91.96, 89.23, 85.61, 84.97, 72.87, 68.98, 62.57, 50.83, 44.13; HRMS (ESI): m/z calcd for C27H24O3 ([M + Na]+): 419.1618; found: 419.1617.
anti-3-Phenyl-1,7-di-p-tolylhepta-1,6-diyne-3,5-diol (
anti-4cb
). White solid, yield 11%, m.p. 84.0–87.0 °C; 1H NMR (500 MHz, CDCl3): δ = 7.74 (d, J = 7.4 Hz, 2H), 7.43–7.28 (m, 7H), 7.15 (d, J = 7.9 Hz, 2H), 7.09 (d, J = 7.9 Hz, 2H), 5.51 (dt, J = 10.9, 2.6 Hz, 1H), 4.38–4.35 (m, 1H), 3.26 (s, 1H), 2.51 (dd, J = 14.7, 10.7 Hz, 1H), 2.35 (d, J = 18.5 Hz, 6H), 2.33–2.28 (m, 1H); 13C NMR (125 MHz, CDCl3): δ = 144.49, 138.85, 138.64, 131.71, 131.56, 129.08, 129.01, 128.33, 127.87, 125.28, 119.15, 89.18, 88.22, 87.42, 85.40, 73.75, 62.18, 51.45, 21.43; HRMS (ESI): m/z calcd for C27H24O2 ([M + Na]+): 403.1669; found: 403.1672.
syn-3-Phenyl-1,7-di-p-tolylhepta-1,6-diyne-3,5-diol (
syn-4cb
). White solid, yield 8%, m.p. 127.1–128.4 °C; 1H NMR (500 MHz, CDCl3): δ = 7.63 (d, J = 8.1 Hz, 2H), 7.36–7.19 (m, 7H), 7.02 (dd, J = 15.2, 7.9 Hz, 4H), 4.68 (dt, J = 8.8, 3.4 Hz, 1H), 3.77 (s, 1H), 2.98 (s, 1H), 2.68 (dd, J = 14.6, 9.2 Hz, 1H), 2.46 (dd, J = 14.6, 3.3 Hz, 1H), 2.27 (d, J = 9.0 Hz, 6H); 13C NMR (125 MHz, CDCl3): δ = 143.56, 138.79, 138.58, 131.65, 131.58, 129.00, 128.95, 128.40, 127.80, 125.36, 119.17, 119.09, 90.67, 88.33, 86.06, 85.56, 72.43, 60.39, 51.25, 21.45; HRMS (ESI): m/z calcd for C27H24O2 ([M + Na]+): 403.1669; found: 403.1669.
(syn,syn)-3-Phenyl-1,9-di-p-tolylnona-1,8-diyne-3,5,7-triol (
syn,syn-5cb
). White solid, yield 9%, m.p. 116.0–121.1 °C; 1H NMR (500 MHz, CDCl3): δ = 7.69 (d, J = 7.8 Hz, 2H), 7.36 (dt, J = 29.8, 7.4 Hz, 5H), 7.19–7.07 (m, 6H), 4.84 (s, 1H), 4.42 (t, J = 12.2 Hz, 1H), 3.66 (d, J = 11.4 Hz, 2H), 3.44 (d, J = 6.3 Hz, 1H), 2.48 (dd, J = 14.4, 9.6 Hz, 1H), 2.35 (s, 6H), 2.17 (d, J = 14.1 Hz, 1H), 2.05 (td, J = 13.8, 3.4 Hz, 1H), 1.81 (dd, J = 14.3, 4.1 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 144.08, 138.91, 138.46, 131.66, 131.62, 129.08, 128.90, 128.43, 127.73, 125.34, 118.95, 91.15, 88.50, 85.95, 85.37, 72.94, 67.21, 61.52, 50.75, 42.71, 21.48; HRMS (ESI): m/z calcd for C29H28O3 ([M + Na]+): 447.1931; found: 447.1928.
(syn,anti)-3-Phenyl-1,9-di-p-tolylnona-1,8-diyne-3,5,7-triol (
syn,anti-5cb
). White solid, yield 9%, m.p. 121.9–122.7 °C; 1H NMR (500 MHz, CDCl3): δ = 7.63 (d, J = 7.5 Hz, 2H), 7.37–7.31 (m, 5H), 7.21 (d, J = 7.8 Hz, 2H), 7.05 (t, J = 10.3Hz, 4H), 4.73 (t, J = 4.0 Hz, 1H), 4.15 (s, 1H), 4.02 (t, J = 8.9 Hz, 1H), 3.84 (s, 1H), 3.38 (s, 1H), 2.42 (dd, J = 14.3, 9.5 Hz, 1H), 2.30 (s, 6H), 2.14 (d, J = 14.4 Hz, 1H), 2.09–2.02 (m, 1H), 1.82 (s, 1H); 13C NMR (125 MHz, CDCl3): δ = 144.09, 138.76, 138.44, 131.59, 131.54, 129.02, 128.92, 128.33, 127.61, 125.32, 119.30, 119.04, 91.30, 88.55, 85.65, 85.02, 72.79, 68.94, 62.48, 50.77, 44.08, 21.41; HRMS (ESI): m/z calcd for C29H28O3 ([M + Na]+): 447.1931; found: 447.1932.
(anti,anti)-3-Phenyl-1,9-di-p-tolylnona-1,8-diyne-3,5,7-triol (
anti,anti-5cb
). White solid, yield 9%, m.p. 121.7–122.7 °C; 1H NMR (500 MHz, CDCl3): δ = 7.73 (d, J = 7.6 Hz, 2H), 7.39 (t, J = 7.5 Hz, 2H), 7.33–7.30 (m, 3H), 7.26 (d, J = 8.0 Hz, 2H), 7.00 (t, J = 7.3 Hz, 4H), 5.20 (t, J = 9.7 Hz, 1H), 4.92 (q, J = 5.1 Hz, 1H), 4.25–4.11 (m. 2H), 3.51–3.43 (m, 1H), 2.34 (s, 6H), 2.20 (dd, J = 14.3, 10.5 Hz, 1H), 2.07 (dt, J = 14.2, 3.5 Hz, 1H), 1.95–1.86 (m, 2H); 13C NMR (125 MHz, CDCl3): δ = 145.09, 138.55, 138.33, 131.71, 131.64, 128.99, 128.94, 128.32, 127.80, 125.20, 119.39, 119.21, 89.72, 88.51, 87.19, 85.64, 74.45, 69.16, 61.80, 50.90, 42.56, 21.45, 21.43; HRMS (ESI): m/z calcd for C29H28O3 ([M + Na]+): 447.1931; found: 447.1922.
anti-1,7-Bis(4-methoxyphenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
anti-4cc
). White solid, yield 13%, m.p. 150.7–151.2 °C; 1H NMR (500 MHz, CDCl3): δ = 7.70 (d, J = 7.8 Hz, 2H), 7.41 (d, J = 8.6 Hz, 2H), 7.35 (t, J = 7.6 Hz, 2H), 7.30–7.22 (m, 3H), 6.82 (d, J = 8.6 Hz, 2H), 6.77 (d, J = 8.6 Hz, 2H), 5.45 (d, J = 10.6 Hz, 1H), 4.35 (s, 1H), 3.76 (d, J = 15.1 Hz, 6H), 3.30 (d, J = 3.4 Hz, 1H), 2.46 (dd, J = 14.7, 10.6 Hz, 1H), 2.26 (dd, J = 14.7, 2.1 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 159.85, 159.71, 144.60, 133.28, 133.14, 128.31, 127.83, 125.29, 114.37, 114.34, 113.95, 113.88, 88.55, 87.62, 87.19, 85.18, 73.76, 62.18, 55.22, 51.52; HRMS (ESI): m/z calcd for C27H24O4 ([M + Na]+): 435.1567; found: 435.1567.
syn-1,7-Bis(4-methoxyphenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
syn-4cc
). White solid, yield 8%, m.p. 100.8–102.7 °C; 1H NMR (500 MHz, CDCl3): δ = 7.70 (d, J = 7.7 Hz, 2H), 7.44–7.36 (m, 4H), 7.32 (t, J = 7.3 Hz, 3H), 6.81 (dd, J = 13.5, 8.6 Hz, 4H), 4.74 (dt, J = 9.1, 3.5 Hz, 1H), 3.92–3.87 (m, 1H), 3.79 (d, J = 7.2 Hz, 6H), 3.11 (s, 1H), 2.74 (dd, J = 14.6, 9.2 Hz, 1H), 2.52 (dd, J = 14.6, 3.5 Hz, 1H);
13
C NMR (125 MHz, CDCl3): δ = 159.81, 159.67, 143.67, 133.23, 133.16, 128.38, 127.76, 125.37, 114.35, 114.23, 113.88, 113.83, 90.06, 87.71, 85.84, 85.35, 72.42, 60.40, 55.25, 55.21, 51.30; HRMS (ESI): m/z calcd for C27H24O4 ([M + Na]+): 435.1567; found: 435.1569.
(syn,syn)-1,9-Bis(4-methoxyphenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
syn,syn-5cc
). White solid, yield 24%, m.p. 103.1–105.3 °C; 1H NMR (500 MHz, CDCl3): δ = 7.66 (d, J = 7.3 Hz, 2H), 7.39 (t, J = 7.3 Hz, 4H), 7.35–7.26 (m, 3H), 6.80 (t, J = 9.2 Hz, 4H), 4.76 (dt, J = 8.8, 3.7 Hz, 1H), 4.13–4.03 (m, 2H), 3.85 (d, J = 1.6 Hz, 1H), 3.79 (d, J = 1.9 Hz, 6H), 3.29 (d, J = 3.4 Hz, 1H), 2.45 (dd, J = 14.5, 9.5 Hz, 1H), 2.19–2.07 (m, 2H), 1.85 (dt, J = 14.3, 3.2 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 159.74, 159.54, 144.14, 133.16, 133.09, 128.32, 127.59, 125.31, 114.43, 114.15, 113.86, 113.77, 90.62, 87.86, 85.42, 84.78, 72.79, 68.99, 62.53, 55.20, 50.74, 44.05; HRMS (ESI): m/z calcd for C29H28O5 ([M + Na]+): 479.1829; found: 479.1823.
(syn,anti)-1,9-Bis(4-methoxyphenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
syn,anti-5cc
). White solid, yield 11%, m.p. 143.6–144.0 °C; 1H NMR (500 MHz, CDCl3): δ = 7.72 (d, J = 7.2 Hz, 2H), 7.41–7.37 (m, 4H), 7.32 (t, J = 8.6 Hz, 3H), 6.79 (dd, J = 8.8, 3.0 Hz, 4H), 4.95–4.92 (m, 1H), 4.86 (t, J = 10.9 Hz, 1H), 4.42 (s, 1H), 4.12 (s, 1H), 3.80 (d, J = 3.7 Hz, 6H), 3.05 (d, J = 3.6 Hz, 1H), 2.17 (dd, J = 14.4, 10.3 Hz, 1H), 2.11–2.04 (m, 1H), 1.98–1.87 (m, 2H); 13C NMR (125 MHz, CDCl3): δ = 159.68, 159.60, 145.10, 133.23, 133.16, 128.29, 127.73, 125.20, 114.37, 113.87, 113.82, 89.12, 87.87, 86.76, 85.11, 74.27, 70.48, 62.39, 55.23, 55.20, 51.09, 44.21; HRMS (ESI): m/z calcd for C29H28O5 ([M + Na]+): 479.1829; found: 479.1826.
(anti,anti)-1,9-Bis(4-methoxyphenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
anti,anti-5cc
). White solid, yield 7%, m.p. 106.5–107.4 °C; 1H NMR (500 MHz, CDCl3): δ = 7.72 (d, J = 7.4 Hz, 2H), 7.41–7.25 (m, 7H), 6.71 (dd, J = 11.0, 8.5 Hz, 4H), 5.22 (t, J = 9.4 Hz, 1H), 4.91 (q, J = 5.0 Hz, 1H), 4.36 (s, 1H), 4.24 (s, 1H), 3.78 (d, J = 4.9 Hz, 6H), 3.67 (d, J = 6.0 Hz, 1H), 2.18 (dd, J = 14.4, 10.4 Hz, 1H), 2.09–2.04 (m, 1H), 1.93–1.81 (m, 2H); 13C NMR (125 MHz, CDCl3): δ = 159.64, 159.51, 145.12, 133.26, 133.18, 128.30, 127.76, 125.19, 114.54, 114.34, 113.82, 113.78, 89.04, 87.82, 86.94, 85.39, 74.46, 69.23, 61.88, 55.17, 50.86, 42.43; HRMS (ESI): m/z calcd for C29H28O5 ([M + Na]+): 479.1829; found: 479.1800.
anti-1,7-Bis(4-chlorophenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
anti-4cd
). White solid, yield 21%, m.p. 146.1–147.0 °C; 1H NMR (500 MHz, CDCl3): δ = 7.71 (d, J = 7.4 Hz, 2H), 7.46–7.22 (m, 11H), 5.46 (dt, J = 10.9, 2.4 Hz, 1H), 4.44 (s, 1H), 3.49 (d, J = 3.1 Hz, 1H), 2.53–2.48 (m, 1H), 2.30 (dd, J = 14.7, 1.9 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 144.02, 134.82, 134.63, 133.02, 132.86, 128.70, 128.63, 128.43, 128.07, 125.15, 120.63, 120.57, 90.69, 89.64, 86.16, 84.22, 73.72, 62.01, 51.08; HRMS (ESI): m/z calcd for C25H18O2Cl2 ([M − H]−): 419.0611; found: 419.0614.
syn-1,7-Bis(4-chlorophenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
syn-4cd
). White solid, yield 15%, m.p. 130.5–131.9 °C; 1H NMR (500 MHz, CDCl3): δ = 7.67 (d, J = 7.5 Hz, 2H), 7.42 (t J = 7.5 Hz, 2H), 7.37–7.33 (m, 3H), 7.29–7.23 (m, 6H), 4.69 (d, J = 9.2 Hz, 1H), 3.96 (s, 1H), 3.18 (d, J = 3.6 Hz, 1H), 2.78–2.73 (m, 1H), 2.52 (dd, J = 14.6, 3.3 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 143.06, 134.75, 134.57, 132.94, 132.85, 128.61, 128.56, 128.51, 127.99, 125.25, 120.66, 120.55, 92.22, 89.79, 84.71, 84.32, 72.45, 60.24, 50.67; HRMS (ESI): m/z calcd for C25H18O2Cl2 ([M − H]−): 419.0611; found: 419.0611.
(syn,syn)-1,9-Bis(4-chlorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
syn,syn-5cd
). White solid, yield 3%, m.p. 153.1–154.0 °C; 1H NMR (500 MHz, CDCl3): 7.67 (d, J = 7.4 Hz, 2H), 7.44–7.26 (m, 11H), 4.81–4.78 (m, 1H), 4.16–4.05 (m, 2H), 3.76 (s, 1H), 3.22 (d, J = 3.4 Hz, 1H), 2.50 (dd, J = 14.5, 9.5 Hz, 1H), 2.21 (dd, J = 14.4, 1.9 Hz, 1H), 2.14–2.09 (m, 1H), 1.87 (dt, J = 14.2, 3.1 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 143.68, 134.78, 134.52, 132.93, 132.86, 128.67, 128.58, 128.47, 127.83, 125.25, 120.81, 120.60, 92.90, 90.09, 84.33, 83.88, 72.89, 68.94, 62.50, 50.56, 43.93; HRMS (ESI): m/z calcd for C27H22O3Cl2 ([M + Na]+): 487.0838; found: 487.0840.
(syn,anti)-1,9-Bis(4-chlorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
syn,anti-5cd
). White solid, yield 5%, m.p. 155.5–156.9 °C; 1H NMR (500 MHz, CDCl3): δ = 7.67 (d, J = 7.2 Hz, 2H), 7.41–7.17 (m, 11H), 4.82 (td, J = 6.1, 3.6 Hz, 1H), 4.38 (dt, J = 9.6, 2.1 Hz, 1H), 3.76 (s, 1H), 3.60 (d, J = 1.7 Hz, 1H), 3.43 (d, J = 6.4 Hz, 1H), 2.49 (dd, J = 14.5, 9.6 Hz, 1H), 2.17 (dd, J = 14.5, 2.1 Hz, 1H), 2.08–2.02 (m, 1H), 1.79 (ddd, J = 14.4, 6.0, 2.3 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 143.67, 134.83, 134.46, 132.94, 128.69, 128.51, 127.84, 125.28, 120.87, 120.53, 92.78, 90.09, 84.49, 84.18, 72.97, 67.16, 61.43, 50.41, 42.51; HRMS (ESI): m/z calcd for C27H22O3Cl2 ([M + Na]+): 487.0838; found: 487.0842.
(anti,anti)-1,9-Bis(4-chlorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
anti,anti-5cd
). White solid, yield 3%, m.p. 125.5–126.2 °C; 1H NMR (500 MHz, CDCl3): δ = 7.67 (d, J = 7.2 Hz, 2H), 5.16 (t, J = 7.5 Hz, 2H), 7.34–7.31 (m, 3H), 7.25 (d, J = 8.2 Hz, 2H), 7.18 (t, J = 7.9 Hz, 4H), 5.16 (t, J = 10.0 Hz, 1H), 4.91 (q, J = 5.0 Hz, 1H), 4.33 (s, 1H), 4.15 (s, 1H), 3.59 (s, 1H), 2.20 (dd, J = 14.4, 10.4 Hz, 1H), 2.10–2.04 (m, 1H), 1.92 (d, J = 14.4 Hz, 1H), 1.86 (ddd, J = 14.4, 5.4, 2.5 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 144.65, 134.74, 134.55, 132.92, 132.84, 128.62, 128.59, 128.43, 127.99, 125.09, 120.84, 120.66, 91.42, 90.15, 85.90, 84.42, 74.49, 69.16, 61.76, 50.75, 42.35, 26.89; HRMS (ESI): m/z calcd for C27H22O3Cl2 ([M + Na]+): 487.0838; found: 487.0807.
anti-1,7-Bis(4-fluorophenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
anti-4ce
). White solid, yield 17%, m.p. 108.9–109.3 °C; 1H NMR (500 MHz, CDCl3): δ = 7.62 (d, J = 7.6 Hz, 2H), 7.42–7.14 (m, 7H), 6.90 (dt, J = 27.9, 8.3 Hz, 4H), 5.37 (d, J = 10.7 Hz, 1H), 4.39 (s, 1H), 3.47 (s, 1H), 2.41 (dd, J = 14.7, 10.7 Hz, 1H), 2.19 (d, J = 14.6 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.69 (d, JC-F = 249.1 Hz), 162.56 (d, JC-F = 248.3 Hz), 144.16, 133.73 (d, JC-F = 8.2 Hz), 133.56 (d, JC-F = 8.8 Hz), 128.40, 128.00, 125.17, 118.25 (d, JC-F = 3.5 Hz), 118.21 (d, JC-F = 3.6 Hz), 115.73 (d, JC-F = 10.0 Hz), 115.51 (d, JC-F = 10.6 Hz), 89.47, 88.48, 86.20, 84.24,73.70, 61.99, 51.18; HRMS (ESI): m/z calcd for C25H18O2F2 ([M − H]−): 387.1202; found: 387.1203.
syn-1,7-Bis(4-fluorophenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
syn-4ce
). White solid, yield 12%, m.p. 101.7–102.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.68 (d, J = 7.6 Hz, 2H), 7.42 (t, J = 8.0 Hz, 4H), 7.38–7.31 (m, 3H), 6.98 (dt, J = 13.9, 8.6 Hz, 4H), 4.70 (dt, J = 9.2, 3.4 Hz, 1H), 3.97–3.93 (m, 1H), 3.18–3.14 (m, 1H), 2.76 (dd, J = 14.6, 9.2 Hz, 1H), 2.52 (dd, J = 14.6, 3.3 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.66 (d, JC-F = 248.4 Hz), 162.55 (d, JC-F = 248.3 Hz), 143.21, 133.67 (d, JC-F = 8.2 Hz), 133.57 (d, JC-F = 8.4 Hz), 127.94, 125.28, 118.26 (d, JC-F = 3.6 Hz), 118.16 (d, JC-F = 3.6 Hz), 115.62 (d, JC-F = 7.2 Hz), 115.45 (d, JC-F = 6.9 Hz), 91.01, 88.57, 84.77, 84.36, 72.43, 60.25, 50.82; HRMS (ESI): m/z calcd for C25H18O2F2 ([M − H]−): 387.1202; found: 387.1203.
(syn,syn)-1,9-Bis(4-fluorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
syn,syn-5ce
). White solid, yield 22%, m.p. 147.3–147.9 °C; 1H NMR (500 MHz, CDCl3): δ = 7.66 (d, J = 7.7 Hz, 2H), 7.45–7.37 (m, 4H), 7.32 (t, J = 7.1 Hz, 3H), 6.98 (dt, J = 11.4, 8.5 Hz, 4H), 4.77 (m, 1H), 4.05 (t, J = 5.4 Hz, 2H), 3.80 (s, 1H), 3.24 (d, J = 3.3 Hz, 1H), 2.47 (dd, J = 14.5, 9.5 Hz, 1H), 2.18 (d, J = 14.4 Hz, 1H), 2.13–2.04 (m, 1H), 1.84 (d, J = 14.3 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.70 (d, JC-F = 248.3 Hz), 162.55 (d, JC-F = 248.3 Hz), 143.81, 133.66 (d, JC-F = 8.2 Hz), 133.58 (d, JC-F = 8.2 Hz), 128.45, 127.80, 125.27, 118.39 (d, JC-F = 3.6 Hz), 118.18 (d, JC-F = 3.6 Hz), 115.67 (d, JC-F= 13.4 Hz), 115.49 (d, JC-F= 13.5 Hz), 91.65, 88.82, 84.42, 83.92, 72.89, 69.01, 62.59, 50.63, 43.97; HRMS (ESI): m/z calcd for C27H22O3F2 ([M − H]−): 431.1464; found: 431.1466.
(syn,anti)-1,9-Bis(4-fluorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
syn,anti-5ce
). White solid, yield 9%, m.p. 114.2–16.8 °C;1H NMR (500 MHz, CDCl3): δ = 7.67 (d, J = 7.7 Hz, 2H), 7.44–7.38 (m, 4H), 7.33 (d, J = 7.5 Hz, 1H), 7.23 (d, J = 8.4, 5.6 Hz, 2H), 6.99–6.94 (m, 4H), 4.82 (d, J = 3.9 Hz, 1H), 4.37 (t, J = 9.4 Hz, 1H), 3.92 (s, 1H), 3.73 (s, 1H), 3.56 (d, J = 6.2 Hz, 1H), 2.51–2.45 (m, 1H), 2.16 (d, J = 14.7 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.66 (d, JC-F = 248.7 Hz), 162.49 (d, JC-F = 248.2 Hz), 143.76, 133.66 (d, JC-F = 2.5 Hz), 133.59 (d, JC-F = 2.2 Hz), 128.42, 127.72, 125.27, 118.42 (d, JC-F = 3.5 Hz), 118.12 (d, JC-F = 3.6 Hz), 115.61 (d, JC-F = 21.7 Hz), 115.41 (d, JC-F = 21.8 Hz), 91.58, 88.78, 84.45, 84.19, 72.88, 67.13, 61.31, 50.32, 42.50; HRMS (ESI): m/z calcd for C27H22O3F2 ([M − H]−): 431.1464; found: 431.1466.
(anti,anti)-1,9-Bis(4-fluorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
anti,anti-5ce
). White solid, yield 3%, m.p. 120.1–120.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.70 (d, J = 7.2 Hz, 2H), 7.39 (t, J = 7.0 Hz, 4H), 7.32 (dd, J = 8.3, 5.7 Hz, 3H), 6.90 (q, J = 6.7 Hz, 4H), 5.17 (t, J = 9.6 Hz, 1H), 4.91 (q, J = 5.6 Hz, 1H), 4.41 (s, 1H), 4.23 (s, 1H), 3.68 (d, J = 5.9 Hz, 1H), 2.19 (dd, J = 14.4, 10.4 Hz, 1H), 2.09–2.04 (m, 1H), 1.92 (d, J = 14.4 Hz, 1H), 1.86 (ddd, J = 14.4, 5.5, 2.5 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.58 (d, JC-F = 248.5 Hz), 162.53 (d, JC-F = 248.3 Hz), 144.75, 133.65 (d, JC-F = 8.4 Hz), 133.54 (d, JC-F = 8.2 Hz), 128.39, 127.93, 125.09, 118.45 (d, JC-F = 3.5 Hz), 118.28 (d, JC-F = 3.5 Hz), 115.60 (d, JC-F = 5.4 Hz), 115.42 (d, JC-F = 5.4 Hz), 90.13, 88.88, 85.90, 84.40, 74.42, 69.14, 61.67, 50.71, 42.33; HRMS (ESI): m/z calcd for C27H22O3F2 ([M + Na]+): 455.1429; found: 455.1432.
anti-1,7-Bis(3-fluorophenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
anti-4cf
). White solid, yield 19%, m.p. 87.7–89.6 °C; 1H NMR (500 MHz, CDCl3): δ = 7.68 (d, J = 1.9 Hz, 2H), 7.36 (t, J = 1.9 Hz, 2H), 7.30–7.12 (m, 6H), 7.05–6.95 (m, 3H), 5.43 (dt, J = 10.5, 2.5 Hz, 1H), 4.38 (s, 1H), 3.35 (s, 1H), 2.48 (dd, J = 14.7, 10.6 Hz, 1H), 2.27 (dd, J = 14.7, 1.9 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.29 (d, JC-F = 245.4 Hz), 162.22 (d, JC-F = 245.2 Hz), 143.97, 129.97 (d, JC-F = 8.6 Hz), 129.86 (d, JC-F = 8.6 Hz), 128.45, 128.08, 127.70 (d, JC-F = 2.5 Hz), 127.54 (d, JC-F = 2.5 Hz), 125.16, 124.00 (t, JC-F = 9.4 Hz), 118.63 (d, JC-F = 20.1 Hz), 118.46 (d, JC-F = 20.2 Hz), 116.12 (d, JC-F = 21.4 Hz), 115.93 (d, JC-F = 21.6 Hz), 90.72, 89.66, 86.00, 83.99, 73.66, 61.93, 51.07; HRMS (ESI): m/z calcd for C25H18O2F2 ([M − H]−): 387.1202; found: 387.1208.
syn-1,7-Bis(3-fluorophenyl)-3-phenylhepta-1,6-diyne-3,5-diol (
syn-4cf
). White solid, yield 16%, m.p. 141.7–142.0 °C; 1H NMR (500 MHz, CDCl3): δ = 7.71 (d, J = 7.6 Hz, 2H), 7.45 (t, J = 7.7 Hz, 2H), 7.37 (t, J = 7.3 Hz, 1H), 7.31–7.22 (m, 3H), 7.20–7.13 (m, 2H), 7.11–6.98 (m, 3H), 4.73 (dt, J = 9.0, 3.5 Hz, 1H), 3.92 (s, 1H), 3.14 (d, J = 3.7 Hz, 1H), 2.78 (dd, J = 14.6, 9.1 Hz, 1H), 2.56 (dd, J = 14.6, 3.4 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.22 (d, JC-F = 245.1 Hz), 162.19 (d, JC-F = 245.1 Hz), 143.03, 129.84 (t, JC-F = 8.5 Hz), 128.54, 128.04, 127.62 (d, JC-F = 2.4 Hz), 127.53 (d, JC-F = 2.0 Hz), 125.27, 124.04 (d, JC-F = 9.3 Hz), 123.92 (d, JC-F = 9.2 Hz), 118.59 (d, JC-F = 11.3 Hz), 118.41 (d, JC-F = 11.3 Hz), 115.96 (t, JC-F = 21.2 Hz), 92.15, 89.76, 84.64, 84.21, 72.43, 60.22, 50.64; HRMS (ESI): m/z calcd for C25H18O2F2 ([M − H]−): 387.1202; found: 387.1201.
(syn,syn)-1,9-Bis(3-fluorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
syn,syn-5cf
). White solid, yield 5%, m.p. 125.5–126.7 °C; 1H NMR (500 MHz, CDCl3): δ = 7.69 (d, J = 7.7 Hz, 2H), 7.44 (t, J = 7.6 Hz, 2H), 7.37 (t, J = 7.3 Hz, 1H), 7.33–7.24 (m, 3H), 7.17 (t, J = 7.7 Hz, 2H), 7.11–7.01 (m, 3H), 4.82–4.79 (m, 1H), 4.17 (s, 1H), 4.07 (t, J = 9.0 Hz, 1H), 3.82 (s, 1H), 3.30 (d, J = 3.4 Hz, 1H), 2.52 (dd, J = 14.5, 9.5 Hz, 1H), 2.25–2.11 (m, 2H), 1.88 (dt, J = 14.3, 3.4 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.31 (d, JC-F = 245.4 Hz), 162.27 (d, JC-F = 245.0 Hz), 143.64, 129.95 (d, JC-F = 8.6 Hz), 129.85 (d, JC-F = 8.6 Hz), 128.52, 127.91, 127.64 (d, JC-F = 2.6 Hz), 127.55 (d, JC-F = 2.6 Hz), 125.29, 124.21 (d, JC-F = 9.4 Hz), 124.02 (d, JC-F = 9.4 Hz), 118.61 (d, JC-F = 8.0 Hz), 118.43 (d, JC-F = 8.0 Hz), 116.09 (d, JC-F = 21.0 Hz), 115.85 (d, JC-F = 21.0 Hz), 92.90, 90.09, 84.22, 83.79, 72.91, 68.92, 62.45, 50.57, 43.96; HRMS (ESI): m/z calcd for C27H22O3F2 ([M − H]−): 431.1464; found: 431.1460.
(anti,anti)-1,9-Bis(3-fluorophenyl)-3-phenylnona-1,8-diyne-3,5,7-triol (
anti,anti-5cf
). White solid, yield 3%, m.p. 87.3–89.5 °C; 1H NMR (500 MHz, CDCl3): δ = 7.77 (d, J = 7.6 Hz, 2H), 7.46 (t, J = 7.6 Hz, 2H), 7.39 (t, J = 7.3 Hz, 1H), 7.28–7.16 (m, 5H), 7.15–7.04 (m, 3H), 5.22 (t, J = 9.6 Hz, 1H), 4.99 (q, J = 5.3 Hz, 1H), 4.42 (d, J = 4.0 Hz, 1H), 4.24 (s, 1H), 3.69 (d, J = 3.6 Hz, 1H), 2.27 (dd, J = 14.5, 10.4 Hz, 1H), 2.17–2.11 (m, 1H), 2.00 (d, J = 14.4 Hz, 1H), 1.94 (ddd, J = 14.5, 5.6, 2.6 Hz, 1H); 13C NMR (125 MHz, CDCl3): δ = 162.20 (d, JC-F = 245.2 Hz), 144.59, 129.85 (t, JC-F = 7.9 Hz), 128.43, 127.99, 127.64 (d, JC-F = 1.9 Hz), 127.54 (d, JC-F = 1.9 Hz), 125.06, 124.15 (d, JC-F = 9.2 Hz), 123.96 (d, JC-F = 9.2 Hz), 118.48 (d, JC-F = 10.9 Hz), 118.30 (d, JC-F = 10.92 Hz), 115.99 (d, JC-F = 21.3 Hz), 115.79 (d, JC-F = 21.3 Hz), 91.27, 90.01, 85.70, 84.25, 74.38, 69.12, 61.63, 50.67, 42.24; HRMS (ESI): m/z calcd for C27H22O3F2 ([M + Na]+): 455.1429; found: 455.1431.